Abstract
This study was performed to investigate the significance of gastric juice analysis (GJA) as a diagnostic criterion of a positive challenge in a standard oral cow's milk challenge (OCC) to confirm typical cow's milk protein-induced enterocolitis (CMPIE). Data from 16 CMPIE patients (aged 14 to 44 days) were analyzed. A standard OCC was openly executed using 0.15 g/kg of protein. Three symptoms (vomiting, lethargy, and bloody or pus-like stool), and four laboratory findings (GJA [3 hr], changes in peripheral blood absolute neutrophil count [ANC] [6 hr], C-reactive protein [6 hr], and stool smear test for occult blood or leukocytes) were observed after OCC. Before OCC, baseline studies were conducted; a stool smear test, blood sampling, and GJA. Positive OCC results were; vomiting (87.5%) (observed 1-3 hr after OCC), lethargy (62.5%) (1-3 hr), bloody or pus-like stool (43.8%) (6-10 hr), abnormal GJA (93.8%), an ANC rise >3,500 cells/µL (93.8%), and an abnormal stool smear test (75.0%). A single GJA test after a standard OCC is a sensitive diagnostic criterion of a positive challenge, and may provide an early confirmatory diagnosis of CMPIE. An investigation of positive OCC outcomes helps to find out a diagnostic algorithm of criteria of a positive challenge in CMPIE.
The diagnosis of typical cow's milk protein-induced enterocolitis (CMPIE) is generally made through clinical practice (1-3). CMPIE is a non-IgE mediated hypersensitivity disorder, and food-specific IgE test findings (e.g., skin prick test, RAST, Uni-CAP) are typically negative in this condition (1, 4). Hypothetically, the patch test may have a role in the diagnosis of gastrointestinal allergy without evidence of IgE, but it has not been sufficiently evaluated (5) and is under a careful study (6) in this disorder. Therefore, oral cow's milk challenge (OCC) remains the valid diagnostic standard for CMPIE (6, 7).
The exemplary diagnostic criteria derived from OCC in CMPIE were suggested by Powell (2, 3). These were mainly composed of vomiting and/or diarrhea that had not been present during the baseline period. However, Powell observed that 28.6% of challenges with cow's milk or soy formula did not produce vomiting in patients with CMPIE (2). Therefore, if a patient shows no vomiting or only regurgitatory driveling during the early period after OCC, a longer observation duration may be needed to determine challenge positivity. The Powell's criteria also place a focus on diarrhea and stool smear testing and require positive staining for fecal eosinophils. From the authors' experience, first diarrhea may be observed about 6-10 hr after OCC as a delayed adverse reaction. Moreover, an increase in peripheral blood absolute neutrophil count (ANC) to >3,500 cells/µL after OCC is a sensitive diagnostic criterion, but this result is obtained 6 hr after OCC (1-3). Thus, the recognition and confirmation of adverse reactions after challenge may be delayed in some CMPIE patients. Because 15-20% of reactions after OCC in CMPIE may lead to shock (1, 2), the delayed recognition of adverse reactions may induce potentially harmful clinical situations (e.g., shock and septic condition) (8).
On the other hand, high levels of tumor necrosis factor-α (TNF-α) expression with villous atrophy in duodenal mucosa (9), methemoglobinemia (10), and severe vomiting or bloody stool after OCC (1-5) are characteristic features of CMPIE. These observations indicate that massive inflammation may occur in the proximal small bowel mucosa of CMPIE patients. Projectile vomiting after OCC is thus observed to be the earliest symptom and the most important diagnostic criterion (1). Therefore, we assumed that this inflammation might be found in gastric juice analysis (GJA) at the early period after OCC.
In the present study, we investigated the significance of a GJA as a diagnostic criterion of a positive challenge for the early and confirmatory diagnosis of CMPIE in a standardized OCC. In addition, we suggested an advanced standard OCC protocol for the diagnosis of this disorder.
We analyzed the data of 16 consecutive CMPIE patients (11 males and 5 females; aged 14 to 44 days) who had been diagnosed using a standardized OCC between March 2003 and July 2006, and who exhibited vomiting, a lethargic condition, or diarrhea and who underwent a stool smear test, blood sampling, and GJA. Patients were recruited from an inpatients unit at Dongsan Medical Center, Keimyung University School of Medicine. Informed consent was obtained from all parents of children administered OCC. All study procedures were approved by the Keimyung University Institutional Review Board.
When a neonate or early infant on a cow's milk formula- or on cow's milk formula and breast mixed-fed was admitted with vomiting or diarrhea, initial clinical findings of a failure to gain weight and serum hypoalbuminemia on admission were regarded as indexes of suspicion for CMPIE (11). When feeding was possible, protein hydrolysate formula (HA®, Maeil Dairy Industry Co., Seoul, Korea) was fed before the diagnosis and electrolytes and acid-base imbalance, anemia, hypoalbuminemia, underlying infection, surgical conditions, metabolic disease, immune disorders, or other underlying diseases were corrected or excluded. When patients showed a stable general condition with good weight gain, which required about 5-7 days of hospitalization in most cases, we performed a single and open standardized OCC. The purpose of the diagnostic challenge was to predict cow's milk intolerance as early and positively as possible, while minimizing the risk of severe reactions (3). The challenge protocol (Table 1) was designed on the basis of the guidelines suggested by previous reports (1-4, 12). Challenges were performed early at 9 a.m. on a weekday under physician supervision with emergency therapies in place and an intravenous line inserted. Before OCC, baseline studies were performed, i.e., a stool smear test for occult blood or leukocytes, blood sampling for peripheral blood leukocyte count, ANC, and C-reactive protein (CRP), and GJA for leukocyte count. Patients were then given a single open OCC consisting of a total of 0.15 g of cow milk protein per kilogram of body weight, and were then observed for 4 hr in the fasting state. The challenge dose of cow's milk mixed with protein hydrolysate formula was the approximate amount of formula that a patient could consume at a single feeding (about 50-100 mL). Patients remained under physician's observation for 12 hr, regardless of the challenge outcome. When adverse reactions occurred, blood pressure monitoring and intravenous fluid therapy were conducted with fasting for about 12 hr (5). Hartmann's solution (30 mL/kg) was administered for an hour and subsequently dextrose saline was infused. Re-feeding with a protein hydrolysate formula was started at 12 hr after OCC. When adverse reactions did not develop, protein hydrolysate formula was refed at 4 hr post-OCC under careful observation for 24 hr post-OCC. When no adverse reactions occurred over 24 hr post-OCC, cow's milk formula was fed and stable general condition with no further adverse symptoms for 3 days was regarded as a negative challenge.
After OCC, seven parameters composed of clinical symptoms and laboratory findings were monitored (Table 1); three symptoms, namely, vomiting, lethargy, and diarrhea, and four laboratory tests, namely, GJA for leukocytes (3 hr), a peripheral blood leukocyte count and ANC (6 hr), CRP (6 hr), and a stool smear test for occult blood or leukocytes. Laboratory personnel were unaware of clinical challenge findings. A positive challenge was defined as the presence of two or more positive results of these seven parameters, based on and modified from the previous suggestions (1-4, 12). The early phase diagnosis was defined as a confirmatory diagnosis made within 4 hr of OCC. This was evaluated with 3 parameters composed of vomiting, lethargy, and GJA using the early phase diagnostic criteria defined as when either vomiting and lethargy or GJA was positive.
GJA data at 30 min (n=4), 1 hr (n=10), and 2 hr (n=7) after OCC were collected between March 1996 and March 1999 (unpublished data of J.B.H.) and each positive outcome to challenge was 0%, 20.0%, and 0%. Results of GJA revealed many RBCs in 1 patient and occult blood positivity in 2 patients with a negative challenge. Hence, we analyzed gastric juice 3 hr after OCC in the present study, and we assessed GJA positivity as a leukocyte count >10 cells/high-powerfield (HPF). Occult blood positivity or a rise in RBC count was excluded in the assessment of GJA, due to possible trauma resulting from tube insertion. During the fasting state after challenge, a neonatal oro-gastric feeding tube (5-Fr, Korean Medical Supply Co., Seoul, Korea) coated with jelly without a carrier fluid was smoothly inserted into the stomach. Two milliliters of air was infused, and then 1-3 mL of gastric juice was aspirated and analyzed for leukocyte count. The control group for the GJA was composed of age-matched 8 cricopharyngeal incoordination or idiopathic hypertrophic pyloric stenosis patients, who were assessed as having no inflammation in the upper gastrointestinal tract.
We excluded vague clinical symptoms and laboratory results from positive findings. A positive vomiting result was regarded as projectile vomiting, not as regurgitatory vomiting or driveling, and had to occur more than twice and absent during the baseline period. A positive lethargy result to challenge was defined as a sleepy pale or cyanotic appearance with relative insensitivity to stimulation, which had not been present during the baseline period. Lethargy in sleeping time was excluded. A positive diarrhea result was defined as a bloody and/or a pus-like stool, not observed during baseline. A relatively loose mucoid stool was not regarded as positive because patients had been fed a protein hydrolysate formula, which often results in this type of stool. A positive GJA challenge was defined as a leukocyte count exceeding 10 cells/HPF. A positive ANC challenge was regarded as a rise to >3,500 cells/µL from baseline (1-5, 7, 8), and a positive CRP challenge was defined as an increase to >1 mg/dL. A positive stool smear test was defined as the presence of occult blood and/or a leukocyte count of >5 cells/HPF, not present at baseline. Diaper rash and anal fissure were excluded as possible blood sources.
OCC was performed on 17 patients with indexes of suspicion of CMPIE during the study period and CMPIE was diagnosed in 16 (94.1%) patients. Positive results to OCC were; vomiting (87.5%) (observed 1-3 hr after OCC), lethargic condition (62.5%) (1-3 hr), a bloody or a pus-like stool (43.8%) (6-10 hr), abnormal GJA (93.8%), ANC >3,500 cells/µL (93.8%), CRP >1 mg/dL (0%), and an abnormal stool smear test (75.0%) (Table 1). No patient showed adverse reactions involving skin or respiration. GJA to challenge was positive in 15 of 16 patients; many WBCs per HPF were observed in 14 patients and 20 WBCs/HPF were present in a single patient. None of the 8 control patients showed positivity to GJA. Baseline GJA tests of OCC patients were all negative. The mean peripheral blood leukocyte count rose from 13,347±4,711 cells/µL to 24,891±16,935 cells/µL (p=0.001), and a mean rise of leukocyte count was 11,544± 13,102 (4,540-13,220) cells/µL. The mean ANC rose from 5,807±2,654 cells/µL to 18,942±13,536 cells/µL (p=0.001) with a mean rise of 12,135±10,982 (2,065-14,873) cells/µL. The mean CRP showed no significant change after OCC (before OCC 0.13±0.17 mg/dL; after 0.13±0.18 mg/dL).
All 16 patients suspected of having CMPIE were confirmingly diagnosed in the early phase after OCC. GJA to challenge was positive in 15 of 16 CMPIE patients (Fig. 1); GJA was positive in 9 of 10 patients with vomiting (+) and lethargy (+); GJA was also positive in all 4 patients with vomiting (+) and lethargy (-); moreover, GJA was positive in all 2 patients with vomiting (-) and lethargy (-). Although all 16 patients with CMPIE were diagnosed in the early phase using the early phase diagnostic criteria including GJA parameter, only 10 (62.5%) patients satisfied the diagnostic criteria without GJA (p=0.009).
Of the seven parameters monitored, CRP was excluded as a criterion because no significant change was observed after OCC. Interpretation of the remaining six parameters resulted in an OCC positivity algorithm for the diagnosis of CMPIE (Fig. 2).
Although OCC is a diagnostic standard for CMPIE, it has serious associated dangers because adverse reactions may lead to shock (1, 2, 6, 8). Thus the early recognition of positive adverse reactions after OCC is necessary to avoid the delayed diagnosis or to preclude for multiple challenges to obtain definite reactions, which may bring out clinically undesirable results (e.g., septic condition and malnutrition) in the challenged patient.
If vomiting is clearly projectile and lethargic condition involves a pale appearance with/without cyanosis during the early phase after OCC, it is not difficult clinically to establish the diagnosis of CMPIE (1). However, 10 CMPIE patients (62.5%) in our study satisfied these diagnostic criteria. If no vomiting or driveling and just a moderately sleepy condition without pale or cyanotic appearance are observed after OCC, it is difficult to regard these symptoms as definite adverse reactions. Under these clinical circumstances, GJA may be functional in a confirmatory diagnosis of CMPIE, without a baseline test, during the early phase after OCC. GJA at 3 hr after OCC was found to be a highly sensitive diagnostic criterion. Symptoms of typical CMPIE commonly start before 2 months of age and thus this disorder mainly occur in neonate or early infancy (1, 5, 11). Based on our clinical experience, we think that oro-gastric tube insertion may be less invasive in neonate or early infancy than in children, because of the relatively fine tube, decreased gag reflex (13), and short oro-gastric distance.
Peripheral blood leukocyte count and ANC increased significantly at 6 hr after OCC in all CMPIE patients of this study. In Powell's case series (2, 3), a rise in ANC of over 3,500 cells/µL was observed in all patients with a mean rise to 9,900 (5,500-16,800) cells/µL. In the present study, an ANC of >3,500 cells/µL was observed in 15 patients (92.9%), with the exception of one patient with an ANC increase of 2,065 cells/µL. In Sicherer et al.'s case series (1), an ANC to >3,500 cells/µL was observed in 9 of 10 patients (90.0%) with an average rise to 7,774 (2,449-13,538) cells/µL. Sicherer et al. also noted that in case of 8 negative challenges, ANC rose by an average of 467 (-458-1,376) cells/µL. In the clinical situation without vomiting or vague lethargy condition after OCC, an ANC rise is a highly definite diagnostic parameter. However, this criterion is confirmed 6 hr after OCC as a late phase adverse reaction (Fig. 2) and is also needed twice blood sampling including a baseline study. We think that, in neonate or early infancy, oro-gastric tube insertion seems to be less invasive than twice blood drawing for ANC. Therefore, when vomiting or lethargy after OCC is not apparent or vague, a single GJA without a baseline study is a sensitive criterion and may induce an early phase diagnosis of CMPIE (Fig. 1).
We also suggest an advanced standard OCC protocol in CMPIE. The recommended amount of cow's milk protein administered for OCC has been 0.6 g protein per kilogram of body weight (2-4, 12). After noting reactions at lower doses, a lower total amount of protein (0.15-0.3 g protein per kilogram of body weight or lower) was recommended, particularly when the infant concerned has a history of a severe reaction after a small ingestion (1, 14). Our standardized OCC was performed using only 0.15 g of cow's milk protein per kilogram of body weight. Nevertheless, this dose proved to be sufficient to induce symptoms in all our CMPIE patients and the results obtained using a single open challenge allowed a diagnosis of CMPIE in all cases. In a negative challenged patient with the 3-day cow's milk reintroduction period, there were no adverse reactions identified. Moreover, our standardized OCC protocol consists of early protein hydrolysate formula feeding, and later, when the patient attains a stable general condition, we perform OCC for CMPIE diagnosis. These approaches may allow the patient with indexes of suspicion of CMPIE (11) to safely progress to diagnosis. However, it should always be borne in mind that OCC in CMPIE has attendant dangers due to the possibility of an overwhelming bowel inflammation (9, 10).
Before OCC, emergency facilities and therapies should be in place and considerations of intravenous access are mandatory for the management of possible adverse reactions, particularly of hypotension or shock (5, 7). About 50% of positive challenges require physician-supervised treatment (5). Hence, in neonates or early infants with suspicion of CMPIE, OCC must be performed in an inpatient unit under physicians observation. No patient in the present study developed hypotension or shock, perhaps due to our early fluid resuscitation protocol and a lower challenge dose. We performed OCC in the early morning on weekdays to ensure the availability of careful observation and appropriate emergency treatment. If vomiting or lethargy develops as adverse reactions, intravenous fluid therapy is mandatory to prevent shock or dehydration. In our experience, vomiting and lethargy may persist for about 8-12 hr in a few patients. Hence, our fasting protocol after OCC is extended to 12 hr when a patient shows an adverse reaction.
We suggest advanced diagnostic criteria in combination with a single open standard OCC protocol that would provide the confirmatory diagnostic approach to CMPIE. A diagnostic algorithm composed of vomiting, a lethargic condition, and GJA induced by a standardized OCC may provide an early phase diagnosis of CMPIE. When vomiting or lethargy after OCC is not apparent or vague, a single GJA without a baseline study after a standard OCC is a sensitive diagnostic criterion of a positive challenge, and may provide an early phase diagnosis of CMPIE.
Figures and Tables
Fig. 1
Results of the early phase diagnosis* evaluated with 3 parameters composed of vomiting, lethargy, and gastric juice analysis (GJA) in typical cow's milk protein-induced enterocolitis (CMPIE). *, When either vomiting and lethargy or GJA was positive within 4 hr after oral cow's milk challenge, we defined it as the early phase diagnosis.
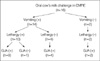
Fig. 2
Algorithm after oral cow's milk challenge for the diagnosis* of typical cow's milk protein-induced enterocolitis. *, We defined a positive challenge as the presence of two or more positive results of the six parameters and the early phase† diagnosis as a diagnosis made within 4 hr of an oral cow's milk challenge using the early phase diagnostic criteria defined as when either vomiting and lethargy or gastric juice analysis was positive. ANC, absolute neutrophil count.

References
1. Sicherer SH, Eigenmann PA, Sampson HA. Clinical features of food protein-induced enterocolitis syndrome. J Pediatr. 1998. 133:214–219.

2. Powell GK. Milk- and soy-induced enterocolitis of infancy. Clinical features and standardization of challenge. J Pediatr. 1978. 93:553–560.
3. Powell GK. Food protein-induced enterocolitis of infancy: Differential diagnosis and management. Compr Ther. 1986. 12:28–37.
4. Burks AW, Casteel HB, Fiedorek SC, Williams LW, Pumphrey CL. Prospective oral food challenge study of two soybean protein isolates in patients with possible milk or soy protein enterocolitis. Pediatr Allergy Immunol. 1994. 5:40–45.

5. Sicherer SH. Food protein-induced enterocolitis syndrome: case presentations and management lessons. J Allergy Clin Immunol. 2005. 115:149–156.

6. Fogg MI, Brown-Whitehorn TA, Pawlowski NA, Spergel JM. Atopy patch test for the diagnosis of food protein-induced enterocolitis syndrome. Pediatr Allergy Immunol. 2006. 17:351–355.

7. Sicherer SH. Food allergy: when and how to perform oral food challenges. Pediatr Allergy Immunol. 1999. 10:226–234.

8. Sicherer SH. Clinical aspects of gastrointestinal food allergy in childhood. Pediatrics. 2003. 111:1609–1616.
9. Chung HL, Hwang JB, Park JJ, Kim SG. Expression of transforming growth factor beta1, transforming growth factor type I and II receptors, and TNF-alpha in the mucosa of the small intestine in infants with food protein-induced enterocolitis syndrome. J Allergy Clin Immunol. 2002. 109:150–154.
10. Murray KF, Christie DL. Dietary protein intolerance in infants with transient methemoglobinemia and diarrhea. J Pediatr. 1993. 122:90–92.

11. Hwang JB, Lee SH, Kang YN, Kim SP, Suh SI, Kam S. Indexes of suspicion of typical cow's milk protein-induced enterocolitis. J Korean Med Sci. 2007. 22:993–997.

12. McDonald PJ, Goldblum RM, Van Sickle GJ, Powell GK. Food protein-induced enterocolitis: altered antibody response to ingested antigen. Pediatr Res. 1984. 18:751–755.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download