Abstract
Recently diffuse large B cell lymphoma (DLBCLs) was reported to be subdivided into germinal center B-cell-like (GCB) and activated B-cell-like (ABC) subgroups by using cDNA microarray and immunohistochemical markers. Tissue microarray blocks were created from 51 nodal DLBCLs with c ntrol tissue. Immunohistochemical staining for the above markers were performed. The median follow-up period was 26 months. Nodal DLBCLs were subclassified into GCB [CD10+ or CD10-/Bcl-6+/MUM1-, n=17 (33%)] and non-GC subgroups [CD10-/Bcl-6- or CD10-/Bcl- 6+/MUM1+, n=35 (67%)], and were alternatively subclassified into pattern A [+ for GCB marker only, n=12 (23%)], B [Co-positive for both markers, n=13 (33%)], C [+ for activation marker only, n=18 (35%)], and D [- for both markers, n=9 (17%)]. Upon survival analysis, the GCB groups showed a relatively better survival than non-GC groups (p=0.0748). Also, pattern C (p=0.0055) and CD138+ (p=0.0008) patients had significantly lower survival rates. By multivariate analysis, CD138 expression alone was considered as an independent risk factor (p=0.031). In summary, our results add to the registration of prognostic implications for previously reported DLBCL subgroups. CD138 may play an important role as a poor prognostic marker. By using immunohistochemistry, a prognostically important subclassification of DLBCLs is possible.
Diffuse large B cell lymphoma (DLBCL) is the most common type of non-Hodgkin's lymphoma in Western countries, and accounts for approximately 60% of patients with B-cell lymphomas in East Asia (1,2). Although these tumors are designated as a single disease entity by the World Health Organization (WHO), the diversity of clinical presentations and pathologic, genetic, and molecular characteristics strongly suggest that these neoplasms represent a eterogenous group of tumors (3). Despite the use of anthracyclin-based chemotherapy, long-term disease-free survival can only be achieved in about 40% of patients (1). Therefore, it is important to identify the patients who may benefit from more aggressive or experimental therapies at diagnosis.
Alizadeh et al. recently reported that DLBCL can be divided into prognostically significant subgroups with germinal center B-cell-like (GCB), activated B-cell-like (ABC), or type 3 gene expression profiles using cDNA microarray (3). The GCB group had a significantly better survival rate than the ABC group (3). The type 3 group was heterogeneous and not well defined, but had a poor outcome similar to the ABC group (3). Their results have been confirmed by another study demonstrating that the gene expression profiles predict the survival of DLBCL patients after chemotherapy (4).
More recently, there have been several studies subdividing DLBCLs into prognostically important subgroups by using an immunohistochemical panel (5-9). However, the resulting data have been controversial, with several studies showing a significantly better survival rate for the GCB group and others finding no difference in survival between the GCB and non-GC groups (5-9).
The aim of this study was to investigate the expression of CD10, Bcl-6, MUM1, CD138, and Bcl-2 in nodal DLBCLs, and to analyze the relationship between immunohistochemical profile and outcome in nodal DLBCLs. Thus, we also evaluated the use of an immunohistochemical profile to subdivide DLBCLs into prognostically significant subgroups by using germinal center B-cell (CD10 and Bcl-6) and activation (MUM1 and CD138) markers with a tissue microarray (TMA).
The study group consisted of 51 patients with de novo nodal DLBCLs including five patients with de novo tonsillar DLBCLs diagnosed at Hanyang University Medical Center from 1995 to 2002, and classified according to WHO criteria based on morphological examination of imprints, paraffin sections, and immunophenotyping.
Hematoxylin and eosin-stained sections from each paraffin-embedded, formalin-fixed block were used to define diagnostic areas. In addition, two random, representative 0.6 mm cores were obtained from each case and inserted in a grid pattern into a recipient paraffin block using a tissue arrayer (Beecher Instruments, Silver Spring, MD, U.S.A.). For the control group, three cases of follicular lymphoma and three cases of reactive tonsil were included in each TMA block. Four-µm sections were then cut from each TMA block and stained with antibodies to CD10, Bcl-6, MUM1, CD138, Bcl-2, and MIB1, as listed in Table 1, using the avidin-biotin method. Each core was evaluated independently by two pathologists for the percentage of tumor cells stained by visual estimation, and recorded in 10% increments. Disagreements were resolved by joint review on a multihead microscope. For each case, the core with the highest percentage of stained tumor cells was used for analysis. For CD10, Bcl-6, Bcl-2, MUM-1 and CD 138, cases were considered positive if 30% or more of the tumor cells were stained with an antibody based on previous studies.
Immunoperoxidase results for CD10, Bcl-6, MUM1, and CD138 were used to subclassify the patients. We divided the DLBCLs into subgroups according to two different methods proposed by Hans et al. and Chang et al., which are shown in Table 2 (8,9). According to Hans et al., patients were separated into GCB and non-GC groups (8). If CD10 was positive, regardless of Bcl-6, MUM-1 or Bcl-6 status, DLBCLs were subclassified as GCB. The remaining patients were classified as non-GC. However, according to Chang et al. method, the cases could be subclassified into four patterns: positive GCB marker only (A), positive GCB and activation marker (B), positive activation marker only (C), and all negative (D) (9).
The Kaplan-Meier method was used to estimate overall survival distributions. Overall survival was calculated from the time from diagnosis to the date of death or last contact. Patients who were alive at last contact were censored in the overall survival analysis. The log-rank test was used to compare the clinical characteristics between the TMA subgroups. Univariate and multivariate analysis was performed using the Cox regression method. Stepwise selection was used to determine variables that were independent predictors of overall survival. The SPSS 11.0 Statistical software (U.S.A.) program was used for data analysis. A value of p<0.05 was considered as significant and 0.05≤p<0.10 as relatively significant.
Clinical data are summarized in Table 3, which represent patients in GCB and non-GC subgroups. Clinical data for survival analysis were available for 51 patients with a minimum follow up period of 16 months. There were 30 males and 21 females with a median age of 59 yr (age range, 19-83 yr). The median follow up period was 26 months. The clinical features of the two DLBCL subgroups (GCB vs. non-GC) did not differ in any regard. Similarly, of the four patterns (A, B, C, and D) observed, clinical parameters did not differ (data not shown).
Out of the total 51 patients, 11 (22.0%, except for one undetermined patient) were positive for CD10, 20 (39.2%) for Bcl-6, 23 (45.1%) for Bcl-2, 16 (31.4%) for MUM1, and 8 (15.7%) for CD138. When the patients were subdivided according to the two different methods stated above, 21 patients (42.0%, except for one patient with undetermined CD10 status) were subclassified as GCB and 29 (58.0%) as non-GC. Using the four group classification, 17 patients (33.3%) were subclassified as pattern A, seven (13.7%) as B, 14 (27.5%) as C, and 13 (25.5%) as D.
Tumor expression of CD10 was associated with better overall survival, which was relatively significant (p=0.0992, Fig. 1A). On the other hand, tumor expression of Bcl-6 also tended to convey better survival, but not to a significant level (p=0.2509, Fig. 1B). MUM1 expression did not result in any difference in overall survival between the two groups (p=0.5207, Fig. 2A). In contrast, CD138 positive patients showed a strikingly worse overall survival rate, although there were only eight patients in total (p=0.0008, Fig. 2B). No significant difference in survival was found between the Bcl-2 positive and negative groups (p=0.5307, not shown). In view of the International Prognostic Index, the low IPI group showed a better survival, which was relatively significant in our study (p=0.0978, data not shown).
When we divided patients into GCB and non-GC subgroups, the GCB subgroup showed a better survival rate than the non-GC subgroup at a relatively significant level (p=0.0748, Fig. 3A). When separately considering patients with low or high IPI scores, the GCB groups also had a better survival rate than the non-GC subgroup with low (p=0.5307, Fig. 3B) and high IPI scores (p=0.1534, Fig. 3C). On the other hand, when we divided the patients into patterns A, B, C, and D, pattern C showed a strikingly poor survival. In contrast, there were no remarkable differences among patterns A, B, and D. When we divided them into two groups, pattern C had a significantly lower survival rate compared to pattern A or B (Fig. 4A). Furthermore, when separately considering those patients with low (Fig. 4B) or high IPI scores (Fig. 4C), four pattern C patients with high IPI scores had a strikingly lower survival than others.
By univariate Cox proportional hazards regression analysis, our results showed that expression of the individual CD138 marker and pattern C was associated with an increased relative risk of death in DLBCL patients (Table 4). Moreover, CD 10 expression, GCB subgroup classification, and IPI scores made relatively significant impacts on survival. Most importantly, using a multivariate Cox proportional hazards regression analysis, only the expression of CD138 alone was statistically significant as an independently poor prognostic factors, controlling for IPI status (Table 5).
CD138 was expressed in eight of the 51 DLBCL patients. The clinical and immunohistochemical characteristics of all eight patients with CD138 expression are summarized in Table 6. Six patients were negative for germinal center B cell markers. However, one patient was positive for both CD10 and Bcl-6 (Fig. 5), and another patient for Bcl-6 alone. These patients were classified into subgroups B and GCB, respectively. The first 80-yr-old male patient expired six months later. In contrast, the second 28-yr-old female patient, who had received bone marrow transplantation, was still alive at follow up 68 months later. The remaining six patients were subclassified as pattern C and non-GCB. These patients died in a relatively short period. Most of the patients with CD138 expression showed generally high Ki-67 labeling indices.
TMA is a useful and cost-effective tool that allows rapid evaluation of immunohistochemical staining of a large number of tumors simultaneously (10). TMA appears to be particularly useful for the immunohistochemical characterization of malignant lymphomas (10). The TMA immunostaining results have been shown to agree with whole tissue section staining in 86% to 100% of patients (11). When compared with whole section immunohistochemistry, TMA demonstrates superior immunostaining consistency between cases because most cases are located on the same TMA section (10). Quantitation of the staining results is also easier because each tissue core can be completely viewed under one intermediate-power microscopic field. Furthermore, the use of TMA preserves the tissue in the paraffin blocks for future studies. In the present study, we selected two different, relatively well preserved and representative areas from each tumor for use in the TMA blocks. Thus, we could easily and consistently evaluate the immunohistochemical staining results.
The diversity in the clinical presentation, morphology, immunophenotype, and genetic and molecular alterations strongly suggests that DLBCL is a heterogenous group of B-cell lymphomas rather than a single clinicopathologic entity (12-14). The primary site of the lymphoma, either the lymph node or different extranodal territories, has been suggested as a criterion that might distinguish the two different groups of DLBCL, nodal and extranodal, with particular clinicobiological characteristics and different natural history (15,16). However, the current classification of lymphomas is largely based on the clinicopathologic features and, at present, does not take into consideration the primary site of the lymphoma; it is simply regarded as additional information (12). In the present study, 69 patients with primary de novo nodal DLBCLs were selected for a uniform study population. In general, Waldeyer's ring, whose lymphoid tissue is similar to that of the lymph node, is currently included among nodal areas (16). Therefore, we also included seven DLBCLs of the Waldeyer's ring in the present study.
CD10 is a human membrane associated neutral endopeptidase, and antibodies against CD10 embedded in paraffin sections are available (17). CD10 is expressed in a variety of human tissues, but its expression is restricted in the germinal center cells of reactive lymphoid tissues (17). There have been several studies examining CD10 expression in DLBCLs by immunohistochemistry, but the resulting data are conflicting (6-9,18-24). Chang et al. and Ohshima et al. have reported that CD10 expression is associated with a better DLBCL prognosis (9,19). In contrast, Uherova et al. and Xu et al. have suggested that CD10 expression is associated with poor outcome (21,24). In the present study, patients with CD10 expression showed a better survival rate at a marginally significant level (p=0.0992).
Bcl-6 is a zinc-finger protein that acts as a transcriptional repressor and is expressed in germinal center B cells and a subset of CD4+ T cells (25,26). Immunohistochemical studies of Bcl-6 expression and its relationship to DLBCL patient outcomes are limited in number. Hans et al. reported Bcl-6 expression evaluated by immunohistochemistry to be associated with a better prognosis (8). Likewise, other studies have reported that Bcl-6 expression predicts a better overall survival, whereas another study found no difference in overall survival related as to Bcl-6 expression (6,8,9,27). Our results demonstrated that patients with Bcl-6 expression tended to show better survival, but not at a statistically significant level (p=0.2509).
Regarding the prognostic value of Bcl-2 expression in DLBCLs, most studies have shown no difference in prognosis between Bcl-2 positive and negative patients, whereas a recent study by Biasoli et al. suggested that Bcl-2 expression, especially in the high IPI group, is associated with a significantly decreased survival (8,22,28,29). Our results show no difference in survival between Bcl-2 positive and negative groups, similar to the results of most previous studies, except for Biasoli et al. (22).
MUM1/IRF-4 (multiple myeloma-1/interferone regulatory factor-4) is a lymphoid specific member of the interferon regulatory factor family of transcription factors, and is normally expressed in plasma cells and a minor subset of germinal center cells (30,31). Studies have shown that MUM1 expression may denote the late stage of germinal center B-cell differentiation (3,30,31). Moreover, Hans et al. reported that MUM1 expression identified non-GCB patients (8). Thus, we used MUM1 as an activated B cell marker in the present study. Prognostic significance of MUM1 expression in DLBCL patients has been limited. Hans et al. and Chang et al. reported that expression of MUM1 is associated with a significantly worse survival (8,9). However, our results demonstrate no difference in survival between MUM1 positive and negative groups.
CD138, also known as syndecan-1, is expressed in antigen-stimulated B cells and denotes terminal differentiation toward plasma cells (32). The clinical significance of CD138 expression in DLBCL patients has not been well documented. In the present study, patients with CD138 expression showed a strikingly worse survival. However, there were only eight CD138 positive cases.
Recently, there have been several efforts to classify DLBCL patients into prognostically implicated subgroups using cDNA microarray and immunohistochemical expression profiles (3,6-9). However, the methods remain controversial. As a part of the efforts to classify DLBCL patients into prognostically implicated subgroups, the present study used an immunohistochemical panel of GCB-cell and activation markers as stated above, such as CD10, Bcl-6, MUM1, and CD 138, on paraffin-embedded tissues. Hans et al. reported that the expression of CD10, Bcl-6, and MUM1 can be combined to divide DLBCL patients into GCB and non-GC subgroups with outcomes similar to that predicted by cDNA microarray analysis as they have previously reported (8). Chang et al. also reported that the expression patterns of CD10, Bcl-6, MUM1/IRF4, and CD138 of GCB cell and activation markers by immunohistochemistry correlate with the prognosis of DLBCL patients (9). Similarly, Biasoli et al. reported that the expression of CD10 could predict a favorable outcome, especially in the low-risk IPI group, and the expression of Bcl-2 was an independent poor prognostic factor (22). In contrast, Colomo et al. reported that a differentiation profile using CD 10, Bcl-6, MUM1, and CD138 was associated with particular clinicopathological features, but was not essential for predicting DLBCL patient outcome (6). More recently, Fabiani et al. reported that CD10 expression in DLBCLs did not influence survival (23). In the present study, patients in the GCB group showed a relatively better overall survival rate than those in the non-GC subgroup (p=0.0748). Similarly, patients subclassified as pattern C (CD10-, Bcl-6-/MUM1+ or CD138+) showed a significantly lower survival rate than pattern A (CD 10+ or Bcl-6+/MUM1-, CD138-) or B patients (CD10+ or Bcl-6+/MUM1+ or CD138+) (p=0.0055). Our results further support that immunohistochemical expression profiles using CD10, Bcl-6, MUM1, and CD138 could predict DLBCL patient outcome.
More interestingly, patients with CD138 expression showed a strikingly worse survival. Upon univariate and multivariate analysis, CD138 expression proved to be an independent poor prognostic factor regardless of IPI status. Even though expression pattern C proved to be an independent poor prognostic factor, it was largely influenced and dependent on CD 138 expression. Thus, its prognostic importance needs to be mentioned in conjunction with that of CD138 expression. As shown in Table 6, six patients of eight patients showing CD138 expression were negative for germinal center B cell markers. Thus, they were subclassified into the non-GC and pattern C groups. However, one patient was positive for both CD10 and BCL-6, and another patient for BCL-6 alone. These patients were subclassified into the GCB and pattern B groups. In particular, an 80 yr-old male patient (patient 1) as shown in Table 6 expired at six months. In contrast, a 28-yr-old female patient (patient 2) was still alive at follow up 68 months later. After a more detailed search for clinical information about this patient, the authors discovered that she received bone marrow transplantation after her DLBCL diagnosis. The remaining six patients in the non-GC and pattern C groups all expired in relatively short period (patients 3-8). Colomo et al. reported that CD138 is highly expressed in the plasmablastic variant of DLBCL, the so-called plasmablastic lymphoma, which is usually associated with HIV infection, and has a highly aggressive clinical behavior (33). Plasmablastic lymphoma was characterized by immunoblastic morphology and a plasma cell phenotype with absent or weak B-cell markers and strong reactivity for plasma cell associated antigens. Patients with CD138 expression in the present study might be diagnosed with plasmablastic lymphoma. However, all eight CD138 expression tumors did not show the morphologic criteria of plasmablastic lymphoma upon histologic review. They were also diffusely and strongly positive for B-cell markers such as CD20 and CD79a. Moreover, despite a diligent search for clinical records of all patients with CD138 expression, there was no evidence of HIV-1 infection or immunosuppression. Thus, the possibility of plasmab lastic lymphoma having an aggressive clinical behavior could be ruled out. Therefore, our results suggest that CD138 expression may play an important role in clinical outcome as a poor prognostic factor. Unfortunately, CD138 expression has been reported at an extremely low expression rate, as shown by the present study.
In conclusion, our results add to the registration of prognostic implications of previously reported DLBCL subgroups. In addition, CD138 may play an important role as a poor prognostic marker. Further investigations examining CD138 expression that include more patients are necessary to reach more specific conclusions. Finally, by using an immunohistochemical panel for CD10, BCL-6, MUM-1, and CD138 expression, prognostically important subclassification of DLBCL patients was attained.
Figures and Tables
 | Fig. 1Kaplan-Meier survival analysis in DLBCL according to CD10 (A) and Bcl-6 expression (B). (A) Patients with CD10 expression above 30% demonstrate significantly better survival. (B) Patients with Bcl-6 expression above 30% tend to have a relatively better survival, but not at a significant level. |
 | Fig. 2Kaplan-Meier survival analysis in DLBCL according to MUM1 (A) and CD138 expression (B). (A) MUM1 expresssion does not influence the overall survival between positive and negative groups. (B) Patients with CD138 expression show a strikingly worse overall survival, although there were only eight positive cases. |
 | Fig. 3Kaplan-Meier survival analysis in DLBCL between GCB and non-GC subgroups. (A) The GCB subgroup demonstrates a longer survival than the non-GC subgroup. (B, C) When separately considering patients with low or high IPI scores, the GCB groups possess a longer survival in both low (B) and high IPI score groups (C). |
 | Fig. 4Kaplan-Meier survival analysis in DLBCL among patterns A, B, and C. (A) The overall survival rate curve of pattern C shows a significantly lower survival rate compared to patterns A and B. (B, C) When separately considering those patients with low or high IPI scores, pattern C also shows a strikingly poor survival rate in both low (B) and high IPI score groups (C). |
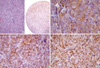 | Fig. 5Histologic and immunohistochemical findings of an 80-yr-old male CD138 positive patient (case 1). (A) Histologic findings of DLBCL patient 1. (B, C, D, E) Tumor cells were diffusely positive for CD138 (B: ×40, C: ×200), CD10 (D: ×200), and BCL-6 (E: ×200). This patient died within six months. |
References
1. A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin's lymphoma. The Non-Hodgkin's Lymphoma Classification Project. Blood. 1997. 89:3909–3918.
3. Alizadeh AA, Eisen MB, Davis RE, Ma C, Lossos IS, Rosenwald A, Boldrick JC, Sabet H, Tran T, Yu X, Powell JI, Yang L, Marti GE, Moore T, Hudson J Jr, Lu L, Lewis DB, Tibshirani R, Sherlock G, Chan WC, Greiner TC, Weisenburger DD, Armitage JO, Warnke R, Levy R, Wilson W, Grever MR, Byrd JC, Botstein D, Brown PO, Staudt LM. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature. 2000. 403:503–511.

4. Rosenwald A, Wright G, Chan WC, Connors JM, Campo E, Fisher RI, Gascoyne RD, Muller-Hermelink HK, Smeland EB, Giltnane JM, Hurt EM, Zhao H, Averett L, Yang L, Wilson WH, Jaffe ES, Simon R, Klausner RD, Powell J, Duffey PL, Longo DL, Greiner TC, Weisenburger DD, Sanger WG, Dave BJ, Lynch JC, Vose J, Armitage JO, Montserrat E, Lopez-Guillermo A, Grogan TM, Miller TP, Le-Blanc M, Ott G, Kvaloy S, Delabie J, Holte H, Krajci P, Stokke T, Staudt LM. Lymphoma/Leukemia Molecular Profiling Project. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med. 2002. 346:1937–1947.

5. Barrans SL, Carter I, Owen RG, Davies FE, Patmore RD, Haynes AP, Morgan GJ, Jack AS. Germinal center phenotype and bcl-2 expression combined with the International Prognostic Index improves patient risk stratification in diffuse large B-cell lymphoma. Blood. 2002. 99:1136–1143.

6. Colomo L, Lopez-Guillermo A, Perales M, Rives S, Martinez A, Bosch F, Colomer D, Falini B, Montserrat E, Campo E. Clinical impact of the differentiation profile assessed by immunophenotyping in patients with diffuse large B-cell lymphoma. Blood. 2003. 101:78–84.

7. Linderoth J, Jerkeman M, Cavallin-Stahl E, Kvaloy S, Torlakovic E. Nordic Lymphoma Group Study. Immunohistochemical expression of CD23 and CD40 may identify prognostically favorable subgroups of diffuse large B-cell lymphoma: a Nordic Lymphoma Group Study. Clin Cancer Res. 2003. 9:722–728.
8. Hans CP, Weisenburger DD, Greiner TC, Gascoyne RD, Delabie J, Ott G, Muller-Hermelink HK, Campo E, Braziel RM, Jaffe ES, Pan Z, Farinha P, Smith LM, Falini B, Banham AH, Rosenwald A, Staudt LM, Connors JM, Armitage JO, Chan WC. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood. 2004. 103:275–282.
9. Chang CC, McClintock S, Cleveland RP, Trzpuc T, Vesole DH, Logan B, Kajdacsy-Balla A, Perkins SL. Immunohistochemical expression patterns of germinal center and activation B-cell markers correlate with prognosis in diffuse large B-cell lymphoma. Am J Surg Pathol. 2004. 28:464–470.

10. Zettl A, Meister S, Katzenberger T, Kalla J, Ott MM, Muller-Hermelink HK, Ott G. Immunohistochemical analysis of B-cell lymphoma using tissue microarrays identifies particular phenotypic profiles of B-cell lymphomas. Histopathology. 2003. 43:209–219.

11. Milanes-Yearsley M, Hammond ME, Pajak TF, Cooper JS, Chang C, Griffin T, Nelson D, Laramore G, Pilepich M. Tissue micro-array: a cost and time-effective method for correlative studies by regional and national cancer study groups. Mod Pathol. 2002. 15:1366–1373.

12. Gatter KC, Warnke RA. Jaffe ES, Harris NL, Stein H, editors. Diffuse large B-cell lymphoma. WHO Classification of Tumors: Pathology and Genetics of Tumors of Haematopoietic and Lymphoid Tissues. 2001. Lyon, France: IARCPress;171–174.
13. Pileri SA, Dirnhofer S, Went P, Ascani S, Sabattini E, Marafioti T, Tzankov A, Leoncini L, Falini B, Zinzani PL. Diffuse large B-cell lymphoma: one or more entities? Present controversies and possible tools for its subclassification. Histopathology. 2002. 41:482–509.

15. Moller MB, Pedersen NT, Christensen BE. Diffuse large B-cell lymphoma: clinical implications of extranodal versus nodal presentation; a population-based study of 1575 cases. Br J Haematol. 2004. 124:151–159.
16. Lopez-Guillermo A, Colomo L, Jimenez M, Bosch F, Villamor N, Arenillas L, Muntanola A, Montoto S, Gine E, Colomer D, Bea S, Campo E, Montserrat E. Diffuse large B-cell lymphoma: clinical and biological characterization and outcome according to the nodal or extranodal primary origin. J Clin Oncol. 2005. 23:2797–2804.
17. Dogan A, Bagdi E, Munson P, Isaacson PG. CD10 and BCL-6 expression in paraffin sections of normal lymphoid tissue and B-cell lymphomas. Am J Surg Pathol. 2000. 24:846–852.

18. Takeshita M, Iwashita A, Kurihara K, Ikejiri K, Higashi H, Udoh T, Kikuchi M. Histologic and immunohistologic findings and prognosis of 40 cases of gastric large B-cell lymphoma. Am J Surg Pathol. 2000. 24:1641–1649.

19. Ohshima K, Kawasaki C, Muta H, Muta K, Deyev V, Haraoka S, Suzumiya J, Podack ER, Kikuchi M. CD10 and Bcl10 expression in diffuse large B-cell lymphoma: CD10 is a marker of improved prognosis. Histopathology. 2001. 39:156–162.

20. Go JH, Yang WI, Ree HJ. CD10 expression in primary intestinal large B-cell lymphomas: its clinical significance. Arch Pathol Lab Med. 2002. 126:956–960.
21. Uherova P, Ross CW, Schnitzer B, Singleton TP, Finn WG. The clinical significance of CD10 antigen expression in diffuse large B-cell lymphoma. Am J Clin Pathol. 2001. 115:582–588.

22. Biasoli I, Morais JC, Scheliga A, Milito CB, Romano S, Land M, Pulcheri W, Spector N. CD10 and Bcl-2 expression combined with the International Prognostic Index can identify subgroups of patients with diffuse large-cell lymphoma with very good or very poor prognoses. Histopathology. 2005. 46:328–333.

23. Fabiani B, Delmer A, Lepage E, Guettier C, Petrella T, Briere J, Penny AM, Copin MC, Diebold J, Reyes F, Gaulard P, Molina TJ. Groupe d'Etudes des Lymphomes de l'Adulte. CD10 expression in diffuse large B-cell lymphomas does not influence survival. Virchows Arch. 2004. 445:545–551.
24. Xu Y, McKenna RW, Kroft SH. Comparison of multiparameter flow cytometry with cluster analysis and immunohistochemistry for the detection of CD10 in diffuse large B-Cell lymphomas. Mod Pathol. 2002. 15:413–419.

25. Falini B, Mason DY. Proteins encoded by genes involved in chromosomal alterations in lymphoma and leukemia: clinical value of their detection by immunocytochemistry. Blood. 2002. 99:409–426.

26. Cattoretti G, Chang CC, Cechova K, Zhang J, Ye BH, Falini B, Louie DC, Offit K, Chaganti RS, Dalla-Favera R. BCL-6 protein is expressed in germinal-center B cells. Blood. 1995. 86:45–53.

27. Zhang A, Ohshima K, Sato K, Kanda M, Suzumiya J, Shimazaki K, Kawasaki C, Kikuchi M. Prognostic clinicopathologic factors, including immunologic expression in diffuse large B-cell lymphomas. Pathol Int. 1999. 49:1043–1052.

28. Kramer MH, Hermans J, Wijburg E, Philippo K, Geelen E, van Krieken JH, de Jong D, Maartense E, Schuuring E, Kluin PM. Clinical relevance of BCL2, BCL6, and MYC rearrangements in diffuse large B-cell lymphoma. Blood. 1998. 92:3152–3162.

29. Kawasaki C, Ohshim K, Suzumiya J, Kanda M, Tsuchiya T, Tamura K, Kikuchi M. Rearrangements of bcl-1, bcl-2, bcl-6, and c-myc in diffuse large B-cell lymphomas. Leuk Lymphoma. 2001. 42:1099–1106.

30. Falini B, Fizzotti M, Pucciarini A, Bigerna B, Marafioti T, Gambacorta M, Pacini R, Alunni C, Natali-Tanci L, Ugolini B, Sebastiani C, Cattoretti G, Pileri S, Dalla-Favera R, Stein H. A monoclonal antibody (MUM1p) detects expression of the MUM1/IRF4 protein in a subset of germinal center B cells, plasma cells, and activated T cells. Blood. 2000. 95:2084–2092.

31. Tsuboi K, Iida S, Inagaki H, Kato M, Hayami Y, Hanamura I, Miura K, Harada S, Kikuchi M, Komatsu H, Banno S, Wakita A, Nakamura S, Eimoto T, Ueda R. MUM1/IRF4 expression as a frequent event in mature lymphoid malignancies. Leukemia. 2000. 14:449–456.

32. Carbone A, Gloghini A, Gaidano G, Franceschi S, Capello D, Drexler HG, Falini B, Dalla-Favera R. Expression status of BCL-6 and syndecan-1 identifies distinct histogenetic subtypes of Hodgkin's disease. Blood. 1998. 92:2220–2228.




 PDF
PDF ePub
ePub Citation
Citation Print
Print






 XML Download
XML Download