Abstract
The aim of study was to describe Mycoplasma pneumoniae epidemics in a hospital-based population. Special attention was paid to the relationship between antibody titer to M. pneumoniae and sex, age, and atopy. During the eight 6-month periods between January 2000 and December 2003, serum samples were obtained from 1,319 Korean children who presented with respiratory symptoms, and were examined for antibodies to M. pneumoniae using the indirect particle agglutination test. Geometric mean antibody titers peaked in the second half of 2000 and then decreased gradually, a second peak occurred in the second half of 2003. Likewise, the frequency of high antibody titers (≥1:640) also peaked during these two periods. Antibody titers in children aged 0-3 yr were lower than in older children during both peak periods and for 2 yr after the first peak. Sex and atopy had no effect on antibody titers. During the years 2000-2003, geometric mean antibody titers and the frequencies of high antibody titers varied with time. These changes suggest a cyclic pattern of M. pneumoniae infection, with two epidemic peaks separated by 3 yr.
Mycoplasma pneumoniae is a well known causative agent of acute respiratory infection in childhood. It is a cause of primary atypical pneumonia, but many infections are asymptomatic or cause only mild symptoms, such as pharyngitis and bronchitis (1). Thus clinical findings are seldom diagnostic for M. pneumoniae infection.
M. pneumoniae infections are endemic in large urban areas and epidemic increases are observed at 3- to 7-yr intervals (2-6). In Denmark the disease occurred in a regular pattern of epidemics every 4.5 yr during the period 1958-1974 (2). Epidemics with an interval of 7 yr were reported in Seattle, U.S.A. in 1966-1967 and 1974 (3), and in Japan epidemic peaks occurred regularly at 4-yr intervals during the period 1980-1992 (4). A few studies on epidemics of M. pneumoniae infections have been undertaken in Korea (7-10), which describe epidemics occurring at 3- to 4-yr intervals. However, these studies focused on the number of community acquired pneumonia cases caused by M. pneumoniae among hospitalized patients, and no epidemiologic study of M. pneumoniae infections has been conducted on the basis of a serologic diagnosis in subjects representing general population.
The standard laboratory methods for the diagnosis of M. pneumoniae infections have been culture and serology. The agent is fastidious and grows slowly, limiting the usefulness of culture for routine purposes (11). Serology is more sensitive for detecting acute infection than culture. A 4-fold rise in antibody titer in acute and convalescent sera is considered necessary for the diagnosis of current M. pneumoniae infection (12). However, a significant rise in antibody titer can not be demonstrated unless the first blood specimen is taken within 10 days of the onset of illness (3) or unless convalescent serum is obtained at proper time intervals (13). Furthermore, serologic tests with paired sera are not suited for the detection of asymptomatic infection. Thus most seroepidemiologic studies confirmed the existence of epidemics by display of the distribution of seropositive cases in time on the basis of testing single serum samples (2, 14-16).
The aim of the present study was to describe the presence of M. pneumoniae epidemics in a hospital-based population by retrospective analysis of serologic data over a period of 4 yr. Special attention was paid to the relationship between antibody titer to M. pneumoniae and sex, age, and atopy during childhood.
The study population comprised 1,319 Korea children aged ≤15 yr, who presented at the outpatient clinic of Seoul National University Hospital for the first time, with acute or chronic respiratory symptoms. Respiratory symptoms included cough, wheezing or other noisy breathing, a runny or stuffed nose, or respiratory difficulties. Children with immunodeficiency disorder were excluded from the study. During the eight 6-month periods between January 2000 and December 2003, serum samples were investigated for anti-M. pneumoniae antibodies. The numbers of samples collected in each period were; 123 and 111 in the first and second halves of 2000, 147 and 167 in the first and second halves of 2001, 172 and 212 in the first and second halves of 2002, 203 and 184 in the first and second halves of 2003, respectively. Only one sample was obtained per subject, and subjects enrolled in one period were not included in any other period. The mean±SD age of all subjects was 6.0±3.4 yr with a male to female ratio of 2:1. Subjects were grouped according to age: 0-3 (n=417), 4-6 (n=508), and 7-15 yr (n=394), to allow antibody titers to be examined by age. Parents provided written informed consent for their children to participate in the study. The study protocol was approved by the Hospital Ethics Committee.
Anti-M. pneumoniae antibodies in serum specimens were titrated using the indirect particle agglutination test (Serodia-MycoII, Fujirebio, Japan), according to the manufacturer's instructions. This test is based on the principle that gelatin particles sensitized with M. pneumoniae cell membrane components are agglutinated in the presence of M. pneumoniae antibody. Blood samples were drawn into Vacutainer SST tubes (Becton Dickinson, Franklin Lakes, NJ, U.S.A.) and centrifuged at 2,000 r.p.m. for 10 min. The separated serum was stored at -20℃ until required for titer determination. The cut-off value for a positive result was 1:40 (17). The demonstration of high antibody titers (≥1:640) was considered evidence of recent M. pneumoniae infection (10, 18).
In children aged 4 yr and over, atopy was defined as at least one positive skin-prick test response (≥3 mm wheal diameter) for a panel of 12 common aeroallergens, in the presence of positive and negative controls (19). In children less than 4 yr old who could not undertake the skin-prick test, atopy was defined as the presence of serum IgE antibodies ≥0.7 kU/L against at least one allergen and/or total IgE ≥200 kU/L (20, 21). Serum total and specific IgE were measured using the Coat-A-Count® Total IgE IRMA (Diagnostic Products Co., Los Angeles, CA, U.S.A.) and using the BLAST system (Bio-Line, Brussels, Belgium), respectively. Whole blood was collected by sterile venipuncture, serum was separated, and stored frozen (-80℃) until assayed. Determinations were made according to the manufacturer's specifications. Specific IgE was measured to house dust mite (Dermatophagoides pteronyssinius, Dermatophagoides farinae) and cockroach (Blatella germanica). Neither skin-prick tests nor IgE tests were performed on 30 individuals (15 in 1st half of 2000, 9 in 2nd half of 2000, and 5 in 2nd half of 2003).
Geometric mean titers and their corresponding 95% confidence intervals (CIs) were obtained by transformation of the antibody titer levels to a logarithmic scale, subsequent calculation of arithmetic means and 95% CIs, and back-transformation of the results to the original scale. Two-way ANOVA was used to evaluate the difference of antibody titers between eight periods with age as another grouping factor, and was followed by Tukey's multiple comparison test. To compare antibody titers between the three age groups at each period, one-way ANOVA was used. Antibody titers were compared in males and females, and between atopic and non-atopic subject groups for each period using the Student's t test. A p-value of less than 0.05 was considered statistically significant.
Fig. 1 shows changes in geometric mean antibody titers over the eight 6-month periods. Two peaks of mean antibody titers were noted to be separated by 3 yr. Titers peaked in the second half of 2000 (geometric mean: 262.0, 95% CI: 174.8-392.8), and then decreased gradually, but remained elevated for one year (101.7, 78.9-131.2 in the first half of 2001; 61.1, 50.9-73.3 in the second half of 2001), compared to baseline titer in the first half of 2000 (35.5, 30.8-41.0), and then achieved baseline (35.6, 31.6-40.1 in the first half of 2002; 38.1, 34.0-42.7 in the second half of 2002; 41.7, 36.3-47.9 in the first half of 2003). However, the peak recurred in the second half of 2003 (151.8, 118.5-194.4).
Likewise, the frequency of high antibody titers (≥1:640) peaked in the second half of 2000, gradually declined, and then re-peaked in the second half of 2003. The frequencies of high antibody titers in these periods were as follows: 0.8% and 37.8% in the first and second halves of 2000, 17.7% and 6.6% in the first and second halves of 2001, 0.6% and 1.4% in the first and second halves of 2002, 3.4% and 30.4% in the first and second halves of 2003, respectively (Fig. 2). In addition, the frequency of positive antibody titers (≥1:40) changed in same manner during the study periods. The 95 and 97.5 percentiles for anti-M. pneumoniae antibody titer during these periods were 1:160 and 1:320 in the first half of 2000, 1:20,480 and 1:20,480 in the second half of 2000, 1:1,280 and 1:2,560 in the first half of 2001, 1:640 and 1:1,280 in the second half of 2001, 1:160 and 1:320 in the first half of 2002, 1:320 and 1:320 in the second half of 2002, 1:320 and 1:640 in the first half of 2003, and 1:5,120 and 1:5,120 in the second half of 2003, respectively.
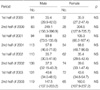
An examination of antibody titers by age group showed that the youngest children aged 0-3 yr had lower titers than those aged 4-6 yr or ≥7 yr during the two peak periods and during the 2-yr period after the first peak, whereas no significant difference was found between the latter two age groups (Table 1). Table 2 shows antibody titers to M. pneumoniae in 8 study periods by sex. The differences were not statistically significant, although females had slightly higher titers than males during all periods except for the first half of 2002. Antibody titers to M. pneumoniae were not significantly different between atopic and non-atopic subjects throughout the study (Table 3).
Antibody titers to M. pneumoniae rose to a peak level in the second half of 2000 and then decreased gradually, and re-peaked in the second half of 2003. This was duplicated by frequencies of high antibody titers (≥1:640). Antibody titers in children aged 0-3 yr were lower than those in older children during the two peak periods and during the 2-yr period following the first peak. Sex and atopy were found to have no effect on antibody titers throughout the study.
This study was based on a retrospective analysis of results from testing single sera, obtained in subjects who visited our outpatient clinic for the first time with acute or chronic respiratory symptoms. Serologic tests with paired sera are not practical in patients with no evidence of lower respiratory infection with M. pneumoniae. Furthermore, it is difficult to verify a significant rise of antibody titer in all cases with current M. pneumoniae infection because its rise is highly dependent on the time of presentation. Moreover, long persistence of antibodies following a M. pneumoniae infection (22) may affect interpretation of our results. Therefore, our data concerning antibody titer to M. pneumoniae may be the consequence of past infection as well as current or recent infection with M. pneumoniae. It should be noted that the patients reviewed in the present study do not precisely match the general population with regard to age or sex distribution. Younger children visit our clinic more frequently, and males outnumber females by about two to one, among those who present with respiratory symptoms.
A single titer of ≥1:640 is regarded as indicative of recent M. pneumoniae infection (10, 18). We found that the frequency of high titers (≥1:640) showed two peaks with a 3-yr interval and these coincided with the peaks of mean antibody titers, which reflects a cycle of M. pneumoniae epidemics during the study period. Our results were parallel with studies from different regions of the world, which described the cyclic nature of epidemics of M. pneumoniae infections (2-6). What governs the periodicity of these epidemics is not well known. The community-based transmission of M. pneumoniae diseases among children may have resulted in protective immunity in a significant proportion of the population. The duration of protective immunity in an individual has been estimated from a few published cases to last about 4 yr with a range of 2 to 10 yr (23-25). Due to the limited duration of protective immunity, the balance between protected and susceptible individuals would be lost with time, and thus the following epidemic could develop.
In the present study, the frequency of higher titers of ≥1:640 was 37.8% at the first peak (2nd half of 2000). This frequency is slightly higher than the previously reported prevalence of infection of all clinical syndromes caused by M. pneumoniae, which ranged from 10 to 35% during epidemic periods (3, 26-28). A single high titer is unlikely to indicate only current infection with M. pneumoniae. Furthermore, our subjects may include children with inapparent infection, whereas the study populations in most previous studies were confined to those with acute respiratory symptoms. Epidemic patterns of M. pneumoniae are not identical between different regions and over time, which may also explain the difference between our frequency and the prevalence previously reported (3, 14, 27).
The levels of antibody to M. pneumoniae and the proportion of high titers (≥1:640, data not shown) were not different between males and females during the study period. Lind and Bentzon reported significantly more boys than girls in a seropositive population (27), but provided no explanation for this difference in gender distribution. However, more recently Rastawicki et al. demonstrated no gender difference in patients with M. pneumoniae infection (14), which is in agreement with the findings of the present study.
We found that antibody titers of children aged 0-3 yr were lower compared to those of children aged 4-6 yr and ≥7 yr at the two epidemic peaks and for 2 yr after the first peak, with no significant difference between the two latter age groups. Moreover, the frequency of high titers by age group showed a similar trend (data not shown). This finding is in accordance with the results of previous seroepidemiologic studies, which found a low prevalence of M. pneumoniae infection in children under 3 yr of age (16, 27). But more recently Layani-Milon et al. demonstrated no significant difference in its infection rates between children under 4 yr of age and children in other age groups and adults (28). Other explanations can also be considered. First, the degree of antibody response to M. pneumoniae may be affected by age. A few studies have demonstrated that most symptomatic disease occurs in older children and young adults even though M. pneumoniae infection is common in infants and young children, and that antibody response in younger children is of lesser magnitude than in older children and adults (15, 27, 29). In this context, it has been presumed that an infection with M. pneumoniae in infancy may enhance the immune response so that subsequent infection becomes more severe, resulting in pneumonia with an increased antibody response as a result of anamnestic recall (29, 30). Another alternative or concomitant possibility is that lower antibody levels in children aged 0-3 yr may be due to different decay rates of antibody by age. Foy et al. demonstrated that antibody titers to M. pneumoniae declined more slowly in adults with M. pneumoniae pneumonia than in young children (<5 yr of age) (22).
Intracellular localization is now appreciated for M. pneumoniae (31), against which the host response has been reported to be associated with cell mediated immunity (29, 32), thus it is assumed that the activation of cell-mediated immune responses would be important for protective immunity, like the association between resistance to Chlamydia pneumoniae, a well-known intracellular microbe and the induction of Th1 responses (33-36). More recently Ferrari and coworkers found elevated antibody titers to C. pneumoniae more frequently in atopic subjects and postulated that atopic disease might be associated with a less efficient clearance of C. pneumoniae or with a more severe infection and consequently higher specific antibody titers (37). While no studies on the relationship between atopy and M. neumoniae serum antibody titers have been performed, we speculate that atopic subjects have higher anti-M. pneumoniae antibody titers than non-atopic subjects as in the case of C. pneumoniae. However, the present study indicates that atopy had no effect on antibody titers throughout the study period, which is contrary to expectation.
In conclusion, over the years 2000-2003, geometric mean antibody titers and the frequencies of high antibody titers to M. pneumoniae varied. These changes suggested a cyclic pattern of M. pneumoniae infection, with two epidemic peaks separated by 3 yr. However, it must be underlined that the existence of epidemics was inferred from changes in antibody titers with time based on testing single sera in the present study. In addition, age, but neither sex nor atopy, was found to affect antibody titers.
Figures and Tables
 | Fig. 1Antibody titers to Mycoplasma pneumoniae during the eight consecutive 6-month periods (2000-2003). Closed circles and short horizontal bars indicate geometric means and 95% confidence intervals, respectively. *p<0.001; †p<0.01, compared with the titer in the first half of 2000. |
 | Fig. 2Distributions of antibody titers to Mycoplasma pneumoniae during the eight consecutive 6-month periods. |
Table 1
Antibody titers to Mycoplasma pneumoniae during the eight consecutive 6-month periods, by age in years

References
1. Atmar RL, Greenberg SB. Pneumonia caused by Mycoplasma pneumoniae and the TWAR agent. Semin Respir Infect. 1989. 4:19–31.
2. Lind K, Benzon MW, Jensen JS, Clyde WA Jr. A seroepidemiological study of Mycoplasma pneumoniae infections in Denmark over the 50-year period 1946-1995. Eur J Epidemiol. 1997. 13:581–586.
3. Foy HM, Kenny GE, Cooney MK, Allan ID. Long-term epidemiology of infections with Mycoplasma pneumoniae. J Infect Dis. 1979. 139:681–687.

4. Ito I, Ishida T, Osawa M, Arita M, Hashimoto T, Hongo T, Mishima M. Culturally verified Mycoplasma pneumoniae pneumonia in Japan: a long-term observation from 1979-99. Epidemiol Infect. 2001. 127:365–367.

5. Foy HM, Kenny GE, McMahan R, Mansy AM, Grayston JT. Mycoplasma pneumoniae pneumonia in an urban area. Five years of surveillance. JAMA. 1970. 214:1666–1672.

6. Joosting AC, Harwin RM, Coppin A, Battaglia P, van der Hoef P. A serological investigation of Mycoplasma pneumoniae infection on the Witwatersrand. S Afr Med J. 1976. 50:2134–2135.
7. Kang KS, Woo H. Pattern of occurrence of Mycoplasma pneumoniae pneumonia in admitted children: southern central Korea, from 1989 to 2002. J Korean Pediatr Soc. 2003. 46:474–479.
8. Park HY, Woo CW, Choung JT, Son CS, Tockgo YC. Trend of the Mycoplasma pneumonia: during recent 9 years period. Pediatr Allergy Respir Dis. 1995. 5:49–59.
9. Yoo HS, Kang HY, Jeoung BJ, Kim KE, Lee KY. A clinical study of Mycoplasma pneumoniae pneumonia. Pediatr Allergy Respir Dis. 1995. 5:112–122.
10. Hong JY, Nah SY, Nam SG, Choi EH, Park JY, Lee HJ. Occurrence of Mycoplasma pneumoniae pneumonia in Seoul, Korea, from 1986 to 1995. J Korean Pediatr Soc. 1997. 40:607–613.
11. Waris ME, Toikka P, Saarinen T, Nikkari S, Meurman O, Vainionpaa R, Mertsola J, Ruuskanen O. Diagnosis of Mycoplasma pneumoniae pneumonia in children. J Clin Microbiol. 1998. 36:3155–3159.
12. Waites KB. New concepts of Mycoplasma pneumoniae infections in children. Pediatr Pulmonol. 2003. 36:267–278.
13. Daxboeck F, Krause R, Wenisch C. Laboratory diagnosis of Mycoplasma pneumoniae infection. Clin Microbiol Infect. 2003. 9:263–273.

14. Rastawicki W, Kaluzewski S, Jagielski M. Occurrence of serologically verified Mycoplasma pneumoniae infections in Poland in 1970-1995. Eur J Epidemiol. 1998. 14:37–40.
15. Ponka A, Ukkonen P. Age-related prevalence of complement-fixing antibody to Mycoplasma pneumoniae during an 8-year period. J Clin Microbiol. 1983. 17:571–575.
16. Bosnak M, Dikici B, Bosnak V, Dogru O, Ozkan I, Ceylan A, Haspolat K. Prevalence of Mycoplasma pneumoniae in children in Diyarbakir, the south-east of Turkey. Pediatr Int. 2002. 44:510–512.

17. Barker CE, Sillis M, Wreghitt TG. Evaluation of Serodia Myco II particle agglutination test for detecting Mycoplasma pneumoniae antibody: comparison with mu-capture ELISA and indirect immunofluorescence. J Clin Pathol. 1990. 43:163–165.

18. Choi SK, Jung JA, Kim KH, Kim GH. Study of seroprevalence of antimycoplasma antibody in healthy children and its diagnostic value. J Korean Pediatr Soc. 1998. 41:489–497.
19. Host A, Andrae S, Charkin S, Diaz-Vazquez C, Dreborg S, Eigenmann PA, Friedrichs F, Grinsted P, Lack G, Meylan G, Miglioranzi P, Muraro A, Nieto A, Niggemann B, Pascual C, Pouech MG, Rance F, Rietschel E, Wickman M. Allergy testing in children: why, who, when and how? Allergy. 2003. 58:559–569.

20. Wittig HJ, Belloit J, De Fillippi I, Royal G. Age-related serum immunoglobulin E levels in healthy subjects and in patients with allergic disease. J Allergy Clin Immunol. 1980. 66:305–313.

21. Tang ML, Coleman J, Kemp AS. Interleukin-4 and interferon-gamma production in atopic and non-atopic children with asthma. Clin Exp Allergy. 1995. 25:515–521.
22. Foy HM, Kenny GE, Cooney MK, Allan ID, van Belle G. Naturally acquired immunity to pneumonia due to Mycoplasma pneumoniae. J Infect Dis. 1983. 147:967–973.

23. Foy HM, Kenny GE, Seri R, Ochs HD, Allan ID. Second attacks of pneumonia due to Mycoplasma pneumoniae. J Infect Dis. 1977. 135:673–677.

24. Biberfeld G. Antibody responses in Mycoplasma pneumoniae infection in relation to serum immunogloblins, especially IgM. Acta Pathol Microbiol Scand Sect B. 1971. 79:620–634.
25. Nakamura S, Ebisawa I, Kitamoto O, Sato T. Persistence of serum antibody following Mycoplasma pneumoniae infection. Am Rev Respir Dis. 1970. 101:620–622.
26. Broughton RA. Infections due to Mycoplasma pneumoniae in childhood. Pediatr Infect Dis. 1986. 5:71–85.

27. Lind K, Bentzon MW. Ten and a half years seroepidemiology of Mycoplasma pneumoniae infection in Denmark. Epidemiol Infect. 1991. 107:189–199.
28. Layani-Milon MP, Gras I, Valette M, Luciani J, Stagnara J, Aymard M, Lina B. Incidence of upper respiratory tract Mycoplasma pneumoniae infections among outpatients in Rhone-Alpes, France, during five successive winter periods. J Clin Microbiol. 1999. 37:1721–1726.
29. Fernald GW, Collier AM, Clyde WA Jr. Respiratory infections due to Mycoplasma pneumoniae in infants and children. Pediatrics. 1975. 55:327–335.
30. Brunner H, Horswood RL, Chanock RM. More sensitive methods for detection of antibody to Mycoplasma pneumoniae. J Infect Dis. 1973. 127:Suppl. S52–S55.

31. Dallo SF, Baseman JB. Intracellular DNA replication and long-term survival of pathogenic mycoplasmas. Microb Pathog. 2000. 29:301–309.

32. Radisic M, Torn A, Gutierrez P, Defranchi HA, Pardo P. Severe acute lung injury caused by Mycoplasma pneumoniae: potential role for steroid pulses in treatment. Clin Infect Dis. 2000. 31:1507–1511.
33. Holtmann H, Shemer-Avni Y, Wessel K, Sarov I, Wallach D. Inhibition of growth of Chlamydia trachomatis by tumor necrosis factor is accompanied by increased prostaglandin synthesis. Infect Immun. 1990. 58:3168–3172.

34. Williams DM, Magee DM, Bonewald LF, Smith JG, Bleicker CA, Byrne GI, Schachter J. A role in vivo for tumor necrosis factor alpha in host defense against Chlamydia trachomatis. Infect Immun. 1990. 58:1572–1576.

35. Sher A, Coffman RL. Regulation of immunity to parasites by T cells and T cell-derived cytokines. Annu Rev Immunol. 1992. 10:385–409.

36. Kaufman SH. Immunity to intracellular bacteria. Annu Rev Immunol. 1993. 11:129–163.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download