Abstract
To evaluate the toxicities and efficacies of two fractionation regimens of high-dose-rate brachytherapy in uterine cervical cancer, patients were stratified by stage Ib-IIa versus IIb-IVa, and randomly assigned to receive 3 Gy fractions (group A) or 5 Gy fractions (group B). External radiotherapy was performed using a 10 MV radiography with a daily 1.8 Gy up to 30.6 Gy to the whole pelvis, and then with a midline shield up to 45.0 Gy. Brachytherapy was performed with 3 Gy × 10 times or 5 Gy × 5 times, and this was followed by booster brachytherapy of a smaller fraction to the residual tumor. Between August 1999 to July 2000, 46 patients were eligible. Median follow-up period was 42 months (5-49). The range of age was 37-83 yr (median, 58). The three-year disease-specific survival rates of group A (n=23) and B (n=23) were 90.5%, 84.9%, respectively (p=0.64). The three-year pelvic control rates of group A and B were 90.0% and 90.9%, respectively (p=0.92). The incidences of late complications of the rectum or bladder of grade 2 or greater in groups A and B were 23.8% and 9.1%, respectively (p=0.24). Our study showed that the results of two regimens were comparable. Fractionation regimen using 5 Gy fractions seems to be safe and effective, and offers shorter treatment duration.
In Korea, invasive uterine cervical cancer was ranked as the fourth most common female cancer in 2002, although its incidence is decreasing gradually (1). Radiotherapy with or without chemotherapy using a combination of external beam radiotherapy (EBRT) and intracavitary radiotherapy (ICR) is an important treatment modality for any stage of cervical cancer. The role of ICR is to deliver a high dose of radiation directly to the tumor while sparing the adjacent normal tissues. ICR has been performed by low-dose-rate (LDR) or high-dose-rate (HDR) brachytherapy. Many centers have reported treatment results, which show that the two modalities are comparable (2, 3). HDR brachytherapy has advantages over LDR in that staff is not exposed to a radiation hazard, the treatment time is shorter, there is no need for hospitalization, no risk of anesthesia and thromboembolism, etc. Many centers in Korea have adopted HDR brachytherapy and more than 70% of cervical cancer patients referred for curative radiotherapy are treated using HDR brachytherapy (4). Although many studies on the use of HDR brachytherapy in cervix cancer are available worldwide, the optimum treatment scheme remains unclear. Moreover, because concurrent chemoradiotherapy is now viewed as the standard treatment for locally advanced cervical cancer, optimum HDR brachytherapy fractions, especially in combination with chemotherapy, need to be settled urgently.
In 2000, the American Brachytherapy Society (ABS) published its recommendations for the use of HDR brachytherapy and recommended a range of 4 to 8 fractions (5). But they also warned that these schedules had not been thoroughly tested clinically and their use requires great caution. Much literature is available regarding optimal fractionation regimens, but the majority of studies have been retrospective (6-10). The results of a Medline search showed that only one prospective randomized trial that compared two ICR fractionation regimens had been undertaken (11). But, in the study, radiation doses were not compared with the concept of a biological effective dose (BED) (12, 13) and chemotherapy was not performed.
So, we performed a prospective randomized study and compared two fractionation regimens of 3 Gy and 5 Gy of HDR brachytherapy in cervical cancer patients treated by curative radiotherapy with or without concurrent chemotherapy, and tried to evaluate the toxicities and efficacies of these two regimens with respect to biological effective doses at various reference points.
This study was designed as a prospective randomized study conducted at the Chonnam National University Hospital. From August 1999 to July 2000, patients with carcinoma of the uterine cervix were enrolled in the study, and randomly assigned to receive ICR fractionation regimens of fraction size 3 Gy or 5 Gy. These two fraction sizes were selected for the following reasons. In Korea, the 5 Gy fraction is most commonly used, and the 3 Gy fraction is the smallest fraction used. The latter may have more potential to spare normal tissue radiobiologically, at the expense of more protracted fractionation. Before randomization, patients were stratified according to the International Federation of Gynecology and Obstetrics (FIGO) as stage Ib-IIa or stage IIb-IVa. Other stratification factors were not considered because of the relatively small study sample size.
Eligibility criteria consisted of a pathologically verified squamous cell carcinoma or adenocarcinoma of the cervix, and an Eastern Cooperative Oncology Group (ECOG) performance status index of 0-2. The patients had no history of previous malignancy, and hepatic, renal, and cardiopulmonary functions were adequate. Pretreatment evaluation included a complete medical history taking and a physical examination, and baseline blood tests, chest radiography, sigmoidoscopy, cystoscopy, and intravenous pyelography, and if necessary, computed tomography (CT) or magnetic resonance imaging (MRI) of the abdomino-pelvis. Informed consents were obtained from all patients, and the study protocol was approved by the institutional review board of Chonnam National University Hospital.
Patients in each stratum of stage Ib-IIa and stage IIb-IVa were assigned to either an ICR regimen of 3 Gy fractions (Group A) or 5 Gy fractions (Group B) by using a computer to randomly generate numbers. Prior to ICR, all patients underwent EBRT to the whole pelvis. EBRT was performed using a 10 MV linear accelerator using the 4-field box technique in daily fractions of 1.8 Gy, 5 days per week. EBRT prior to midline shield was delivered up to around 30.6 Gy over 3.5 weeks regardless of stage to allow a more controlled comparison of cumulative BED. After ICR started, further EBRT with midline shield was delivered on non-ICR weekdays up to total dose of 45 Gy, and if necessary, a booster dose of 5.4-14.4 Gy was delivered to the lateral pelvic wall.
Brachytherapy was performed by using a remotely controlled afterloading system containing a high-dose-rate radioisotope of 192Ir (Varisource®, Varian, U.S.A.). A curved uterine tandem and paired ovoids were used as applicators. Applicator insertion was done on an outpatient basis with non-narcotic analgesics. For ICR simulation, orthogonal radiography films of anteroposterior and lateral view were taken with the applicators inserted, and the position of rectum, bladder, and the A point were defined according to ICRU 38 recommendations (14). The reference points of the rectum and bladder were identified on films by inserting a Foley catheter balloon filled with 5 mL of contrast media and by filling the rectum with 50 mL of barium. The rectal point dose was estimated at the anterior rectal wall on a line from the middle of the ovoid sources. ICR isodose curves were optimized by adjusting the dwell times of a 5 mm-length stepping source at each position for the three applicators. ICR was initially administered as a fraction size of 3 Gy to the A point, 3 times a week in 10 fractions (30 Gy) in group A, and 5 Gy twice a week in 5 fractions (25 Gy) in group B. This was planned as a similar total BED at A point for both groups. At this time, follow-up gynecological examinations or radiological studies, such as MRI, were undertaken to assess tumor shrinkage. According to the magnitude of the tumor regression and the resultant anatomical changes of the uterus, a secondary ICR boost was performed at a much smaller fraction size to A point as well as to the rectal and bladder reference point, with sufficient coverage of the residual tumor. This additional ICR boost was delivered 5 times in group A and 3 times in group B. The biological effective dose was calculated by using a linear-quadratic model with α/β ratio of 10 for tumors and an α/β ratio of 3 for the rectum and bladder. When calculating BED, we did not consider tumor repopulation during treatment. The equations used for the BED calculation at each reference point were as follows:
Chemotherapy was recommended in patients with adequate hepatic, renal, and cardiopulmonary functions, who were diagnosed as FIGO Ib-IIa with a bulky tumor (greater than 4 cm) or radiological pelvic lymphadenopathy, and in all patients with FIGO IIb or greater. The chemotherapy agents used were 5-FU (1,000 mg/day for day 1-5, intravenous infusion) and cisplatin (110 mg on day 1, intravenously). The timing of chemotherapy was planned to start at the same time as the external radiotherapy, and was repeated every 4 weeks for a total of four courses.
Each institution in Korea has different HDR equipment and treatment policies, which makes it difficult to conduct a multicenter study with an optimal sample size. Thus, we conducted as a single institutional pilot study and enrolled patients over a period of 12 months. The endpoints used to compare the effect of the two fractionation regimens were survival, pelvic tumor control, and complications. Toxicity was assessed using the Radiation Therapy Oncology Group (RTOG) grading system (15). Overall survival was defined as the observed length of life from the date of protocol entry to death or the last follow-up. Disease-specific survival was calculated from the time of entry to death due to disease or until the last follow-up. Patients who developed a central or a pelvic relapse were considered as pelvic failures. We assessed the tumor response 8 weeks after completing radiotherapy. Patients were followed up every 2 months over the first year, 3 monthly during the next 3 yr, and then 6 monthly during the subsequent years. Actuarial survival rates were calculated using the Kaplan-Meier method, and differences between survival curves were assessed using the log-rank test. Comparisons between categorical variables were based on the χ2 test or Fisher's exact test. Comparisons of continuous variables between independent groups were performed using the Mann-Whitney U test. The Cox proportional hazards model was used to assess the importance of various covariables. The Statistical Package for the Social Sciences (SPSS) was used for statistical analysis.
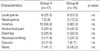
A total of 56 patients were enrolled between August 1999 and July 2000 and each treatment group included 28 patients (Table 1). However, ten patients did not complete the planned treatment. Of these, seven patients refused treatment and three patients requested transfers to other hospitals. The resultant total number of eligible patients was 46 with 23 patients in each group. The median follow-up time was 42 months (range, 5-49 months) and the follow-up rate was 43/46 (93.5%) in all patients, 21/23 in group A and 22/23 in group B. The age range of all patients was 37-83 yr (median, 58 yr). The numbers of patients according to FIGO stage Ib1, Ib2, IIa, IIb, IIIa, and IIIb were 8 (17.4%), 3 (6.5%), 10 (21.7%), 22 (47.8%), 1 (2.2%), and 2 (4.3%), respectively. Detailed data on patient characteristics are listed in Table 2, and these characteristics were not significantly different in the two groups. Radiological lymphadenopathy (LAP) was defined as pelvic or paraaortic lymph node (PAN) enlargement by CT or MRI. Maximum tumor diameter was measured by CT or MRI. Detailed data on treatment characteristics are listed in Table 3. Radiotherapy duration and ICR number differed in the two groups because of the protracted fractionation in group A. Six patients were treated by ICR more than 15 times. In these patients, secondary or even tertiary ICR boost sessions with much smaller fractions were possible because rectal or bladder reference point doses were negligible. One patient who had paraaortic LAP was treated by extended field radiotherapy up to 45 Gy. Total physical dose at the A point including EBRT and ICR seemed to be larger in group A than in group B but the median total BED10 at the A points were similar in the two groups.
The 3-yr overall survival rate (3YOSR) of all 46 patients was 76.1% (Fig. 1). According to FIGO stage, the 3YOSRs of Ib-IIa (n=21) and IIb-III (n=25) were 90.5% and 64.0%, respectively (p=0.04, Fig. 2), and the 3YOSRs of groups A and B were 78.3% and 73.9%, respectively (p=0.85, Fig. 3). There was no significant difference in 3YOSRs by age (<60 yr vs. ≥60), histology, tumor diameter (≤4 cm vs. >4), radiological LAP (pelvic vs. paraaortic vs. none), radiotherapy duration (≤60 days vs. >60), total A point BED10 (≤110 Gy vs. >110), or chemotherapy (yes vs. no). A total of 11 patients died at the time of analysis, five patients from cervical cancer and the other six patients without evidence of cervical cancer. Thereby, the 3-yr disease-specific survival rate (3YDSSR) of all patients was 87.8%. The 3YDSSRs of groups A and B were 90.5% and 85.0%, respectively (p=0.71, Fig. 4). According to FIGO stage, the 3YDSSRs of Ib-IIa and IIb-III were 90.5% and 84.9%, respectively (p=0.64).
Forty-five of 46 patients (97.8%) achieved an initial complete response. Four patients (9.3%) developed pelvic failures among 43 patients who were evaluable for tumor control (Table 4). In group A (n=21), two patients developed combined pelvic and distant failures at 9 and 20 months, respectively; a 3-yr pelvic control rate of 90.0% (Fig. 5). In group B (n=22), one patient had local failure at 19 months and another patient developed combined failure at 18 months; a 3-yr pelvic control rate of 90.9%. Distant metastasis developed in 5 patients, who had all radiological lymphadenopathy. The sites of involvement were the paraaortic lymph nodes in 2, lung in 2, and one patient had both paraaortic and supraclavicular lymph node metastasis. The two groups were not significantly different in terms of the total failure rate (Table 4).
Acute toxicities, which included hematologic profiles, were evaluated mainly in patients who received concurrent chemotherapy. No patient showed grade 3 or 4 toxicity in terms of the hematologic, gastrointestinal, or genitourinary systems. All acute toxicities were relieved spontaneously or controlled with minor medications (grade 1 or 2, Table 5). Unlike other toxicities, anemia in our patients might reflect the effect of cervical cancer per se, rather than treatment. The two groups were similar in terms of the incidences of acute toxicities.
Excluding three patients, for whom information regarding late complications was unavailable, five of the 21 patients (23.8%) in group A, and two of 22 (9.1%) in group B developed late complications, but this difference was not statistically significant, because of the small sample size (p=0.240). In group A, all five patients developed grade 2 complications (Table 6). In group B, one patient developed grade 2 rectal bleeding and another patient combined rectovaginal and vesicovaginal fistulas of grade 4. The median delay for the occurrence of rectal complications was 16 months, whereas it was 21 months for bladder complications. The mean doses at various reference points were compared in patients with or without rectal complications of grade 2 or 4 in Table 7. As listed in Table 7, ICR A point dose (40.5 Gy vs. 34.9 Gy, p=0.026) and total A point BED10 (119 Gy vs. 110 Gy, p=0.042) were significantly higher in patients with rectal complications. The mean doses at reference points were compared in patients with or without bladder complications of grade 2 or 4 in Table 8. As listed in Table 8, ICR A point dose (43.8 Gy vs. 35.1 Gy, p=0.022) and total bladder point BED3 (144 Gy vs. 117 Gy, p=0.045) were significantly higher in patients with bladder complications.
The use of HDR brachytherapy for the treatment of uterine cervix cancer has been increasing worldwide. Radiobiological aspects are mentioned in the literatures in terms of advantages or disadvantages of HDR brachytherapy versus LDR (16, 17). A better optimization of HDR brachytherapy dose distribution seems to be one of the important advantages. The variation of dwell time of single stepping sources in HDR brachytherapy permits an almost infinite variation of effective source strength. Many centers have reported treatment results and concluded that the results of LDR and HDR are comparable (2, 3). Our results concur with the literature in terms of survivals, failure patterns, and late complications. Although HDR brachytherapy has been used for more than 30 yr, there is still no consensus about the optimum fractionation schedule for the treatment of cervical cancer, and this varies widely in clinical practice. The ABS survey of brachytherapy practice for carcinoma of the cervix in the U.S.A. showed that the median HDR brachytherapy dose was about 30 Gy and that the median dose per fraction was 6 Gy for a median of 5 fractions (18). In our country, most centers use a fraction size of 3 to 5 Gy with a range of 5 to 13 fractions (4).
So far, only one prospective randomized study has attempted to determine the effects of different fractionations (11). This study found no differences in terms of treatment results or in the incidence of late complications between fraction sizes of 7.5 Gy and 6 Gy, which suggests that a small number of fractions (7.5 Gy/fraction) might be advantageous, because of the shorter duration and a lower treatment load. Petereit et al. (19) reviewed 24 articles on HDR brachytherapy for cervical cancer using different regimens, but failed to identify an optimal fractionation schedule. They observed that the technique and experience of individual centers might have played a more important role than attempts to optimize fractionation. From the radiobiological point of view, simple comparisons of various fraction sizes and total physical doses of ICR used in different studies might lead to incorrect interpretations, in addition, doses of EBRT to the whole pelvis also differ widely in studies. This is why the interpretation of optimal fraction size or total doses of ICR of HDR brachytherapy are difficult. Since the concept of BED was accepted in the clinical field, some have reported upon the results of various combinations of EBRT and ICR fractionations in terms of BED10 or BED3. Nag and Gupta (20) developed and published a simple Excel spreadsheet program to assist clinicians in the calculation of equivalent HDR brachytherapy regimen doses. Although we did not publish at that time, we also developed calculation sheets in terms of BED10 or BED3 for various combinations of EBRT and ICR fractionations using Excel, and converted BED into total equivalent doses applied as 1.8 Gy fractions, to allow more practical comparisons with conventional fractionation.
Many studies have been reported upon the acceptable BED of reference points. Lee et al. (6) reported upon the results of brachytherapy using a 3 Gy fraction size, and recommended that rectal BED3 should be kept below 130 Gy. Clark et al. (21) found a dose response relationship and a threshold for late rectal complications above 125 Gy3 at rectal reference point, in a study involving concurrent chemoradiotherapy. Ferrigno et al. (8) reported that the 5-yr late bladder complication rate was higher among patients treated with BED of larger than 125 Gy3 at bladder reference point, although the difference was not statistically significant (17% vs. 9%, p=0.27). Toita et al. (22) suggested that the rectal BED3 should be kept below 100-120 Gy. Ogino et al. (23) reported that no grade 4 rectal complication was observed in any patient with a time-dose factor (TDF) below 130 or a BED below 147. In our study, the mean rectal BED3 of patients with grade 2-4 complications was above 150 Gy, although no significant difference was found between these patients and those with lower grade complications. Mean bladder BED3 of our patients with grade 2-4 complications was above 140 Gy and this differed significantly from those with lower grade complications.
It is difficult to compare late complication rates with those of other studies, because of the different grading systems adopted, and the different follow-up periods used. Fu and Phillips (2) reviewed the treatment results of HDR brachytherapy in the literature. Major rectal and bladder complication rates ranged from 1.4% to 10% and from 0.3% to 4%, respectively. Our study also showed comparable results. However, the relationship between reference rectal and bladder points doses defined by ICRU 38 and the incidence of late complications has not always been accepted. A recent survey upon the use of the ICRU 38 reference point showed that rectum and bladder ICRU points were recorded in only 55% and 58%, respectively, in clinical practice (24). Moreover, those two points were reported in only 18% and 14%, respectively, in literature. The study concluded that comprehensive concepts were needed for reporting dose to points and volumes in target and critical organs. Kapp et al. (25) failed to find a correlation between the maximum doses to the rectum, bladder and the A point, and the development of late sequelae. They concluded that threshold doses for normal tissue in HDR brachytherapy could not be determined by considering doses at single points. In our study, the bladder reference point had a greater predictive value for late complications than the rectal reference point. The current tendency is to consider the volume and dose to organs irradiated, rather than a single point that is presumed to receive a maximum dose (26). Using CT-based brachytherapy planning, Uno et al. (27), in an analysis of 100 patients with stages IIb or IIIb cervical cancer treated with HDR brachytherapy, found that the depth of the 6-Gy isodose volume determined three dimensionally has a predictive value in terms of late rectal complications.
Recently, several randomized trials have shown that better treatment results are obtained using concurrent chemoradiotherapy (28-30), but information regarding concurrent chemoradiotherapy with HDR brachytherapy is scarce, because most experiences of combining chemotherapy and brachytherapy have been based on LDR treatment. Our current treatment policy is to use concurrent chemoradiotherapy for high-risk early stage, or stage IIb or greater patients. Our results show that the acute side effects are tolerable in patients treated with chemoradiotherapy and that late complications are similar, regardless of the use of chemotherapy. The 3 Gy fraction size used in our study is one of the smallest used in Korea, and was adopted to maximize biological advantages compared to 5 Gy. However, this prolongs the overall treatment time, which may directly affect the tumor control rate. Our patients in the 3 Gy fraction arm were treated for a median 65 days, which was seven days longer than those in the 5 Gy arm. Although failure patterns were not different in the two groups in our study, if more patients had been enrolled or the follow-up extended, both groups may have revealed different failure patterns.
In conclusion, our results showed that the two HDR fractionations were comparable in view of survival, failure patterns, and late complications. The development of late complications was found to depend on the absolute BED at each reference point rather than the fraction size. The fractionation regimen involving a 5 Gy fraction size and application twice a week seems to be both safe and effective for the treatment of uterine cervical cancer, and offers a shorter treatment duration.
Figures and Tables
Table 5
Incidence of acute complications of grade 0-2 in patients treated with concurrent chemoradiotherapy (%)
References
1. Ministry of Health and Welfare. Annual report of the Korea Central Cancer Registry Program: Based on registered data from 134 hospitals, January-December, 2001. 2002. Korea:
2. Fu KK, Phillips TL. High-dose-rate versus low-dose-rate intracavitary brachytherapy for carcinoma of the cervix. Int J Radiat Oncol Biol Phys. 1990. 19:791–796.

3. Patel FD, Sharma SC, Negi PS, Ghoshal S, Gupta BD. Low dose rate vs. high dose rate brachytherapy in the treatment of carcinoma of the uterine cervix: a clinical trial. Int J Radiat Oncol Biol Phys. 1994. 28:335–341.

4. Huh SJ. Current status of high dose rate brachytherapy in cervical cancer in Korea and optimal treatment schedule. J Korean Soc Ther Radiol Oncol. 1998. 16:357–366.
5. Nag S, Erickson B, Thomadsen B, Orton C, Demanes JD, Petereit D. The American Brachytherapy Society recommendations for high-dose-rate brachytherapy for carcinoma of the cervix. Int J Radiat Oncol Biol Phys. 2000. 48:201–211.

6. Lee SW, Suh CO, Chung EJ, Kim GE. Dose optimization of fractionated external radiation and high-dose-rate intracavitary brachytherapy for FIGO stage IB uterine cervical carcinoma. Int J Radiat Oncol Biol Phys. 2002. 52:1338–1344.

7. Sood BM, Gorla G, Gupta S, Garg M, Deore S, Runowicz CD, Fields AL, Goldberg GL, Anderson PS, Vikram B. Two fractions of high-dose-rate brachytherapy in the management of cervix cancer: clinical experience with and without chemotherapy. Int J Radiat Oncol Biol Phys. 2002. 53:702–706.

8. Ferrigno R, dos Santos Novaes PE, Pellizzon AC, Maia MA, Fogarolli RC, Gentil AC, Salvajoli JV. High-dose-rate brachytherapy in the treatment of uterine cervix cancer. Analysis of dose effectiveness and late complications. Int J Radiat Oncol Biol Phys. 2001. 50:1123–1135.

9. Le Pechoux C, Akine Y, Sumi M, Tokuuye K, Ikeda H, Yajima M, Yamada T, Tanemura K, Tsunematsu R, Ohmi K. High dose rate brachytherapy for carcinoma of the uterine cervix: comparison of two different fractionation regimens. Int J Radiat Oncol Biol Phys. 1995. 31:735–741.
10. Wong FC, Tung SY, Leung TW, Sze WK, Wong VY, Lui CM, Yuen KK, O SK. Treatment results of high-dose-rate remote afterloading brachytherapy for cervical cancer and retrospective comparison of two regimens. Int J Radiat Oncol Biol Phys. 2003. 55:1254–1264.

11. Chatani M, Matayoshi Y, Masaki N, Teshima T, Inoue T. A prospective randomized study concerning the point A dose in high-dose rate intracavitary therapy for carcinoma of the uterine cervix. The final results. Strahlenther Onkol. 1994. 170:636–642.
12. Dale RG. The application of the linear-quadratic dose-effect equation to fractionated and protracted radiotherapy. Br J Radiol. 1985. 58:515–528.

13. Fowler JF. The linear-quadratic formula and progress in fractionated radiotherapy. Br J Radiol. 1989. 62:679–694.

14. ICRU Report 38. Dose and volume specification for reporting intracavitary therapy in gynecology. 1985. International commission on radiation units and measurements;1–20.
15. Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995. 31:1341–1346.

16. Stitt JA, Fowler JF, Thomadsen BR, Buchler DA, Paliwal BP, Kinsella TJ. High dose rate intracavitary brachytherapy for carcinoma of the cervix: the Madison system: I. Clinical and radiobiological considerations. Int J Radiat Oncol Biol Phys. 1992. 24:335–348.

17. Orton CG. High-dose-rate brachytherapy may be radiobiologically superior to low-dose rate due to slow repair of late-responding normal tissue cells. Int J Radiat Oncol Biol Phys. 2001. 49:183–189.

18. Nag S, Orton C, Young D, Erickson B. The American brachytherapy society survey of brachytherapy practice for carcinoma of the cervix in the United States. Gynecol Oncol. 1999. 73:111–118.

19. Petereit DG, Pearcey R. Literature analysis of high dose rate brachytherapy fractionation schedules in the treatment of cervical cancer: is there an optimal fractionation schedule? Int J Radiat Oncol Biol Phys. 1999. 43:359–366.

20. Nag S, Gupta N. A simple method of obtaining equivalent doses for use in HDR brachytherapy. Int J Radiat Oncol Biol Phys. 2000. 46:507–513.

21. Clark BG, Souhami L, Roman TN, Chappell R, Evans MD, Fowler JF. The prediction of late rectal complications in patients treated with high dose-rate brachytherapy for carcinoma of the cervix. Int J Radiat Oncol Biol Phys. 1997. 38:989–993.

22. Toita T, Kakinohana Y, Ogawa K, Adachi G, Moromizato H, Nagai Y, Maehama T, Sakumoto K, Kanazawa K, Murayama S. Combination external beam radiotherapy and high-dose-rate intracavitary brachytherapy for uterine cervical cancer: analysis of dose and fractionation schedule. Int J Radiat Oncol Biol Phys. 2003. 56:1344–1353.

23. Ogino I, Kitamura T, Okamoto N, Yamasita K, Aikawa Y, Okajima H, Matsubara S. Late rectal complication following high dose rate intracavitary brachytherapy in cancer of the cervix. Int J Radiat Oncol Biol Phys. 1995. 31:725–734.

24. Potter R, Van Limbergen E, Gerstner N, Wambersie A. Survey of the use of the ICRU 38 in recording and reporting cervical cancer brachytherapy. Radiother Oncol. 2001. 58:11–18.
25. Kapp KS, Stuecklschweiger GF, Kapp DS, Poschauko J, Pickel H, Hackl A. Carcinoma of the cervix: analysis of complications after primary external beam radiation and Ir-192 HDR brachytherapy. Radiother Oncol. 1997. 42:143–153.

26. Van Lancker M, Storme G. Prediction of severe late complications in fractionated, high dose-rate brachytherapy in gynecological applications. Int J Radiat Oncol Biol Phys. 1991. 20:1125–1129.

27. Uno T, Itami J, Aruga M, Kotaka K, Fujimoto H, Sato T, Minoura S, Ito H. High dose rate brachytherapy for carcinoma of the cervix: risk factors for late rectal complications. Int J Radiat Oncol Biol Phys. 1998. 40:615–621.

28. Rose PG, Bundy BN, Watkins EB, Thigpen JT, Deppe G, Maiman MA, Clarke-Pearson DL, Insalaco S. Concurrent cisplatin-based radiotherapy and chemotherapy for locally advanced cervical cancer. N Engl J Med. 1999. 340:1144–1153.

29. Whitney CW, Sause W, Bundy BN, Malfetano JH, Hannigan EV, Fowler WC Jr, Clarke-Pearson DL, Liao SY. Randomized comparison of fluorouracil plus cisplatin versus hydroxyurea as an adjunct to radiation therapy in stage IIB-IVA carcinoma of the cervix with negative para-aortic lymph nodes: a Gynecologic Oncology Group and Southwest Oncology Group study. J Clin Oncol. 1999. 17:1339–1348.





 PDF
PDF ePub
ePub Citation
Citation Print
Print













 XML Download
XML Download