Abstract
Traditional risk factors for predicting of cardiovascular disease are not always effective predictors for development of cardiovascular events. This review summarizes several newly developed noninvasive imaging techniques for evaluating carotid plaques and their role in cardiovascular disease risk.
Atherosclerosis is the most frequent cause of coronary and carotid artery disease. Although it rarely causes symptoms in early stages, atherosclerosis can develop at young age and progress throughout a lifetime. The development of atherosclerotic plaques, which occurs at a late stage in atherogenesis, 1) is a progressive process and is caused by the accumulation of lipids and inflammation.2) The composition of atherosclerotic plaques reflects the severity of local atherosclerotic disease. Advanced atherosclerotic plaques contain fibrous tissue, a necrotic lipid-rich core, calcium, and inflammatory cells.3) Destabilization or rupture of atherosclerotic plaques can cause acute thrombosis, leading to life-threatening clinical events such as acute coronary syndrome.4)5) The possibility of rupture is related to characteristics that represent vulnerable plaques, such as a large lipid core, thin fibrous cap, or marked inflammation. 6) Detection of atherosclerotic plaque is critical for preventing future cardiovascular events.
Traditional risk factors like Framingham Risk Score are not always correlated with the development of cardiovascular events.7) Researchers have sought for new imaging techniques for detection of subclinical atherosclerosis. As a result, in addition to the widely used technique of carotid ultrasound, current diagnostic options include coronary artery calcium score, carotid intima-media thickness (cIMT), and carotid plaque. This review summarizes various methods that evaluate carotid plaques using ultrasound and their role in predicting cardiovascular disease risk.
The current American Society of Echocardiography (ASE) guidelines suggest standard screening methods of carotid plaques.8) Circumferential scanning ranging from anterior to posterior angles and imaging the near or far wall of the common carotid artery, bulb, and internal carotid artery segments are required. If a plaque is seen in short axis view, long axis assessment of the plaque is used to corroborate maximum plaque size. Meanwhile, ASE and European Mannheim consensus defined plaque as a focal wall thickening > 50% (or 0.5 mm) of the surrounding IMT, or its cIMT > 1.5 mm.8)9) Plaque may be characterized by its presence or absence, location, thickness, number, irregularity (smooth, irregular, or ulcerated), area, and echodensity (echolucent or echogenic). Carotid plaques have been evaluated by both qualitative (visual) and quantitative methods. Hollander et al.10) identified a correlation between the presence of carotid plaque and an increased risk of stroke and cerebral infarction (about 1.5 fold) irrespective of plaque location. The presence of a carotid plaque was a stronger predictor of coronary heart disease risk than cIMT.11) This shows that qualitative method of evaluating the presence or absence of carotid plaque is important because it may indicate markers of generalized atherosclerosis. However, cardiovascular risk varies widely according to the severity of the carotid plaque. In our clinical cases, carotid plaques were observed in two patients. One patient had a single plaque with low cardiovascular risk (Fig. 1A) and the other patient had multiple complex plaques with recent stroke (Fig. 1B). If the evaluation of carotid plaque is only focused on its presence or absence, it is not possible to determine the difference in risk between the two patients.
To address this issue, many studies have quantified the carotid plaque to more accurately estimate cardiovascular risk (Table 1). Maximum carotid plaque thickness is associated with increased risk of vascular outcomes in a multiethnic cohort. 12) A study by Störk et al.13) looked at the relationship between the total number of carotid plaques and prognosis. They evaluated carotid burden using carotid plaque score (1 point = 1–2 plaques; 2 points = 3–4 plaques; 3 points = 5–6 plaques; and 4 points = 7–12 plaques). As compared to patients with no plaque, the risk of mortality increased 2.9-fold when one to two plaques were present and 4.9-fold when greater than four plaques were present. These findings demonstrated a graded relationship between the number of carotid plaques and mortality. Carotid plaque area, determined by the sum cross-sectional area of all carotid plaques, has also been identified as an independent predictor of future cardiovascular risk.14) Echolucent plaques are lipid-rich, whereas echogenic plaques are rich of fibrous tissue and calcification. Plaque echogenicity can be graded from 1 to 4 based on visual analysis with gradient progressing, from echolucent (defined as a plaque appearing black) to predominant echolucent, predominant echogenic, and, finally, echogenic (defined as a plaque appearing white or almost white).15) Honda et al.16) defined an echolucent plaque as less than -13.4 dB by using integrated backscatter (IBS) analysis. They demonstrated that echolucent carotid plaque with low IBS value was a significant and independent predictor of future coronary events in patients with stable angina. Echolucent plaque was defined by use of the grey-scale median (GSM), a computerized measurement of plaque echogenicity. el-Barghouty et al.17) have contended that a GSM of less than 32 corresponds to a high risk plaque. Additional studies have further confirmed echolucent plaques as predictor of cerebrovascular events.18)19) Prati et al.20) studied whether plaque score was an independent predictor of cerebrovascular ischemic events. The score was based on degree of stenosis and plaque morphology; (degree of stenosis = 0 if lumen obstruction < 40%, = 1 if 40%), (echogenicity = 1 for low echogenicity or echolucent plaque, = 2 for intermediate, and = 3 for hyper-echogenicity), (texture = 0 if homogeneous, and = 1 if heterogeneous), and (surface characteristics = 0 if smooth, = 1 if irregular). A total plaque risk score was calculated from each subject examined, which ranged from 1 to 6. The most powerful predictor of cardiovascular events during their follow-up was total plaque risk score > 4.
Recently, several new ultrasound technologies have been developed for the evaluation of carotid plaques. A study by Lal et al.21) analyzed plaque texture by pixel distribution analysis and histology. Madycki et al.22) found that pixel distribution analysis predicted the risk of perioperative complications more precisely than standard GSM analysis. Another evaluation technique relies on contrast-enhanced ultrasound imaging of the carotid vasa vasorum.23) Coli et al.24) demonstrated a strong correlation between histologic density and the degree of contrast-agent enhancement by ultrasound imaging. Contrast enhanced ultrasound may also be used to detect subclinical atherosclerosis in the carotid arteries, serving as an additional method for the detection of carotid plaque ulceration.25)26)
The final technique quantifies carotid plaque using a three-dimensional (3D) measurement of plaque volume. 3D measurement demonstrated a higher negative predictive value and sensitivity for coronary artery disease (CAD) than two-dimensional (2D) measurement. In particular, total 3D plaque volumes less than 0.09 mL could predict the absence of CAD.27) 3D plaque volume is significantly more sensitive to changes with therapy than measurements of cIMT or total plaque area.28) Sillesen et al.29) found that carotid plaque volume was more closely correlated with coronary atherosclerosis than cIMT, abdominal aortic diameter, or ankle-brachial index in a large number of patients (n = 6101).
Traditional cardiovascular disease risk factors such as old age, gender, smoking, cholesterol levels, blood pressure, and diabetes have long been used to predict cardiovascular risk, but this method has its limitations. Imaging modalities such as carotid ultrasound are superior means of predicting cardiovascular risk.30) Measurement of cIMT has been widely used to predict cardiovascular risk, but it may not be useful for risk stratification in the general population. A meta-analysis by Den Ruijter et al.31) demonstrated that, for the general population, the added predictive value of including common cIMT measurement to the Framingham Risk Score was small and not significant. However, in individuals of intermediate risk, the added value was 3.2% in men and 3.9% in women.
Despite such limited evidence of cardiovascular risk prediction, recent guidelines recommend measuring cIMT in asymptomatic patients of intermediate cardiovascular risk. The ASE consensus statement recommends that common cIMT measurement should always be supplemented by thorough scanning of extracranial carotid arteries for carotid plaques for higher diagnostic accuracy of subclinical vascular diseases.8) Although cIMT is a well-known surrogate marker for subclinical atherosclerosis, thickening of cIMT does not necessarily represent subclinical atherosclerosis. cIMT is primarily a result of the hypertensive thickening of smooth muscles in the arterial media layer, rather than the subintimal changes which are indicative of atherosclerosis. Compared with cIMT, presence of carotid plaque may be more representative for atherosclerosis. Atherosclerosis Risk In Communities (ARIC) trial showed that the addition of plaque to cIMT resulted in a net reclassification improvement of 9.9% in the overall population and 21.7% in the intermediate risk group.11) Carotid plaque, compared with cIMT, was shown to predict CAD events more accurately in a meta-analaysis.32) Ultrasound assessment of carotid plaque was found to have a higher diagnostic accuracy than cIMT for the prediction of future myocardial infarction. In addition, the absence of carotid plaque provided greater assurance, with low event rate (4.0%) of 10-year myocardial infarction. The ARIC trial, which evaluated the presence or absence of carotid plaque without quantification, demonstrated that including plaque with cIMT significantly improved risk prediction of coronary heart disease.11)
Subsequent studies used quantified methods to measure plaques and evaluated risk prediction using quantified plaques. In earlier studies, plaque quantification included the thickness, number, surface, area, and texture of plaques which were detailed by 2D ultrasound. These studies showed significantly improved risk prediction compared to cIMT and traditional risk factors. Studies investigating the evaluation of coronary heart disease based on carotid plaque score also demonstrated its efficacy as a significant predictor for CAD.
Recent developments in new technologies for evaluating carotid plaques show promise in improving our ability to predict the risk of cardiovascular disease. 3D ultrasound shows particular prognostic promise. 3D measurement of carotid plaque volume is more sensitive to the presence of carotid plaque because it creates simultaneous visualization of carotid plaque using all 3 planes, decreasing the odds of missing any plaques present. It also allows for a more accurate assessment of total plaque volume and enhances the ability to observe changes in plaque burden compared to 2D techniques. Further research is required to confirm this, but present evidence suggests that 3D plaque volume measurement will be a stronger predictor of cardiovascular events in patients.
Ultrasound is used for initial evaluation of carotid plaque. The advantage of ultrasound is its low cost and that it is relatively safe as an imaging modality. The presence of carotid plaque on ultrasound is a better predictor of future cardiovascular events compared to cIMT. The disadvantage of ultrasound is that it is dependent on the operator's skill and image quality. The limitation of using carotid plaque in clinical practice is that plaque quantification, such as morphology and volume, has not been well studied yet.
Quantification of carotid plaques is an important step in assessing subclinical atherosclerosis for the prediction of future coronary heart disease. While some studies suggest a plaque scoring method for improved assessment, there is no consensus classification system for carotid plaque severity. In the future, 3D ultrasound is expected to be a more reliable means of evaluating plaque burden and predicting cardiovascular risk.
Figures and Tables
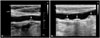 | Fig. 1Carotid ultrasound showed carotid plaques in two patients, one patient has an asymptomatic single plaque with low cardiovascular risk (A), and the other patient has multiple complex plaques with recent stroke (B). The image (A) shows a small amount of smooth homogenous plaque (arrow) located on the far wall of the proximal CCA. In contrast, the image (B) demonstrates moderate to large amount of heterogeneous multiple plaques (arrows) with focal calcification and irregular surface, in mid CCA. CCA: common carotid artery. |
References
1. Hegele RA. The pathogenesis of atherosclerosis. Clin Chim Acta. 1996; 246:21–38.
2. Vaya J. The association between biomarkers in the blood and carotid plaque composition-focusing on oxidized lipids, oxysterols and plaque status. Biochem Pharmacol. 2013; 86:15–18.
3. Kragel AH, Reddy SG, Wittes JT, Roberts WC. Morphometric analysis of the composition of atherosclerotic plaques in the four major epicardial coronary arteries in acute myocardial infarction and in sudden coronary death. Circulation. 1989; 80:1747–1756.
4. Schaar JA, Muller JE, Falk E, Virmani R, Fuster V, Serruys PW, Colombo A, Stefanadis C, Ward Casscells S, Moreno PR, Maseri A, van der Steen AF. Terminology for high-risk and vulnerable coronary artery plaques. Report of a meeting on the vulnerable plaque, June 17 and 18, 2003, Santorini, Greece. Eur Heart J. 2004; 25:1077–1082.
5. Naghavi M, Libby P, Falk E, Casscells SW, Litovsky S, Rumberger J, Badimon JJ, Stefanadis C, Moreno P, Pasterkamp G, Fayad Z, Stone PH, Waxman S, Raggi P, Madjid M, Zarrabi A, Burke A, Yuan C, Fitzgerald PJ, Siscovick DS, de Korte CL, Aikawa M, Juhani Airaksinen KE, Assmann G, Becker CR, Chesebro JH, Farb A, Galis ZS, Jackson C, Jang IK, Koenig W, Lodder RA, March K, Demirovic J, Navab M, Priori SG, Rekhter MD, Bahr R, Grundy SM, Mehran R, Colombo A, Boerwinkle E, Ballantyne C, Insull W Jr, Schwartz RS, Vogel R, Serruys PW, Hansson GK, Faxon DP, Kaul S, Drexler H, Greenland P, Muller JE, Virmani R, Ridker PM, Zipes DP, Shah PK, Willerson JT. From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: part I. Circulation. 2003; 108:1664–1672.
6. Shah PK. Mechanisms of plaque vulnerability and rupture. J Am Coll Cardiol. 2003; 41:4 Suppl S. 15S–22S.
7. Schlendorf KH, Nasir K, Blumental BS. Limitations of the Framingham risk score are now much clearer. Prev Med. 2009; 48:115–116.
8. Stein JH, Korcarz CE, Hurst RT, Lonn E, Kendall CB, Mohler ER, Najjar SS, Rembold CM, Post WS. American Society of Echocardiography Carotid Intima-Media Thickness Task Force. Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: a consensus statement from the American Society of Echocardiography Carotid Intima-Media Thickness Task Force. Endorsed by the Society for Vascular Medicine. J Am Soc Echocardiogr. 2008; 21:93–111. quiz 189-90.
9. Touboul PJ, Hennerici MG, Meairs S, Adams H, Amarenco P, Bornstein N, Csiba L, Desvarieux M, Ebrahim S, Fatar M, Hernandez Hernandez R, Jaff M, Kownator S, Prati P, Rundek T, Sitzer M, Schminke U, Tardif JC, Taylor A, Vicaut E, Woo KS, Zannad F, Zureik M. Mannheim carotid intima-media thickness consensus (2004-2006). An update on behalf of the Advisory Board of the 3rd and 4th Watching the Risk Symposium, 13th and 15th European Stroke Conferences, Mannheim, Germany, 2004, and Brussels, Belgium, 2006. Cerebrovasc Dis. 2007; 23:75–80.
10. Hollander M, Bots ML, Del Sol AI, Koudstaal PJ, Witteman JC, Grobbee DE, Hofman A, Breteler MM. Carotid plaques increase the risk of stroke and subtypes of cerebral infarction in asymptomatic elderly: the Rotterdam study. Circulation. 2002; 105:2872–2877.
11. Nambi V, Chambless L, Folsom AR, He M, Hu Y, Mosley T, Volcik K, Boerwinkle E, Ballantyne CM. Carotid intima-media thickness and presence or absence of plaque improves prediction of coronary heart disease risk: the ARIC (Atherosclerosis Risk In Communities) study. J Am Coll Cardiol. 2010; 55:1600–1607.
12. Rundek T, Arif H, Boden-Albala B, Elkind MS, Paik MC, Sacco RL. Carotid plaque, a subclinical precursor of vascular events: the Northern Manhattan Study. Neurology. 2008; 70:1200–1207.
13. Störk S, van den Beld AW, von Schacky C, Angermann CE, Lamberts SW, Grobbee DE, Bots ML. Carotid artery plaque burden, stiffness, and mortality risk in elderly men: a prospective, population-based cohort study. Circulation. 2004; 110:344–348.
14. Spence JD, Eliasziw M, DiCicco M, Hackam DG, Galil R, Lohmann T. Carotid plaque area: a tool for targeting and evaluating vascular preventive therapy. Stroke. 2002; 33:2916–2922.
15. Mathiesen EB, Bønaa KH, Joakimsen O. Low levels of high-density lipoprotein cholesterol are associated with echolucent carotid artery plaques: the tromsø study. Stroke. 2001; 32:1960–1965.
16. Honda O, Sugiyama S, Kugiyama K, Fukushima H, Nakamura S, Koide S, Kojima S, Hirai N, Kawano H, Soejima H, Sakamoto T, Yoshimura M, Ogawa H. Echolucent carotid plaques predict future coronary events in patients with coronary artery disease. J Am Coll Cardiol. 2004; 43:1177–1184.
17. el-Barghouty N, Nicolaides A, Bahal V, Geroulakos G, Androulakis A. The identification of the high risk carotid plaque. Eur J Vasc Endovasc Surg. 1996; 11:470–478.
18. Kitta Y, Obata JE, Takano H, Nakamura T, Kodama Y, Fujioka D, Saito Y, Kawabata K, Mende A, Kobayashi T, Kugiyama K. Echolucent carotid plaques predict in-stent restenosis after bare metal stenting in native coronary arteries. Atherosclerosis. 2008; 197:177–182.
19. Mathiesen EB, Bønaa KH, Joakimsen O. Echolucent plaques are associated with high risk of ischemic cerebrovascular events in carotid stenosis: the tromsø study. Circulation. 2001; 103:2171–2175.
20. Prati P, Tosetto A, Casaroli M, Bignamini A, Canciani L, Bornstein N, Prati G, Touboul PJ. Carotid plaque morphology improves stroke risk prediction: usefulness of a new ultrasonographic score. Cerebrovasc Dis. 2011; 31:300–304.
21. Lal BK, Hobson RW 2nd, Pappas PJ, Kubicka R, Hameed M, Chakhtoura EY, Jamil Z, Padberg FT Jr, Haser PB, Durán WN. Pixel distribution analysis of B-mode ultrasound scan images predicts histologic features of atherosclerotic carotid plaques. J Vasc Surg. 2002; 35:1210–1217.
22. Madycki G, Staszkiewicz W, Gabrusiewicz A. Carotid plaque texture analysis can predict the incidence of silent brain infarcts among patients undergoing carotid endarterectomy. Eur J Vasc Endovasc Surg. 2006; 31:373–380.
23. Feinstein SB. Contrast ultrasound imaging of the carotid artery vasa vasorum and atherosclerotic plaque neovascularization. J Am Coll Cardiol. 2006; 48:236–243.
24. Coli S, Magnoni M, Sangiorgi G, Marrocco-Trischitta MM, Melisurgo G, Mauriello A, Spagnoli L, Chiesa R, Cianflone D, Maseri A. Contrast-enhanced ultrasound imaging of intraplaque neovascularization in carotid arteries: correlation with histology and plaque echogenicity. J Am Coll Cardiol. 2008; 52:223–230.
25. van den Oord SC, ten Kate GL, Akkus Z, Renaud G, Sijbrands EJ, ten Cate FJ, van der Lugt A, Bosch JG, de Jong N, van der Steen AF, Schinkel AF. Assessment of subclinical atherosclerosis using contrast-enhanced ultrasound. Eur Heart J Cardiovasc Imaging. 2013; 14:56–61.
26. ten Kate GL, van Dijk AC, van den Oord SC, Hussain B, Verhagen HJ, Sijbrands EJ, van der Steen AF, van der Lugt A, Schinkel AF. Usefulness of contrast-enhanced ultrasound for detection of carotid plaque ulceration in patients with symptomatic carotid atherosclerosis. Am J Cardiol. 2013; 112:292–298.
27. Johri AM, Chitty DW, Matangi M, Malik P, Mousavi P, Day A, Gravett M, Simpson C. Can carotid bulb plaque assessment rule out significant coronary artery disease? A comparison of plaque quantification by two- and three-dimensional ultrasound. J Am Soc Echocardiogr. 2013; 26:86–95.
28. Spence JD. Technology insight: ultrasound measurement of carotid plaque--patient management, genetic research, and therapy evaluation. Nat Clin Pract Neurol. 2006; 2:611–619.
29. Sillesen H, Muntendam P, Adourian A, Entrekin R, Garcia M, Falk E, Fuster V. Carotid plaque burden as a measure of subclinical atherosclerosis: comparison with other tests for subclinical arterial disease in the High Risk Plaque BioImage study. JACC Cardiovasc Imaging. 2012; 5:681–689.
30. del Sol AI, Moons KG, Hollander M, Hofman A, Koudstaal PJ, Grobbee DE, Breteler MM, Witteman JC, Bots ML. Is carotid intima-media thickness useful in cardiovascular disease risk assessment? The Rotterdam Study. Stroke. 2001; 32:1532–1538.
31. Den Ruijter HM, Peters SA, Anderson TJ, Britton AR, Dekker JM, Eijkemans MJ, Engström G, Evans GW, de Graaf J, Grobbee DE, Hedblad B, Hofman A, Holewijn S, Ikeda A, Kavousi M, Kitagawa K, Kitamura A, Koffijberg H, Lonn EM, Lorenz MW, Mathiesen EB, Nijpels G, Okazaki S, O'Leary DH, Polak JF, Price JF, Robertson C, Rembold CM, Rosvall M, Rundek T, Salonen JT, Sitzer M, Stehouwer CD, Witteman JC, Moons KG, Bots ML. Common carotid intima-media thickness measurements in cardiovascular risk prediction: a meta-analysis. JAMA. 2012; 308:796–803.
32. Inaba Y, Chen JA, Bergmann SR. Carotid plaque, compared with carotid intima-media thickness, more accurately predicts coronary artery disease events: a meta-analysis. Atherosclerosis. 2012; 220:128–133.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download