Abstract
Situs ambiguous is rare congenital anomaly in adults. In 2 adult patients who admitted for different cardiac problems, situs ambiguous with polysplenia was detected. A 42-year-old male admitted for radio frequent catheter ablation of atrial fibrillation, and he had left-sided inferior vena cava (IVC), hepatic segment of IVC interruption with hemiazygos continuation, multiple spleens and intestinal malrotation. And in a 52-year-old female case who was hospitalized due to infective endocarditis after implanting pacemaker for sick sinus syndrome, multiple spleens, left-sided stomach, bilateral liver with midline gallbladder, and left-sided IVC were found. Those findings were consistent with situs ambiguous with polysplenia, but their features were distinctive.
Situs solitus represents the normal position of heart and viscera relative to midline, and situs inversus indicates mirror-image location of the viscera relative to situs solitus.1) Situs ambiguous (SA) is defined as an abnormality which can be considered to be present when the thoracic and abdominal organs are not clearly lateralized.2)
SA is typically associated with complex cardiovascular malformations. Also, splenic abnormalities and intestinal malrotation are common. Thus SA is usually categorized either as splenic morphology - polysplenia (bilateral left-sidedness, usually with multiple spleens, left isomerism, namely, polysplenia syndrome) or as asplenia (bilateral right-sidedness, with absence of spleen, right isomerism, namely, asplenia syndrome).2)
A 42-year-old male was admitted for radiofrequency ablation of atrial fibrillation (AF). He was diagnosed as AF 4 years ago and took anti-arrhythmic agent, beta blocker and anticoagulant. He had left side weakness due to cerebral infarction of right middle cerebral artery territory 3 years ago. He had a history of hypertension. His family had no history of diseases or congenital abnormality. He had no other symptoms but intermittent palpitation. His heart sound was irregular, but nothing particular was revealed on other physical and laboratory examinations. Double the shadow of thoracic aorta and widening state of superior mediastinum were shown in chest X-ray, but there were no other remarkable matters (Fig. 1). Initial electrocardiogram showed AF with moderate ventricular response (average 60-80 beats/min). On transthoracic and transesophageal echocardiography, no structural cardiac abnormalities were revealed. It seemed that hepatic vein was connected to right atrium through inferior vena cava (IVC) as usual. There was no pulmonary hypertension. We checked coronary multidirectional computed tomography (MDCT) to identify the anatomical variations of the patient's coronary vessels and heart before the ablation procedure. There was a tubular structure which was paralleling with descending thoracic aorta. It was supposed to be an IVC interruption with hemiazygos continuation (Fig. 2A). Hepatic veins were drained to right atrium. Abdomen computed tomography (CT) was performed to evaluate other combined abnormality. Multiple
and round soft tissue densities were detected around the spleen, which were enhanced at the same degree of the spleen. Left-sided colon and right-sided small bowels indicated intestinal malrotation. IVC was located at the left side of aorta, and the hepatic segment of IVC was absent (Fig. 2B-D). By means of venography of IVC through right femoral vein, the interruption of the thoracic IVC with hemiazygos continuation along with aortic arch was confirmed (Fig. 3). All those findings were compatible with SAP.
A 52-year-old female was presented with a history of intermittent fever for a month. She had been DDDR-type pacemaker insertion state for last 8 months due to sick sinus syndrome. Her family history was non-specific. She had no other symptoms and signs of fever. Nothing specific was shown on her physical and laboratory examinations. Her chest X-ray showed no significant lesions, except gastric air detected under the right side of diaphragm and hepatic shadow in the left side abnormally (Fig. 4). Liver dynamic CT was checked to identify the anatomy of her abdominal organs. The symmetric liver and gallbladder with multiple sandy stones were midline. Multiple spleens and stomach were located at the right side of abdomen. Superior mesenteric vein was unusually located anterior to the superior mesenteric artery. The left-sided IVC was crossed the aorta at the level of diaphragm and drained into right atrium (Fig. 5). There was no intraabdominal lesion to develop fever. During hospitalization, methicillin resistant staphylococcus epidermidis was repeatedly incubated on blood cultures. She was referred us for an echocardiographic examination to find any evidence of infection in her heart. Echocardiogram revealed that large multiple mobile vegetations which were attached on the right ventricular pacemaker lead. The vegetations were prolapsed through the tricuspid valve, and the largest diameter of them was 20 mm. Coronary MDCT for the anatomical confirmation of vascular structure was checked before heart surgery. There was left-sided IVC, but no IVC interruption. She got surgery for removal of infected pacemaker lead and vegetation on tricuspid valve. After 4 weeks of antibiotics therapy, there was no longer pathogen growth in blood culture.
Rose et al.4) estimated the minimal incidence of SA 1/40,000 live births. However Gatrad et al.5) obtained 1/24,000 affected in an English population and 1/2,700 in a highly inbred Asian population. The overall prognosis of children with SA had fatally poor prognosis in their early age, which result from the degree of congenital heart diseases.6)
SAP have congenital heart disease in 50-100% of cases.1) In a study of the spectrum of cardiac abnormalities in the 170 fetes with SAP, complete atrio-ventricular septal defect (68%), complete heart block (38%), double outlet right ventricle (23%), right ventricular outflow tract obstruction (21%) and total anomalous pulmonary vein (5%) drainage were detected, and only 58% of 170 survived.7) Therefore, the case reports of SA in middle aged adults are extremely rare. The abnormal arrangement of the abdominal organs was present up to 50% in a review of 146 autopsied cases of SAP.1) But, such cases without congenital cardiac defects in SAP was also rarely reported previously.
There were 5 case reports of adults' SAP in Korea from 1997 to 2010. These cases were different from one another in their compositions of organ arrangement and the mean age of these cases was 25.5 years. Three cases had congenital disease, noncompaction of the ventricular myocardium,8) coarctation of aorta with bicuspid aortic valve9) and intraluminal duodenal diverticulum,10) which were too minor to be detected until adulthood. In 2 cases, they were incidentally detected while respectively evaluating dyspnea due to congestive heart failure with atrial fibrillation11) and constiptation.12)
Among venous malformation, the congenital interruption of the IVC is usually found in about 80% of patients SAP.13) The most typical IVC interruption is complete absence of the hepatic segment of the IVC with azygos continuation.14) Among the 5 Korean adults mentioned above, the IVC interruption was presented in 4 cases, IVC was drained to hemiazygous vein in just one case.8-11) Venous malformations in SAP can be accompanied by various types which have usually no functional problems. But they do in some special circumstances as the 2 followings. In first case, a patient was found to be interrupted IVC while inserting catheter into right femoral vein for radiofrequency ablation of atrio-venticular nodal reentrant tachycardia, only to give the procedure up.15) Secondly, a pacemaker was failed to be inserted in general approach because of persistent left superior vena cava which can be accompanied by SAP.16)
Consequently, the congenital abnormality does not always cause symptoms or medical problems in adults. However, these anatomical misarrangements can cause confusion in diagnosis and can bring problems during invasive procedure. Therefore, careful analysis of systemic anomalies is necessary to be done in medical approaching, especially for all patients expected surgical or medical interventions.14)
References
1. Fulcher AS, Turner MA. Abdominal manifestations of situs anomalies in adults. Radiographics. 2002; 22:1439–1456. PMID: 12432114.
2. Jacobs JP, Anderson RH, Weinberg PM, Walters HL 3rd, Tchervenkov CI, Del Duca D, Franklin RC, Aiello VD, Béland MJ, Colan SD, Gaynor JW, Krogmann ON, Kurosawa H, Maruszewski B, Stellin G, Elliott MJ. The nomenclature, definition and classification of cardiac structures in the setting of heterotaxy. Cardiol Young. 2007; 17(Suppl 2):1–28. PMID: 18039396.
3. Brandenburg VM, Krueger S, Haage P, Mertens P, Riehl J. Heterotaxy syndrome with severe pulmonary hypertension in an adult. South Med J. 2002; 95:536–538. PMID: 12005012.
4. Rose V, Izukawa T, Moës CA. Syndromes of asplenia and polysplenia. A review of cardiac and non-cardiac malformations in 60 cases withspecial reference to diagnosis and prognosis. Br Heart J. 1975; 37:840–852. PMID: 1191445.
5. Gatrad AR, Read AP, Watson GH. Consanguinity and complex cardiac anomalies with situs ambiguus. Arch Dis Child. 1984; 59:242–245. PMID: 6712272.
6. Peoples WM, Moller JH, Edwards JE. Polysplenia: a review of 146 cases. Pediatr Cardiol. 1983; 4:129–137. PMID: 6878069.
7. Pepes S, Zidere V, Allan LD. Prenatal diagnosis of left atrial isomerism. Heart. 2009; 95:1974–1977. PMID: 19304670.
8. Cho YH, Jin SJ, Je HC, Yoon YW, Hong BK, Kwon HM, Kim TH, Rim SJ. A case of noncompaction of the ventricular myocardium combined with situs ambiguous with polysplenia. Yonsei Med J. 2007; 48:1052–1055. PMID: 18159602.
9. Sohn GH, Byun KM, Han HJ, Kim HJ, Choi JO, Lee SC, Park SW. A case of coarctation of aorta with left isomerism associated with inferior vena cava interruption and polysplenia. J Cardiovasc Ultrasound. 2007; 15:27–29.
10. Kim JH, Seo HI, Kim HS, Kim DH, Jeon TY, Song GA, Kim S. Heterotaxia syndrome with intraluminal duodenal diverticulum. J Korean Surg Soc. 2010; 79:75–78.
11. Cheon IS, Rhee YP, Choi BR, Lee SS, Jung WT, Lee JD, Choi DJ, Hwang JY, Seo BG, You JJ. Hemiazygos continuation of left inferior vena cava draining into the right atrium via persistent left superior vena cava: a variant of polysplenia syndrome mimicking aortic dissection. Korean Circ J. 1998; 28:440–447.
12. Park YB, Kim JI, Cheung DY, Cho SH, Park SH, Han JY, Kim JK. A case of multiple intraabdominal anomalies including nonrotation of the intestine and a preduodenal portal vein. Korean J Med. 2007; 73:548–551.
13. Applegate KE, Goske MJ, Pierce G, Murphy D. Situs revisited: imaging of the heterotaxy syndrome. Radiographics. 1999; 19:837–852. discussion 853-4. PMID: 10464794.
14. Bartram U, Fischer G, Kramer HH. Congenitally interrupted inferior vena cava without other features of the heterotaxy syndrome: report of five cases and characterization of a rare entity. Pediatr Dev Pathol. 2008; 11:266–273. PMID: 17378673.
15. Miranda R, Simpson CS, Nolan RL, Diez JC, Michael KA, Redfearn DP, Baranchuk A. Superior approach for radiofrequency ablation of atrio-ventricular nodal reentrant tachycardia in a patient with anomalous inferior vena cava and azygos continuation. Europace. 2010; 12:908–909. PMID: 20207745.
16. Lee S, Cha JH, Han DH. Persistent left superior caval vein with absent right superior caval vein: importance of awareness. Cardiol Young. 2011; 1–3.
Fig. 1
Double the shadow of thoracic aorta (bold arrows) and widening state of superior mediastinum (sharp arrows) were shown in chest X-ray.

Fig. 2
(A) Coronary multidirectional computed tomography: There was an 1.7 cm sized, round, tubular structure which was paralleling with descending thoracic aorta (white arrows). (B-D) On abdomen computed tomography: (B) A dilated hemiazygos vein runs posterior to the descending aorta. (C) Multiple, well-defined round soft tissue densities were detected around the spleen (black stars). IVC was located at the left side of aorta, and hepatic segment of IVC was absent. (D) Left-sided colon (hollow arrow) and right-sided small bowels (bold arrow) indicated gastrointestinal malrotation. IVC: inferior vena cava.

Fig. 3
Venography of IVC through right femoral vein: Interruption of the thoracic IVC with hemiazygos continuation (arrows) along with aortic arch was confirmed. Enlarged hemiazygos vein drained into left brachiocephalic vein and then to superior vena cava. Infra-hepatic segment of IVC was intact. IVC: inferior vena cava.

Fig. 4
The chest X-ray of 52-year-old woman showed gastric air under the right side of diaphragm (arrows), and hepatic shadow in the left side abnormally.
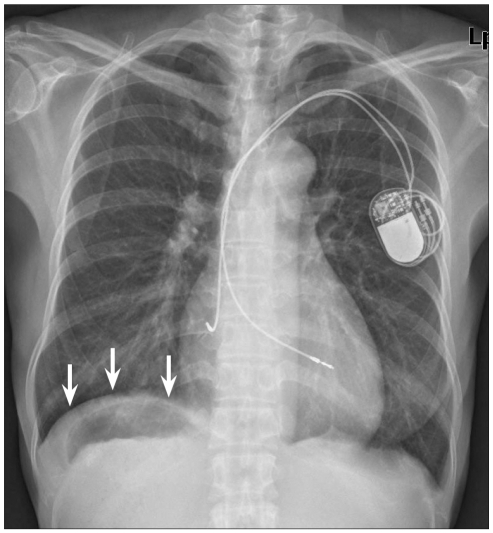
Fig. 5
Liver dynamic computed tomography. A: There were midline symmetric liver (L) and multiple spleens (black stars) and stomach (S) are located at the right side of abdomen. B: Multiple sandy stones in midline gallbladder. Superior mesenteric vein was unusually located anterior to the superior mesenteric artery. C: The left sided inferior vena cava was crossing the aorta at the level of diaphragm and was drained to right atrium. D: Liver was observed in both sides relative to midline.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download