Abstract
Deep vein thrombosis (DVT) is a predisposing condition of pulmonary embolism which can be fatal. Usually, DVT is found in the lower extremities. However, DVT can be occurred in the upper extremities. The usual predisposing conditions of the upper extremity DVT include insertion of central venous catheters and pacemaker wires. Here, we report a case of upper extremity DVT after in vitro fertilization and embryo transfer. The patient was successfully controlled with subcutaneous administration of low molecular weight heparin.
With an increased risk for developing fatal pulmonary thromboembolism, deep vein thrombosis (DVT) is an important clinical entity.1) It can be occurred as complications of insertion of central venous catheters and pacemaker wires.2)3) However, it can be occurred in the pregnancy or as a complication of assisted reproductive techniques (ART). Here, we report a case of DVT of upper extremity after in vitro fertilization (IVF) and embryo transfer (ET).
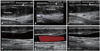
A 34-year-old woman was admitted to our hospital complaining of tingling sense and painful swelling of the right upper extremity for 5 days. The patient diagnosed as primary infertility and underwent IVF after stimulation with a gonadotropin-releasing hormone analog 1 month ago. The IVF was successful with implantation of 3 oocytes. Several days after IVF, she developed slight abdominal distention and dyspnea. Her symptoms persisted for three weeks with conventional treatment. Five days ago, the patient felt edema and pain in her right neck, pain in the subclavian area and tingling sensations with numbness in the right arm. On admission, her body temperature was 37.2℃, pulse rate was 96 bpm, respiratory rate was 20/min, and blood pressure was 108/68 mmHg. Complete blood count showed white blood cell count 11.0×103/uL, hemoglobin 11.4 g/dL and platelet count 167×103/µL. Laboratory results of cardiac enzyme and blood chemistry (SMA20) were within normal range. The hypercoagulability profiles were negative for any deficiencies including protein C and S activities, anti-cardiolipin antibodies (IgG and IgM), lupus anticoagulant, antithrombin III and factor V leiden genotype. An ultrasonographic scan showed thrombosis of the right internal jugular vein extended from the midportion of the vein to the conjoined portion of brachiocephalic and subclavian veins with partial obstruction of the lumen (Fig. 1A, B and C) (Fig. 2A and B). Though there was a risk of developing pulmonary thromboembolism, computerized tomography was not performed due to the increased risk of fetal loss. Fortunately, there was no evidence of increased pulmonary arterial pressure assessed by the echocardiography.
The patient was treated with subcutaneous low-molecular weight heparin [LMWH; nadroparin (Fraxiparine™); Sanofi-Aventis, Belgium] twice a day, which improved her symptoms after 5 days. She was maintained with LMWH twice a day until delivery and she had twin babies without further complications. The follow up ultrasound scan showed normalized internal jugular vein (Fig. 1 D, E and F) (Fig. 2C and D).
Compared to lower extremity DVT, upper extremity DVT (UEDVT) is uncommon disease and represents about 10% of all DVTs.2) It can be associated with mechanical or anatomical factors (e.g. insertion of central venous catheters or narrowing of veins by cervical rib) and hypercoagulable states (e.g. antithrombin deficiency, malignancy and antiphospholipid syndromes).4) Moreover, it can be occurred after ART.4)5) Exogenous gonadotropins and gonadotropin-releasing analogs are used for the follicular induction and maturation in ART.6) Some patients who undergo ART develop ovarian hyperstimulation syndrome (OHSS), which is characterized by ovarian enlargement, hydrothorax, ascites, hemoconcentration, renal impairment, hepatic dysfunction, hypoalbuminemia, and venous thrombosis.6) It can be occurred during 2% to 6% of treatment cycles. Rarely, the hypercoagulable state produced by OHSS may cause DVT.5) Because her hypercoagulability profiles were normal and she underwent IVF and ET, the UEDVT can be associated with ART. However, her symptoms associated with OHSS were mild.
UEDVT can be associated with pulmonary embolism (PE) and its incidence was reported about up to one third of patients with UEDVT.1) Because the patient's vital signs were normal and there was a little evidence of PE, the possibility of PE was assessed as low. Moreover echocardiographic exam showed normal right ventricular function and normal pulmonary arterial pressure assessed by maximal tricuspid regurgitation velocity.
The patient was treated with LMWH inpatient and outpatient settings. Because LMWH has improved bioavailability, longer half-life, and dose-independent renal clearance, it is associated with weight-based subcutaneous administration, a more predictable anticoagulant action, and making unmonitored.7) LMWH has emerged as an effective alternative to conventional unfractionated heparin as initial therapy for DVT.8) It is also suitable for outpatient therapy because of improved bioavailability and more predictable anticoagulant action.9) Moreover, serious potential complications of conventional heparin therapy seem less common with LMWH.10) Because LMWH does not cross the placental barrier, available data support the safety of LMWH for the developing fetus.11)12) So LMWH was successfully used for initial treatment for UEDVT and secondary prophylaxis of PE during pregnancy in this patient.
Figures and Tables
Fig. 1
The longitudinal image of the right neck shows thrombosis of the right internal jugular vein with partial obstruction of the lumen (A: before compression, B: color flow mapping, and C: after compression). The follow up ultrasound scan reveals normalized internal jugular vein (D: before compression, E: color flow mapping, and F: after compression).
References
1. Prandoni P, Polistena P, Bernardi E, Cogo A, Casara D, Verlato F, Angelini F, Simioni P, Signorini GP, Benedetti L, Girolami A. Upper-extremity deep vein thrombosis. Risk factors, diagnosis, and complications. Arch Intern Med. 1997. 157:57–62.

2. Bernardi E, Pesavento R, Prandoni P. Upper extremity deep venous thrombosis. Semin Thromb Hemost. 2006. 32:729–736.

3. Mustafa S, Stein PD, Patel KC, Otten TR, Holmes R, Silbergleit A. Upper extremity deep venous thrombosis. Chest. 2003. 123:1953–1956.
4. Chan WS, Ginsberg JS. A review of upper extremity deep vein thrombosis in pregnancy: unmasking the 'ART' behind the clot. J Thromb Haemost. 2006. 4:1673–1677.

5. Lamon D, Chang CK, Hruska L, Kerlakian G, Smith JM. Superior vena cava thrombosis after in vitro fertilization: case report and review of the literature. Ann Vasc Surg. 2000. 14:283–285.

6. Whelan JG 3rd, Vlahos NF. The ovarian hyperstimulation syndrome. Fertil Steril. 2000. 73:883–896.

7. Hirsh J, Warkentin TE, Shaughnessy SG, Anand SS, Halperin JL, Raschke R, Granger C, Ohman EM, Dalen JE. Heparin and low-molecular-weight heparin: mechanisms of action, pharmacokinetics, dosing, monitoring, efficacy, and safety. Chest. 2001. 119:64S–94S.

8. McRae SJ, Ginsberg JS. Initial treatment of venous thromboembolism. Circulation. 2004. 110:I3–I9.

9. Harrison L, McGinnis J, Crowther M, Ginsberg J, Hirsh J. Assessment of outpatient treatment of deep-vein thrombosis with low-molecular-weight heparin. Arch Intern Med. 1998. 158:2001–2003.

10. Lindhoff-Last E, Nakov R, Misselwitz F, Breddin HK, Bauersachs R. Incidence and clinical relevance of heparin-induced antibodies in patients with deep vein thrombosis treated with unfractionated or low-molecular-weight heparin. Br J Haematol. 2002. 118:1137–1142.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download