Abstract
Background
Granulocyte stimulating factor (G-CSF) and bone marrow mononuclear cells (BM-MNC) were reported to improve cardiac function after myocardial infarction (MI). This study was to examine their combined beneficial effects and mechanisms of actions in reperfused MI, which have not been verified yet.
Methods
Fifteen pigs were divided into 3 groups after a 1-hour balloon occlusion and reperfusion of the left anterior descending coronary artery. G1 (n=5) was a control, G2 (n=5) was a G-CSF injected group (10 ug/kg/day, from day1 to day7 after MI), and G3 (n=5) was an autologous intracoronary BM-MNC infused group after G-CSF treatment.
Results
Modified wall motion indices by echocardiography were similar among 3 groups at 24 hours after MI. However, they improved significantly in G2 and G3 at 35days after MI (p<0.05). The percentage of infarct area/left ventricular myocardial area measured from a triphenyltetrazolium chloride (TTC) stain was lower in G3 than in G1 or G2 (p=0.026). The number of vWF-positive vessels and the expressions of vWF and VE cardherin by RT-PCR were higher in G3 and G2 than in G1 (p<0.05). The number of TUNEL-positive cells and bcl2/bax ratio were not significantly different among 3 groups.
Since ventricular remodeling after myocardial infarction (MI), which causes ventricular dysfunction, is strongly associated with the prognosis1)2), the prevention of remodeling is an important therapeutic concern in the management of myocardial infarction. Ventricular remodeling is usually determined by infarct size, ventricular loading condition, and infarct artery patency.1-3) Although rapid reperfusion of the infarct-related coronary artery is of great importance in salvaging myocardium and limiting infarct size, there are cases of delayed reperfusion and those of irreversible myocardial damage or ventricular dysfunction even after optimal reperfusion in clinical settings. Medically, angiotensin converting enzyme inhibitors and beta blockers can prevent ventricular remodeling and reduce mortality in patients with myocardial infarction.2)4) However, the loss of myocytes and the resulting scar formation are irreversible processes and cannot be regenerated by these agents.2-16)
Recombinant human granulocyte colony stimulating factor (rh G-CSF) is known to proliferate bone marrow stem cells and mobilize hematopoietic bone marrow stem cells into the peripheral blood. They have the capacity to differentiate into cardiac myocytes and vascular smooth muscle cells.17-21) It has been reported that rh G-CSF improved cardiac function and reduced ventricular remodeling by angiogenesis and regenerating myocardium in mice and pigs with permanent left anterior descending artery (LAD) occlusion.20-22) In addition, it was suggested that G-CSF acted directly on cardiomyocytes through G-CSF receptors and promoted their survival after myocardial infarction.7)
Transplantation of bone marrow derived stem cells has been suggested to improve cardiac function after acute myocardial infarction in several experimental and clinical studies.5-16) Bone marrow stem cells can differentiate into cardiomyocyte-like cells23)24) and they supply angioblasts, angiogenic factors, angiogenic ligands, and cytokines, which induce neovascularization in the ischemic tissue.8)
However, the possible combined beneficial effects of both G-CSF and BM-MNC on the injured myocardium have not been assessed and the action mechanism of G-CSF and BM-MNC have not been clearly defined in reperfused myocardial infarction.
In this study, we examined the combined beneficial effects of G-CSF and BM-MNCs on the injured myocardium and their mechanisms of action in improving cardiac function and preventing cardiac remodeling in reperfused acute myocardial infarction in the closed chest pig model.
Acute myocardial infarction was induced by percutaneous balloon angioplasty in 27 cross breed pigs weighing 18-23 kg. 7 pigs died as a result of refractory ventricular fibrillation during the coronary artery occlusion (Fig. 1A). Among the remaining 20 pigs, 15 were appropriate for both echocardiographic examination and tissue analysis. All the experiments were performed according to the "Revised Guide for the Care and Use of Laboratory Animals Available".25)
The animals were anesthetized with intramuscular injections of ketamine hydrochloride (10-15 mg/kg) and xylazine (10 mg/kg) followed by intravenous injections of diazepam and fentanyl through a superficial ear vein. Bone marrow blood was aspirated from the ileum and a baseline echocardiography was performed. After an anterior neck dissection, a 6 Fr. sheath was inserted into a carotid artery and a 6 Fr. guiding catheter was positioned into the ostium of the left coronary artery under fluoroscopic guidance. After performing a coronary angiography (CAG), a floppy guide wire was advanced into the left anterior descending artery (LAD) and a 3.0 mm×13-18 mm balloon was inflated between the first (D1) and the second diagonal (D2) coronary arteries in order to induce a complete occlusion. After 1 hour of occlusion of the LAD, the balloon was deflated and a CAG was performed to confirm the presence of reperfusion.
All the pigs were divided into three groups:
(1) Control group (Group 1, n=5), a group which occluded LAD for 1 hour followed by reperfusion; (2) G-CSF treated group (Group 2, n=5), 24 hours after reperfused myocardial infarction, subcutaneous rh G-CSF (10 ug/kg/day, for 7 days, Dong-A Pharmaceutical; Seoul, Korea) injected group; (3) G-CSF and bone marrow mononuclear cell treated group (Group 3, n=5); intracoronary autologous bone marrow mononuclear cells (6.7×108±4.7×108) infused group after 7 days of rh G-CSF treatment (10 ug/kg/day, for 7days) after reperfused myocardial infarction.
At 5 to 7 days of rh G-CSF treatment after an acute myocardial infarction with reperfusion, bone marrow blood (50 to 60 mL) was aspirated from the ileum under anesthesia with ketamine, xylazine, diazepam, and fentanyl (N=5). BM-MNCs were separated by Ficoll-density-gradient centrifugation using Lymphocyte Separation Medium (LSM: Cellgro, Mediatech, Herndon, VA, USA).
Heparinized blood was diluted 1:1 in phosphate-buffered saline (PBS) and layered carefully over a lymphocyte separation medium (LSM). The tubes were spun at 3,600 rpm x g for 20 minutes and the mononuclear cell layer was harvested from the plasma-LSM interface. RBCs were removed with RBC buffer and BM-MNCs were washed with PBS. Then the BM-MNC suspension was incubated with 1 µg/mL CM-Dil (Molecular Probes) at 37℃ for 5 minutes and at 4℃ for 15 minutes, then washed 2 times with PBS before resuspension in 8 mL of PBS. The mean number of BM-MNCs collected was 6.7×108±4.7×108.
Within 1 hour of the BM-MNC collection and preparation, the cells were infused directly into the LAD through an over-the-wire angioplasty balloon catheter, which was placed between D1 and D2. A total 8 mL of the cell suspension (6.7×108±4.7×108 BM-MNCs) was infused for 20 minutes. The over-the-wire angioplasty balloon was inflated 4 times for 2 to 3 minutes each time in order to prevent the backflow of cells and to facilitate cell contact with infarcted myocardium for a prolonged time. A 2 mL cell suspension was infused at each inflation.
The numbers of circulating white blood cells and monocytes were counted before and at 1, 3, 7, 14, and 28 days after myocardial infarction. Bone marrow blood cells were counted before and at 3, 7, 14, and 28 days after myocardial infarction.
Echocardiographic studies (Acuson Sequoia C256, 5MHz transducer) were performed before and at 1, 3, 7, 14, and 28 days after myocardial infarction. In a left decubitus position, the left ventricular end-diastolic dimension (LVED), left ventricular end-systolic dimension (LVSD), diastolic interventricular septal wall thickness (IVSTd), and posterior wall thickness (PWTd) were measured by M-mode. Left ventricular end-diastolic area (LVAd), left ventricular end-systolic area (LVAs), and fractional area change (FAC) were measured by 2-dimensional echocardiography at the mid-papillary level of the short axis view. For the analysis of regional wall motion, the anteroseptum, which is between the anterolateral and posteromedial papillary muscles at the papillary level of the short axis view, was divided into 5 segments (Fig. 1B). The function of each segment was assessed visually and scored from 1 to 5 (normal: 1, hypokinesis: 2, akinesis: 3, dyskinesis: 4, and aneurysm: 5) at 24 hours and at 35 days after myocardial infarction. On the basis of this wall motion analysis scheme, a modified wall motion score index (MWMSI) was calculated as follows:
MWMSI=Sum of the wall motion scores of 5 segments/5 segments of the anteroseptum between the anterolateral and posteromedial papillary muscles at the papillary level of the short axis view.
Thirty five days after myocardial infarction, the pigs were sacrificed and the infarct size was estimated using triphenyltetrazolium chloride (TTC) staining. The heart was cut into seven transverse slices, six of which were incubated for 15 minutes at 37℃ in 1% (W/V) TTC solution. One was left for RT-PCR analysis. The infarcted area was measured by computed planimetry (NIH IMAGE 1.63, NIH, ML). The infarct size which was measured from each myocardial slice was calculated as a percentage of the left ventricular (LV) area.
Immunohistochemical staining for endothelial cells of the blood vessels was conducted with the anti-von Willebrand factor (vWF) antibody to evaluate angiogenesis in the myocardium. Tissue sections were de-waxed in xylene and rehydrated through a descending ethanol series. Antigen retrieval was performed by incubating with 20 µg/mL proteinase K solution in a humidified chamber for 20 minutes at 37℃ and for another 20 minutes at room temperature. After washing with PBS containing 0.05% Tween 20 (PBT), the sections were blocked with normal goat serum blocking solution in PBT for 30 min. The sections were stained with anti-vWF (DAKO; Glostrup, Denmark) at a dilution of 1 in 500 for 60 min. The sections were linked with a biotinylated link antibody and labeled with streptavidin (LSAB-Kit, DAKO; Glostrup, Denmark). Finally, the sections were developed with diaminobenzidine (DAB, DAKO; Glostrup, Denmark) for 10 min to generate a brown-colored product and lightly counterstained with hematoxylin (Sigma) prior to mounting with Canada Balsam (Junsei, Japan). To count the number of vessels, 15 fields were chosen randomly from remote (non-infarct), border (ischemic), and infarct regions in each group. The number of capillaries in each section was presented as the mean±SE of blood vessels per unit area (mm2).
For the detection of apoptotic cells, a TUNEL assay was carried out on paraffin sections using the in situ Apoptosis Detection Kit (Takara, Japan). After deparaffinization, the slides were treated with 20 µg/mL of proteinase K for 30 min at room temperature (RT). The sections were then incubated with 3% H2O2 in methanol for 5 min to eliminate any endogenous peroxidase. Each section was treated with terminal deoxynucleotidyl transferase (TdT) buffer solution that contained TdT and fluorescein dUTP for 90 min at 37℃. The sections were incubated with anti-fluorescein isothiocyanate (FITC) horseradish peroxidase (HRP) conjugate for 30 min at 37 centigrade, HRP activity was visualized with diaminobenzidine (DAB) and counterstained with 3% methyl green for 5 min. In order to count the number of apoptotic cells, 30 fields were chosen from remote, border, and infarct regions in each sample. The number of apoptotic cells at each section was presented as the mean±SE of TUNEL-positive cells per 104 nucleated cells.
Total RNA was extracted from infarcted tissue, peri-infarcted tissue, and non-infarcted tissue with Trizol (Gibco-BRL; Grand Island, NY, USA). Contaminating DNA was digested by 1 U of RQ1 DNase (Promega; Madison, WI, USA) as recommended by the manufacturer. The first-strand cDNA was synthesized from 0.5 µg of DNase-treated total RNA using 0.5 µg random hexamers (Invitrogen) and 200 U moloney murine leukemia virus (M-MLV) reverse transcriptase (Invitrogen) at 37℃ for 60 min in a volume of 20 µL. The first strand cDNA (1 µL) was used for PCR amplification in a 25 µL reaction mixture. PCR was performed under the following conditions: 94℃ for 30 seconds, 60℃ for 40 seconds, and 72℃ for 40 seconds, with an additional 7-minutes incubation at 72℃ after cycle completion. The PCR reaction was performed for 28 cycles with glyceraldehyde-3-phosphate dehydrogenase (GAPDH) primers and for 35 cycles with vWF, VE-cadherin, bcl-2, bax primers.
The data is presented as the mean±SD. An SPSS 10.0 program was used for statistical analysis. Hematologic data were analyzed using the Mann-Whitney and Wilcoxon methods. Analysis of Variances (ANOVA) was used for echocardiographic data and histopathological data analysis; and time after death official approval was performed using the Bonferroni and Scheffe methods. Statistical significance was considered to be p<0.05.
The WBC numbers, both in the peripheral blood and bone marrow, increased significantly in the G-CSF treated groups (Group 2 and Group 3) at 5 to 7 days after myocardial infarction (peripheral blood, day 0 vs. days 5 to 7, 16389±74/mm3 vs. 75443±180/mm3; bone marrow, day 0 vs. days 5 to 7, 40864±139/mm3 vs. 83992±190/mm3; p<0.05 ) (Fig. 2A and B). Monocyte numbers, both in the peripheral blood and bone marrow also increased significantly in the G-CSF treated groups (Group 2 and Group 3) at 3 to 7 days after myocardial infarction (peripheral blood, day 0 vs. days 5 to 7, 786±17/mm3 vs. 3631±47/mm3; bone marrow, day 0 vs. day 3, 1866±30 /mm3 vs. 4395±34 /mm3; p<0.05) (Fig. 2C and D). However, these changes were not observed in the control group (peripheral blood WBC, day 0 vs. days 5 to 7, 19864±68/mm3 vs. 17815±78/mm3; bone marrow WBC, day 0 vs. days 5 to 7, 36890±110/mm3 vs. 27730±120 /mm3; p=ns) (Fig. 2).
Morphologically, the infarcted myocardial wall was thin and the left ventricular cavity was markedly dilated in the control group. However, these changes were not prominent in the G-CSF and BM-MNC treated groups (Fig. 3A). The percentage of infarct area/LV myocardium area measured at the sliced left ventricle (sum of 6 slices) was less in the G-CSF+BM MNC treated group (Group 3) than in the control group or the G-CSF only treated group (Group 2 and Group 3) (Group 1 vs. Group 2 vs. Group 3, 9.2±2.1% vs. 7.2±1.1% vs. 1.2±1.2%, p=0.026) (Fig. 3B).
We evaluated the number of vessels by immunohistoechemical staining using anti-vWF Ab and the expressions of mature endothelial cell surface molecule such as vWF and VE-cadeherin by real-time RT-PCR in the infarct area, border area and non-infarct area to determine the mechanism of G-CSF and BM-MNC mediated effect on cardiac function and remodeling after reperfused MI. The number of vWF-positive vessels was higher in G-CSF and G-CSF+ BM MNC group than in control group in normal region (Group 1 vs. Group 2 vs. Group 3, 240.9±4.5/mm2 vs. 263.9±10.8/mm2 vs. 275.4±9.7/mm2, p<0.05), in border region (Group 1 vs. Group 2 vs. Group 3, 130.4±7.4/mm2 vs. 166.2±11.5/mm2 vs. 173.7±9.3/mm2, p<0.05), and infarcted region (Group 1 vs. Group 2 vs. Group 3, 88.2±5.0/mm2 vs. 116.4±9.8/mm2 vs. 127.0±7.8/mm2, p<0.05) (Fig. 4). Real-time RT-PCR revealed that the expressions of endothelial markers (vWF and VE-cadherin) markedly increased in G-CSF and G-CSF+BM MNC group than in control group (Fig. 5A). Expression of vWF mRNA was higher in G-CSF and G-CSF+BM MNC group than in control group in normal region (Group 1 vs. Group 2 vs. Group 3, 1.0±0.06-fold vs. 1.47±0.15-fold vs. 1.62±0.19-fold, p<0.05), in border region (Group 1 vs. Group 2 vs. Group 3, 0.65±0.10-fold vs. 1.34±0.19-fold vs. 1.44±0.10-fold, p<0.05), and infarcted region (Group 1 vs. Group 2 vs. Group 3, 0.99±0.15-fold vs. 1.50±0.07-fold vs. 1.84±0.26-fold, p<0.05) (Fig. 5A).
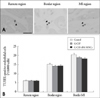
We assessed the number of apoptotic cells by TUNEL assay and mRNA expressions for bcl-2, bax, and the ratio of of bcl-2 mRNA/bax mRNA by real-time RT-PCR in the infarct area, border area and non-infarct area since apoptotic cell death was reported to cause the progression of remodeling after MI and the expansion of MI. The number of TUNEL-positive cells in remote, border and infarcted region was not different among control group, G-CSF group, and G-CSF+BM MNC group (Fig. 6). Furthermore, real-time RT-PCR revealed that bcl-2 mRNA, bax mRNA and the ratios of bcl-2/bax mRNA in remote, border and MI regions were not different among the three groups (Fig. 5B).
Before myocardial infarction, there were no significant differences in the LVEDs, LVSDs, LVAd, LVAs, and FACs among the 3 groups (Table 1). The percent increase in the left ventricular area from the baseline to 35 days after myocardial infarction, measured by 2D echocardiography at the mid-papillary level of the short axis view, tended to be lower in the G-CSF+BM-MNC group than in the control group (Group 1 vs. Group 2 vs. Group 3, 37.5±4.6% vs. 29.6±3.3% vs. 6.7±2.6%; p=0.096) (Table 1). The wall motion index which was calculated at the mid-papillary level of the left ventricle at 24 hours after myocardial infarction was similar among the three groups (Table 2). However, the regional wall motion improved significantly in Group 2 and Group 3 at 35 days after myocardial infarction (Table 2).
The present study demonstrated that the intracoronary infusion of BM-MNCs with G-CSF treatment reduced infarct size, improved ventricular function and prevented ventricular remodeling partly by the enhanced neovascularization in reperfused MI of a closed chest pig model.
G-CSF is a hematopoietic growth factor, which stimulates the bone marrow to produce different types of blood cells by shortening the time of their maturation and increasing the number of cell divisions. In addition to the induction of proliferation and differentiation of hematopoietic stem cells in the bone marrow, G-CSF mobilizes bone marrow stem cells17-21,26-28) and releases angiogenic growth factors, such as vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), and hepatocyte growth factor.29)30) Iwagana et al.22) reported that G-CSF prevented cardiac dysfunction and remodeling after myocardial infarction in pigs through G-CSF-induced activation of Akt and upregulation of VEGF. Recently, it was suggested that G-CSF acted directly on cardiomyocytes to promote their survival after myocardial infarction.7) In that study, G-CSF treatment protected cultured cardiomyocytes from apoptotic cell death, possibly through upregulation of bcl-2 and bcl-xL expression,7)31) which indicated that it had direct protective effects on cardiomyocytes through the G-CSF receptor and the Jak-Stat pathway. Clinically, Kuethe et al.10) reported a significant improvement in regional wall motion and wall perfusion abnormalities with G-CSF treatment in patients after acute myocardial infarction.
The bone marrow contains multipotent adult stem cells that can differentiate into various kinds of cell lineages, such as hematopoietic cells, fibroblasts, osteoblasts, myogenic cells, and endothelial lineage. It has been reported that bone marrow mononuclear cells have the capacity to regenerate cardiac myocytes and blood vessels. As a result, many clinical and experimental trials have been conducted and have demonstrated the beneficial effects of bone marrow mononuclear cells on infarcted myocardium in human. Strauer et al.13) reported that selective intracoronary transplantation of autologous bone marrow mononuclear cells improved cardiac function in humans, possibly by bone marrow cell-associated myocardial regeneration and neovascularization. Recently, the beneficial effect of intracoronary autologous bone marrow cell infusions on left ventricular systolic function in patients after acute myocardial infarction, was demonstrated.16) In an animal study, bone marrow mononuclear cells were implanted directly into the infarcted myocardium and then were proved to enhance the collateral perfusion and regional function of the myocardium in pigs.8) However, others reported that the transdifferentiation of bone marrow cells into cardiomyocytes was quite rare.6)11) Therefore, angiogenesis rather than myocardial regeneration was used to explain the beneficial effect of bone marrow mononuclear cell treatment.
All of these findings suggest that the functions of bone marrow mononuclear cells and G-CSF in myocardial infarction are more complex than were originally anticipated. Although certain mechanisms of actions of bone marrow mononuclear cells and G-CSF on the injured myocardium appear similar, basically, the two treatment modalities were expected to have different mechanisms of action. Bone marrow mononuclear cells provide material for the regeneration of damaged myocardium and induction of angiogenesis. G-CSF proliferates this material-bone marrow mononuclear cells- and enhances regeneration or neovascularization by bone marrow mononuclear cells in the ischemic tissue. Based on this, we expected a possible synergistic beneficial effect from the combined G-CSF and intracoronary BM-MNC treatment in reperfused MI in pigs.
In this study, the numbers of white blood cells and monocytes, both in the peripheral blood and bone marrow, increased significantly in the G-CSF treated groups (Group 2 and Group 3) compared with those of the control group (Group 1). These findings demonstrated that white blood cells and monocytes were proliferated and mobilized by G-CSF.
TTC staining revealed that the infarct size at 35 days after MI was smaller in the BM-MNC with G-CSF treated group than in the other groups in this study. In concordance with this finding, the left ventricular regional wall motion which was assessed by echocardiography, improved more in BM-MNC with G-CSF treated group. Moreover, the ventricular remodeling evaluated by the increase of LV area 35 days after MI tended to be lower in BM-MNC with G-CSF treated group. One of the mechanisms of these improvements could partly be explained by the neovascularization, since the number of vWF positive cells and the expressions of endothelial surface molecules such as vWF and VE-Cadherin, increased in Group 2 and Group 3. However, the addictive beneficial effect of BM-MNCs on morphological and functional improvements of the left ventricle cannot be explained by neovascularization only.
Apoptotic cell death was considered to be responsible for the progression of left ventricular remodeling and the expansion of infarction size.32) Iwagana et al.22) suggested that G-CSF decreased the number of apoptotic endothelial cells and improve cell survival in porcine myocardial infarction model. However, in this study, we couldn't demonstrate differences in the number of apoptotic cells and cell survivals among the three groups. Although more apoptotic cells were seen over the infarct-related myocardium than over the normal myocardium in all the three groups, there were no differences among the three groups in each area (Fig. 6). Moreover, bcl2/bax ratio achieved by RT-PCR assessment of m-RNA, which are an indicator of cell survival, showed no difference among the three groups in each area (Fig. 5B). Since the number of TUNEL positive apoptotic cell itself was quite few, we could not clarify the types of apoptotic cells. Possibly, the optimal reperfusion of an infarct-related coronary artery might reduce the total number of apoptotic cell deaths in all the groups. Our results suggest that both G-CSF and BM-MNC treatment do not seem to have a significant influence on the cell survivals in the optimally reperfused MI.
One of the limitations of this study was that there were no commercially available surface antibodies which could clarify the characteristics of the collected BM-MNCs. Thus, it was not possible to characterize BM-MNCs which were infused via intracoronary arteries. Various approaches need to be considered for analyzing BM-MNCs of pigs, including the detection of specific genes.
The other limitation of this study was relatively small number of the experimental animals. In addition, two pigs of Group 1 and three pigs of Group 2 were excluded in the final analysis because of inadequate tissue preparations or inadequate echocardiographic assessments. Finally, in order to clarify the combined effect of G-CSF and BM-MNCs more precisely, an intracoronary BM-MNC infusion group without G-CSF treatment would need to be enrolled.
Since there was no evidence that the BM-MNCs transplanted after G-CSF treatment transdifferentiated into endothelial cells or myocytes in this study, it still remains to be determined whether the beneficial effects of BM-MNC with G-CSF treatment are due to transdifferentiation of BM-MNC or due to other paracrine effects.
In conclusion, our study suggested that the intracoronary bone marrow mononuclear cell infusion with G-CSF treatment reduced infarct size, improved left ventricular function and prevented ventricular remodeling in reperfused porcine MI model. These prominent combined beneficial effects that resulted in morphological and functional improvements of LV, could partly be explained by the neovascularization. However, neither BM-MNCs nor G-CSF seem to have a significant effect on apoptotic cells and bcl2/bax ratio in the optimally reperfused MI. Autologous intracoronary bone marrow mononuclear cell transplantation with G-CSF for the treatment of myocardial infarction is promising, but still challenging. Further experimental trials with larger numbers of animals are needed to demonstrate the precise role of BM-MNCs with G-CSF in myocardial infarction.
Figures and Tables
Fig. 1
A: Study protocol. B: Analysis of regional wall motion. The anterior wall and interventricular septum at the mid-papillary level were divided into 5 segments for evaluation of regional wall motion. SC: subcutaneous injection, IC: intracoronary injection, PB: peripheral blood examination, BM: bone marrow blood examination, 2D: 2-dimensional echocardiography, LV: left ventricle, RV: right ventricle, AL: anterolateral papillary muscle, PM: posteromedial papillary muscle.
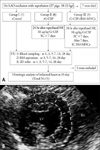
Fig. 2
Analysis of blood cells. A and C: The numbers of circulating white blood cells (WBC) and monocytes in the peripheral blood (PB) were counted preoperatively and at 1, 3, 5-7, 14, and 28 days after myocardial infarction (MI). B and D: The numbers of circulating white blood cells (WBC) and monocytes in the bone marrow (BM) were counted at pre-operatively and at 1, 3, 5-7, 14, and 28 days after myocardial infarction. *Control vs. G-CSF, p<0.05.

Fig. 3
A: Trophenltetrazolium chloride (TTC) staining. The areas of infarcted (pale) and viable (red) myocardium were measured by computed planimetry. B: The infarct size measured from tracing the myocardial slices was calculated as a percentage of the LV area. The infarct size was much smaller in the G-CSF+BM-MNCs treated group than in the control and G-CSF alone groups. The results are given as the mean±SD. *p<0.05.
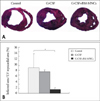
Fig. 4
Analysis of blood vessel endothelial cells by anti-von Willebrand factor (vWF) antibody in porcine heart sections. A: Representative vWF staining (a: control group, b: G-CSF group, c: G-CSF+BM-MNCs in remote region, and d: control group, e: G-CSF group, f: G-CSF+BM-MNCs in border region, g: control group, h: G-CSF group, i: G-CSF+BM-MNCs in MI region). Large vessels (arrow) and capillaries (arrowheads) were stained. Scale bar=50 µm B: The average number of endothelial cells was determined by counting randomly chosen 15 fields from remote, border, and MI regions in each group. Results are given as means±SE. *p<0.05.
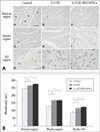
Fig. 5
Real-time PCR analysis. 35 days after MI, the levels of vWF, VE-cadherin, bcl-2 and bax mRNA expression in remote, border, and MI region of hearts were measured using real-time PCR analysis. A: Relative expressions of endothelial cell markers (vWF and VE-cadherin) analyzed through real-time PCR were normalized to that of GAPDH for each sample. B: Bcl-2 and bax gene expression was examined by quantitative real-time RT-PCR and the ratios of bcl-2 mRNA/bax mRNA. The expression levels of each genes were normalized to the house keeping gene GAPDH. Error bars indicate the SE. Results are representative of five independent experiments (n=5). *p<0.05.

Fig. 6
Detection of apoptotic cell death by the terminal deoxynucleotidyl transferase-mediated dUTP-nick end labeling (TUNEL) method in porcine heart sections. A: Representative TUNEL-positive cells (a: control group, b: G-CSF group, c: G-CSF+BM-MNCs in remote region). Arrowheads indicate TUNEL-positive cells. Scale bar=20 µm. B: The average number of TUNEL-positive cells was determined by counting randomly chosen 30 fields from normal, border, and MI regions in each group. Results are given as means±SE
Acknowledgements
The Present study was supported by grants from the Korean Society of Echocardiography (06-04).
References
1. Pfeffer JM, Pfeffer MA, Fletcher PJ, Braunwald E. Progressive ventricular remodeling in rat with myocardial infarction. Am J Physiol. 1991. 260:H1406–H1414.

2. Pfeffer MA, Braunwald E. Ventricular remodeling after myocar-dial infarction. Experimental observations and clinical implications. Circulation. 1990. 81:1161–1172.

3. Braunwald E, Kim CB. Late establishment of patency of the infarct-related artery. 1994. London: Saunders.
4. Schmermund A, Lerman LO, Ritman EL, Rumberger JA. Cardiac production of angiotensin II and its pharmacologic inhibition: effects on the coronary circulation. Mayo Clin Proc. 1999. 74:503–513.

5. Adachi Y, Imagawa J, Suzuki Y, Yogo K, Fukazawa M, Kuromaru O, Saito Y. G-CSF treatment increases side population cell infiltration after myocardial infarction in mice. J Mol Cell Cardiol. 2004. 36:707–710.

6. Balsam LB, Wagers AJ, Christensen JL, Kofidis T, Weissman IL, Robbins RC. Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature. 2004. 428:668–673.

7. Harada M, Qin Y, Takano H, Minamino T, Zou Y, Toko H, Ohtsuka M, Matsuura K, Sano M, Nishi J, Iwanaga K, Akazawa H, Kunieda T, Zhu W, Hasegawa H, Kunisada K, Nagai T, Nakaya H, Yamauchi-Takihara K, Komuro I. G-CSF prevents cardiac remodeling after myocardial infarction by activating the Jak-Stat pathway in cardiomyocytes. Nat Med. 2005. 11:305–311.

8. Kamihata H, Matsubara H, Nishiue T, Fujiyama S, Tsutsumi Y, Ozono R, Masaki H, Mori Y, Iba O, Tateishi E, Kosaki A, Shintani S, Murohara T, Imaizumi T, Iwasaka T. Implantation of bone marrow mononuclear cells into ischemic myocardium enhances collateral perfusion and regional function via side supply of angioblasts, angiogenic ligands, and cytokines. Circulation. 2001. 104:1046–1052.

9. Kang HJ, Kim HS, Zhang SY, Park KW, Cho HJ, Koo BK, Kim YJ, Soo Lee D, Sohn DW, Han KS, Oh BH, Lee MM, Park YB. Effects of intracoronary infusion of peripheral blood stem-cells mobilised with granulocyte-colony stimulating factor on left ventricular systolic function and restenosis after coronary stenting in myocardial infarction: the MAGIC cell randomised clinical trial. Lancet. 2004. 363:751–756.

10. Kuethe F, Richartz BM, Kasper C, Sayer HG, Hoeffken K, Werner GS, Figulla HR. Autologous intracoronary mononuclear bone marrow cell transplantation in chronic ischemic cardiomyopathy in humans. Int J Cardiol. 2005. 100:485–491.

11. Murry CE, Soonpaa MH, Reinecke H, Nakajima H, Nakajima HO, Rubart M, Pasumarthi KB, Virag JI, Bartelmez SH, Poppa V, Bradford G, Dowell JD, Williams DA, Field LJ. Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. Nature. 2004. 428:664–668.

12. Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson SM, Li B, Pickel J, McKay R, Nadal-Ginard B, Bodine DM, Leri A, Anversa P. Bone marrow cells regenerate infarcted myocardium. Nature. 2001. 410:701–705.

13. Strauer BE, Brehm M, Zeus T, Kostering M, Hernandez A, Sorg RV, Kogler G, Wernet P. Repair of infarcted myocardium by autologous intracoronary mononuclear bone marrow cell transplantation in humans. Circulation. 2002. 106:1913–1918.

14. Tomita S, Mickle DA, Weisel RD, Jia ZQ, Tumiati LC, Allidina Y, Liu P, Li RK. Improved heart function with myogenesis and angiogenesis after autologous porcine bone marrow stromal cell transplantation. J Thorac Cardiovasc Surg. 2002. 123:1132–1140.

15. Wojakowski W, Tendera M, Zebzda A, Michalowska A, Majka M, Kucia M, Maslankiewicz K, Wyderka R, Krol M, Ochala A, Kozakiewicz K, Ratajczak MZ. Mobilization of CD34(+), CD117(+), CXCR4(+), c-met(+) stem cells is correlated with left ventricular ejection fraction and plasma NT-proBNP levels in patients with acute myocardial infarction. Eur Heart J. 2006. 27:283–289.

16. Wollert KC, Meyer GP, Lotz J, Ringes-Lichtenberg S, Lippolt P, Breidenbach C, Fichtner S, Korte T, Hornig B, Messinger D, Arseniev L, Hertenstein B, Ganser A, Drexler H. Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial. Lancet. 2004. 364:141–148.

17. Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J, Homma S, Edwards NM, Itescu S. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med. 2001. 7:430–436.

18. Kuethe F, Figulla HR, Herzau M, Voth M, Fritzenwanger M, Opfermann T, Pachmann K, Krack A, Sayer HG, Gottschild D, Werner GS. Treatment with granulocyte colony-stimulating factor for mobilization of bone marrow cells in patients with acute myocardial infarction. Am Heart J. 2005. 150:115.

19. Link DC. Mechanisms of granulocyte colony-stimulating factor-induced hematopoietic progenitor-cell mobilization. Semin Hematol. 2000. 37:25–32.

20. Ohtsuka M, Takano H, Zou Y, Toko H, Akazawa H, Qin Y, Suzuki M, Hasegawa H, Nakaya H, Komuro I. Cytokine therapy prevents left ventricular remodeling and dysfunction after myocardial infarction through neovascularization. Faseb J. 2004. 18:851–853.

21. Orlic D, Kajstura J, Chimenti S, Limana F, Jakoniuk I, Quaini F, Nadal-Ginard B, Bodine DM, Leri A, Anversa P. Mobilized bone marrow cells repair the infarcted heart, improving function and survival. Proc Natl Acad Sci USA. 2001. 98:10344–10349.

22. Iwanaga K, Takano H, Ohtsuka M, Hasegawa H, Zou Y, Qin Y, Odaka K, Hiroshima K, Tadokoro H, Komuro I. Effects of G-CSF on cardiac remodeling after acute myocardial infarction in swine. Biochem Biophys Res Commun. 2004. 325:1353–1359.

23. Makino S, Fukuda K, Miyoshi S, Konishi F, Kodama H, Pan J, Sano M, Takahashi T, Hori S, Abe H, Hata J, Umezawa A, Ogawa S. Cardiomyocytes can be generated from marrow stromal cells in vitro. J Clin Invest. 1999. 103:697–705.

24. Tomita S, Li RK, Weisel RD, Mickle DA, Kim EJ, Sakai T, Jia ZQ. Autologous transplantation of bone marrow cells improves damaged heart function. Circulation. 1999. 100:II247–II256.

25. Bayne K. Revised Guide for the Care and Use of Laboratory Animals available. American Physiological Society. Physiologist. 1996. 39:199208–211.
26. Avalos BR. Molecular analysis of the granulocyte colony-stimulating factor receptor. Blood. 1996. 88:761–777.

27. Berliner N, Hsing A, Graubert T, Sigurdsson F, Zain M, Bruno E, Hoffman R. Granulocyte colony-stimulating factor induction of normal human bone marrow progenitors results in neutrophil-specific gene expression. Blood. 1995. 85:799–803.

28. Demetri GD, Griffin JD. Granulocyte colony-stimulating factor and its receptor. Blood. 1991. 78:2791–2808.

29. Fujii K, Ishimaru F, Kozuka T, Matsuo K, Nakase K, Kataoka I, Tabayashi T, Shinagawa K, Ikeda K, Harada M, Tanimoto M. Elevation of serum hepatocyte growth factor during granulocyte colony-stimulating factor-induced peripheral blood stem cell mobilization. Br J Haematol. 2004. 124:190–194.

30. Ohmi C, Matsuyama H, Tei Y, Yoshihiro S, Shimabukuro T, Ohmoto Y, Naito K. Granulocyte colony-stimulating factor may promote proliferation of human bladder cancer cells mediated by basic fibroblast growth factor. Scand J Urol Nephrol. 2003. 37:286–291.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download