Abstract
Echocardiography can play a key role in the diagnosis, evaluation and triage of patients presenting with acute chest pain in the emergency department, because of its rapid, accurate and repetitive image acquisition. Echocardiography can detect coronary artery disease, complications of acute myocardial infarction, pericardial emergency, acute aortic diseases, and pulmonary embolic events, all of which may cause acute chest pain. Depending on the clinical situation, stress echocardiography or contrast echocardiography may provide additional information. Ongoing technical development of imaging acquisition and analysis in echocardiography will increase its use in the differential diagnosis of acute chest pain.
Many patients visit the emergency department (ED) with a chief complaint of chest pain. These patients require rapid and efficient triage to decide whether they require hospitalization or can be discharged. It is important to allocate resources appropriately to the highest-risk patients who require timely life-saving therapy, and clinicians must be able to identify the cause of chest pain, especially in life-threatening conditions, such as acute ischemic heart disease, cardiac tamponade, acute aortic dissection and pulmonary embolism (PE). Time is essential for the patients with chest pain. Early diagnosis and treatment also reduce morbidity and mortality in the critically ill patients with chest pain.
Generally, electrocardiography (ECG) and cardiac biomarkers (e.g. CK-MB, troponin) can be used for the differential diagnosis of chest pain. However, electrocardiographic changes are often nonspecific in more than 50% of patients with chest pain. Cardiac biomarkers take hours from symptoms onset to exceed the normal range and can be influenced by renal functions.1) Because of these limitations, echocardiography can be useful in assessing patients with chest pain. This modality is ideal in the ED because it is rapid, accurate, and noninvasive and allows repeated examinations. Echocardiography can identify structural and functional changes associated with chest pain. Therefore, echocardiography plays an important role in the evaluation of different causes of chest pain, including coronary artery disease (CAD), pericardial disease, acute aortic disease, cardiomyopathy, and even pulmonary embolic events (Table 1).2) Transthoracic echocardiography (TTE) provides additional imaging capabilities, including Doppler and contrast echocardiography. The ACC/AHA committee provided guidelines for the clinical application of echocardiography in 1997, including indications in patients with acute chest pain (Table 2).3)
Recently, smaller, more portable, and relatively inexpensive hand-held echocardiographic devices are becoming more widely available and may allow physicians greater accessibility to cardiac ultrasound scanning.4) The hand-held echocardiographic examination can be used as an adjunct to information already obtained via the ECG and biomarkers for triage of patients with a low likelihood of cardiac chest pain. Thus, in a patient with normal ECG results, and CK-MB and troponin levels at the time of presentation, the additional absence of left ventricular wall motion abnormalities upon hand-held echocardiographic examination would indicate a sufficiently low risk for acute myocardial ischemia or infarction and may support the decision to discharge the patients directly from the ED.
Regional wall motion abnormalities (RWMA) occur before the ECG changes or the onset of symptoms in the ischemic cascades. Left ventricular (LV) diastolic dysfunction occurs before systolic dysfunction; ECG abnormalities and chest pain are relatively late events (Fig. 1).5) RWMA detected by echocardiography correspond with the coronary artery territories, although there is some variation depending on the individual coronary artery anatomy6) and dominance of the right coronary artery (RCA) and left circumflex arteries (LCX).
The severity of the wall motion abnormality depends on the transmural extent of the infarction, and the circumferential limits depend on the coronary arterial distribution. The ischemic myocardium continues to demonstrate some degree of passive forward motion because of the pulling action of adjacent nonischemic myocardium, but the contractility (systolic thickening) of the ischemic myocardial segments decreases or is absent. In a normal person, the LV free wall thickness increases by more than 40% during systole. Hypokinesis is defined as systolic wall thickening of less than 30%, and akinesis is defined as systolic wall thickening of less than 10%. Dyskinesis means that a myocardial segment moves outward during systole, usually in association with systolic wall thinning.7)
To detect RWMA, the LV is divided into several segments. American Society of Echocardiography scoring system is used most widely for grading the severity of a wall motion abnormality (Table 3).8) A 17-segment model has been proposed recently, which adds the apical cap.9) This 17-segment model (Fig. 2) is preferred for myocardial perfusion studies, which can be compared with other perfusion modalities {cardiac single photon emission computed tomography (SPECT), cardiac magnetic resonance image (MRI), cardiac computed tomography (CT) etc.}. Each segment is assigned a score on the basis of visual assessment of contractility: normal: 1, hypokinesis: 2, akinesis: 3, dyskinesis: 4, and aneurysmal: 5. The wall motion score index (WMSI) is calculated to provide a semi-quantitative measure of the extent of RWMA. For example, a normal contracting LV has a WMSI of 1 (16/16), whereas a dysfunctional LV will have an increasing WMSI proportional to the severity of RWMA. This score correlates with the myocardial infarction size compared with the perfusion defect size on cardiac SPECT imaging. Patients with a WMSI greater than 1.7 have a perfusion defect larger than 20%.10)
The coronary arterial territories of the LV wall are depicted in Fig. 3.9) The anterior, anterolateral, anteroseptal, and apical segments are in the distribution of the left anterior descending artery (LAD). The lateral wall and lateral apex correspond to the LCX distribution. The inferolateral wall is supplied by the posterior descending artery. In 80% of the population, the posterior descending artery arises from the RCA. In the other 20%, the posterior descending artery arises from the LCX (so called, left-dominant). The right ventricle (RV) is supplied by acute marginal branches arising from the RCA. The coronary artery anatomy may differ between individual patients, so the extent of segmental wall motion abnormalities may also differ between patients. In addition, the presence of collateral vasculatures and bypass graft must be considered.
Echocardiography can be useful for direct visualization of coronary arteries.11)12) Detection of the ostia of the left and right coronary arteries and proximal part of the LAD and their flow is helpful for excluding an anomalous origin of a coronary artery, coronary aneurysm, and coronary fistula, associated with chest pain or sudden death. Use of a high-frequency transducer and a special setting of low Nyquist limits allow visualization of the coronary artery tree and evaluation of the coronary flow.12) For instance, coronary flow reserve (CFR), expressed as the ratio of the peak diastolic flow velocity during maximal vasodilatation to that of the basal diastolic flow velocity, can predict coronary artery stenosis and can be used to elucidate other causes of chest pain, including myocardial hypertrophy, microvascular disease, and aortic stenosis.13-15) TTE allows the evaluation of the diastolic coronary flow velocity in the distal LAD. An extremely slow diastolic velocity of less than 13 cm/s may suggest the pathologic flow, which can mean impairment of myocardial perfusion.12)16) The slow diastolic velocity can explain the "slow dye progression" phenomenon of coronary angiography in patients with angina-like chest pain. Retrograde flow at the anterior apex of the heart reflects the presence of collateral flow, which means the total occlusion of the LAD.12)17)
In most cases, a patient's cardiovascular symptoms are worsened by activity or stress. An adequate supply of oxygen to the myocardium is essential for preventing the development of ischemia and infarction. For example, coronary artery stenosis limits an appropriate increase in perfusion in response to an increased demand for blood flow, as occurs during exertion or emotional stress. Stress echocardiography compares the wall motion, myocardial perfusion, pressure gradient, pulmonary pressure, valvular regurgitation, or filling pressure before and after a stress to identify a pathologic condition, that is not apparent at rest. During a stress echocardiography, two-dimensional echocardiographic images at rest and during stress are digitized and displayed in a side-by-side format so that induced RWMAs can be detected easily.
The most common indication for stress echocardiography is the evaluation of myocardial ischemia, but this modality is also available in the evaluation of valvular heart disease, pulmonary hypertension, LV filling pressure (e.g. diastolic heart failure), and exertional dyspnea.
Echocardiography related to exercise or pharmacologic stress to assess the presence of ischemia is safe and effective in patients with acute chest pain, except myocardial infarction (MI) or unstable angina.
Stress echocardiography is performed during exercise or administration of pharmacologic agents. Exercise stress includes a treadmill exercise protocol (e.g. Bruce protocol) or a supine bicycle protocol. For a treadmill study, the patient is imaged at baseline and just after peak exercise. Because exercise-induced RWMA due to ischemia usually lasts for a few minutes after termination of exercise, it is important to acquire the immediate postexercise image to compare with the baseline preexercise image. The supine bicycle protocol can be used to obtain images throughout exercise, which allows for image comparison at low, medium, and high workloads.18) Exercise echocardiography should be terminated if any of the following conditions occurs: (1) severe symptoms (chest pain, dyspnea), (2) severe ischemia (ST-segment depression ≥5 mm or ST-segment elevation), (3) complex ectopy or ventricular tachycardia, (4) hypertensive blood pressure response (>220 mmHg systolic, >110 mmHg diastolic), (5) hypotension (>20 mmHg decrease compared with previous stage), or (6) reaching the target heart rate.19) In one study, with using the wall motion criteria, the sensitivity and specificity of exercise stress echocardiography were 85% and 88%, respectively, compared with 85% and 81% for an exercise thallium scan.20)
When a patient cannot exercise properly, a pharmacologic stress test can be performed to induce ischemia with dobutamine, adenosine or dipyridamole. Dobutamine is used most commonly in stress echocardiography (Fig. 4). Baseline images are obtained, followed by the infusion of dobutamine at incremental levels (usually 5, 10, 20, 30 and 40 ㎍/kg/min) of 3 minutes duration. Patients who do not reach target heart rate even after being given 40 ㎍/kg/min of dobutamine can perform handgrip maneuvers or be given intravenous atropine in doses of 0.1 to 0.25 mg up to total of 1 mg, unless they have contraindications. The test is terminated when the patient's heart rate reaches 85% of maximum predicted heart rate or the evidence of myocardial ischemia, hypotension, significant arrhythmia or drug-related side effects is developd.21) In one study, dobutamine stress echocardiography was found to have a sensitivity of 89.5%, a specificity of 88.9%, and a negative predictive value of 98.5%.22)
Tissue Doppler imaging (TDI), strain rate imaging, or color kinesis are anticipated to improve the accuracy of stress echocardiography.
The LV opacification with imaging microbubbles improves the definition of the LV endocardial border (Fig. 5). This provides better quantification of LV volume and assessment of wall motion analysis. The administration of an echocardiographic contrast enhancing endocardial border has increased the sensitivity of stress echocardiography in the diagnosis of CAD. In one study, the percentage of wall segments visualized increased from 94.4% to 99.8% with the use of LV opacification during peak exercise.23)
Myocardial contrast echocardiography (MCE) allows the assessment of myocardial perfusion with microbubbles, delivered to small myocardial vessels for ventricular enhancement. Hypoperfused areas of myocardium lack the enhancement by a contrast agent. MCE is a reproducible and reliable technique for evaluating of the risk area after coronary occlusion, regional coronary flow reserve, myocardial viability, and the outcome of reperfusion.24)
Innovative ultrasound methods using harmonic imaging have been developed to exploit the interaction between microbubbles and ultrasound and to allow assessment of perfusion.
Harmonic imaging has recently been shown to provide improved endocardial definition relative to tissue harmonic imaging for specific regions of the endocardium, in particular the base and anterior wall.25) In conventional echocardiography, images are derived from ultrasound waves returned at the same frequency as the transmitted waves. However, harmonic frequencies are also produced by the transmitted wave; in harmonic imaging, the machine uses the returning second harmonic to construct images. This improves image quality because there is less distortion of the returned ultrasound waves (they are generated in the heart and only have to pass through tissue once), and few harmonics are produced close to the chest wall where many artifacts arise.26) This technique has recently been shown to be superior to routine evaluation to detect of CAD.27)28) If the amount of microbubbles in the myocardium is sufficient, they can be destroyed with a high mechanical index (>1.5) of ultrasound. In the normal myocardium, the microbubbles are replenished within five to seven cardiac cycles. However, in the myocardium with low or no perfusion, the microbubbles are not replenished normally, and the areas of myocardium affected appear dark or patchy.29)30)
In the hemodynamically unstable patients after acute MI, it is very important to identify immediately the underlying cause and to exclude cardiac rupture to ensure proper treatment. Two-dimensional echocardiography and color Doppler study are useful for identifying the cause in these patients, especially for checking mechanical complications (e.g. ventricular septal rupture, papillary muscle rupture, free wall rupture). Therefore, echocardiography is the mainstay of diagnosis of mechanical complications of MI,31) and patients with unexplained hemodynamic deterioration should be evaluated immediately (Table 4). Thrombolytic therapy and primary percutaneous coronary intervention have been reported to reduce the incidence of mechanical complications.
Ventricular septal rupture occurs in 1-3% of patients after MI, and it occurs during the early phase of acute infarction (almost within the first week). The typical clinical presentation is a new systolic murmur, with abrupt hemodynamic deterioration. The risk factors for development of ventricular septal rupture include old age (>65 years), hypertension, anterior wall infarction, first infarction, or lack of collateral circulations.32) Two-dimensional echocardiography may show a discrete defect, but there may also be multiple serpiginous channels in the necrotic myocardium (Fig. 6). The diagnosis can usually be made by TTE, and experience is essential because the most useful views depend on the location of defect. Subcostal views are particularly useful in the critically ill, supine patient with inferior infarction. Small defects may not be visible, but color Doppler is very sensitive. When TTE is suboptimal for examination, transesophageal echocardiography (TEE) is very useful for detecting the site of the defect, and the presence of multiple defects.
The therapeutic strategy for ventricular septal rupture is early surgery. Without a repair, the outcome is very poor. Early surgical intervention improves the outcome compared with conservative therapy.33) Intra-aortic counterpulsation can be used to stabilize the patient with cardiogenic shock before surgery.
The necrosis and rupture of a papillary muscle causes acute mitral regurgitation (MR) in patients with acute MI. The clinical presentation of papillary muscle rupture is variable. About 50% of patients with acute MR do not have a systolic murmur because of rapid equalization of the LV and left atrial (LA) pressure. In addition, the murmur can be attenuated by a crackled lung sound caused by pulmonary edema. Rupture usually involves the posteromedial papillary muscle, perfused from the posterior descending artery alone, whereas the anterolateral papillary muscle has a dual blood supply from both the diagonal and circumflex arteries.
Two-dimensional echocardiography can detect abnormalities of the mitral valve apparatus, including prolapse of one or both leaflets, a flail leaflet, and liberation of a portion of the papillary muscle (Fig. 7). A papillary muscle rupture should be suspected when there is eccentric jet of MR with a relatively normal-sized LA. TEE is a particularly suitable imaging modality34) and should be performed immediately if this diagnosis is suspected because it provides high-resolution images of the papillary muscles and accurate assessment of the severity of MR.
Papillary muscle rupture and acute severe MR is a life-threatening condition that requires urgent surgical intervention. Once a papillary muscle rupture has been diagnosed, immediate mitral valve replacement and coronary revascularization is necessary for patient's survival.
Rupture of the free wall of the LV is a fatal mechanical complication of acute MI that occurs in about 1% of patients with MI. Free wall rupture is prone to the patients with a transmural MI involving the inferolateral wall in association with the LCX. The administration of thrombolytic agents may promote cardiac rupture, presumably because of a hemorrhagic tendency. Free wall rupture usually presents clinically as hemodynamic collapse and sudden death.
Direct visualization of the free wall rupture is often difficult because it may be only a "slit" in the myocardium and the location of pericardial fluid may not correlate with the area of rupture (Fig. 8). It is rare for a color Doppler study to show blood flow into the pericardium. When the diagnosis is uncertain, the use of left heart contrast may delineate the tear. An intrapericardial thrombus is often present and is very characteristic. It appears as an echo-dense mass that may be mobile and undulating within the pericardial space or immobile and impinging on cardiac chambers.35) Negative echocardiographic findings should not exclude myocardial rupture, if the clinical suspicion is high.
The mortality is so high unless appropriate treatment is given, that clinical suspicion, early detection and prompt surgical intervention are indispensable for good clinical outcome.
A pseudoaneurysm is characterized by a small neck communication that connects the LV and aneurismal cavity. The majority of pseudoaneurysms are located in inferoposterior or inferolateral regions, in association with the RCA or LCX.
It is important to distinguish a pseudoaneurysm, which has a high possibility of spontaneous rupture, from a true aneurysm, which seldom ruptures spontaneously. In a true aneurysm, there is continuity of the myocardium in the region of dilatation, in contrast to the loss of this continuity in a pseudoaneurysm with an abrupt area of rupture. An echocardiographic feature of pseudoaneurysm is the narrow neck. The ratio of the diameter of entry and the maximal diameter of the pseudoaneurysm is usually <0.5, although this feature is not always reliable and has only a 60% sensitivity.36) Spectral and color Doppler studies demonstrate characteristic "to-and-pro" flow through the rupture site. Contrast echocardiography can be helpful in locating the point of communication. Fig. 9 demonstrates the difference between a true aneurysm and a pseudoaneurysm.
The RV is involved in acute MI frequently. RV infarction occurs in up to 50% of inferior wall MI, but hemodynamically significant RV infarction occurs infrequently.37) Many RCA occlusions do not result in significant RV infarction because of the lower RV oxygen demand, higher oxygen extraction ratio, greater systolic/diastolic flow ratio, and collateral supply. However, once a patient develops cardiogenic shock due to RV infarction, the mortality is so high that initial recognition and treatment are very important. The clinical settings of a suspected RV infarction are unexplained hypotension or low cardiac output after acute MI, marked hypotension after administration of nitrates, and unexplained hypoxemia after acute MI. Patients who have an RV infarction present with increased jugular venous pressure but clear lung fields. Echocardiography plays an important role in the diagnosis and assessment of RV infarction. The RV is dilated and hypokinetic to akinetic (Fig. 10). Tricuspid regurgitation may appear and become significant because of dilatation of the tricuspid annulus. TDI may provide additional evidences of RV infarction. In one study, a tricuspid valve annulus peak velocity less than 12 cm/s had a sensitivity of 81%, specificity of 82%, and negative predictive value of 92% for RV infarction.38)
When a segmental wall motion abnormality is present in the LV cavity, it is prone to form a thrombus, most commonly in the LV apex resulting from occlusion of the LAD. The peak timing of thrombus formation after MI is 3 days, but thrombus may occur as early as within few hours in large areas of apical akinesis and may occur a few weeks after acute MI.
Because of concern about a potential embolic event, patients with a large apical infarction or ventricular aneurysm should be given anticoagulation properly.
Two-dimensional echocardiography is the most practical and reliable imaging modality for detecting a LV thrombus. The echocardiographic finding of a thrombus is a mass that is distinct from the endocardium and protruding to a variable extent into the LV cavity (Fig. 11). Characteristically, a thrombus has a nonhomogeneous echo density. The base of attachment to the wall may be broad in a sessile thrombus, or narrow in a pedunculated thrombus, which has a greater chance of embolization. Contrast echocardiography is also helpful in detecting a LV thrombus.
Hemodynamically insignificant pericardial effusion is common after MI, especially after a large transmural infarction. Pericardial friction rubs and/or pericardial pain are frequently encountered in patients with a transmural MI. It is important to identify the chest pain of pericarditis accurately, because failure to be aware of it may lead to the incorrect diagnosis of recurrent ischemic pain and/or infarct extension, with resulting inappropriate use of anticoagulants, nitrates, beta-blockers, or coronary angiography.
Echocardiography is a sensitive technique for diagnosing pericardial effusion, although the absence of fluid does not exclude pericarditis. Echocardiography can identify a site for percutaneous drainage if required and can be used to monitor the procedure. The effusion does not increase morbidity and mortality in MI but serves as another indicator of a large area infarction. Larger effusion or echocardiographic dense effusions representing hemorrhage should always make the clinicians consider a free wall rupture.39)
In patients with severe hemodynamic compromise, TTE may be limited by mechanical ventilation, recent cardiac surgery, or an inability to position the patient adequately. In this situation, TEE has proven to be efficacious, especially in ruling out complications related to cardiac rupture and for assessing global and regional ventricular function.40) With careful sedation and close monitoring, TEE can be performed safely even in patients with early acute MI.41) However, it does have chances of potential complications, TEE should be performed by an experienced physician. It is relatively contraindicated in patients with esophageal diseases-for example, varices, stricture, esophagitis, scleroderma-and may be hazardous in patients with severe coagulopathy or poor respiratory function.
Acute pericardial abnormalities can range from the pleuritic chest pain of pericarditis to marked hemodynamic collapse and even death from tamponade. Echocardiography is the most powerful tool for diagnosing pericardial diseases, especially in emergent clinical settings (e.g. pericardial effusion, pericardial tamponade). When a pericardial effusion or tamponade needs to be drained, pericardiocentesis can be performed most safely under the guidance of two-dimensional echocardiography.42) TEE is also helpful in measuring pericardial thickness, in evaluating diastolic function from pulmonary vein, and in detecting loculated pericardial effusion or other structural abnormalities of the pericardium.
The filling of the pericardial space with fluid or blood results in a pericardial effusion, detected as an echo-free space. A small amount of effusion may be detected usually as a posterior echo-free space in the parasternal long axis view in the ventricular systolic phase. When the amount of pericardial effusion is massive, the heart may have a "swinging" motion in the pericardial cavity. However, the swinging motion is not always present in cardiac tamponade. Cardiac tamponade can occur with a small amount of pericardial effusion if the effusion accumulates rapidly.43)
Two-dimensional echocardiographic features of a tamponade include early diastolic collapse of the RV, late diastolic right atrial inversion, abnormal ventricular septal motion, respiratory variation in ventricle chamber size, and plethora of the inferior vena cava with blunted respiratory changes.
The Doppler echocardiographic findings of pericardial effusion and tamponade are more sensitive than the two-dimensional echocardiographic features.44) Cardiac tamponade makes the four cardiac chamber pressures equal. During inspiration, the venous return and RV filling increase and that result in a leftward septal shift. Therefore, the Doppler ultrasound shows that tricuspid and pulmonic valve flow velocities increase markedly during inspiration, while pulmonic vein, mitral, and aortic flow velocities diminish.
The most effective therapy of tamponade is elimination of the pericardial fluid. If manifestations of tamponade appear, pericardial drainage must be performed immediately because relief of the intrapericardial pressure may be lifesaving. Intravenous saline may be administered as the patient is being readied. Pericardiocentesis is very useful for lifesaving in tamponade, but a blind percutaneous attempt has a high rate of complications, including pneumothorax, puncture of the cardiac wall, and even death. Two-dimensional echocardiography provides guidance of the needle by locating the optimal site of the puncture, by determining the depth of the pericardial effusion, usually from the subcostal view. The position of the puncture needle can be confirmed by imaging with administration of agitated saline.45) Two-dimensional echocardiography can be used to monitor the result of pericardiocentesis.
Because of the high mortality rate and the need for early medical and surgical treatment in acute aortic diseases, rapid and accurate diagnostic techniques are indispensable. In this aspect, echocardiography can play a key role in diagnosis acute aortic diseases. Compared with other modalities (CT, MRI), echocardiography has the advantage of being applicable in any hospital department-without the need to transfer the patient, who is often in an unstable hemodynamic situation, is being monitored, and has an intravenous line in place. In this section, we focus on aortic dissection, a potentially life-threatening condition.
Aortic dissections are classified by the location of the tear and the extent of involvement. The diagnosis and identification of the location and extent of an aortic dissection are important for optimal treatment. For instance, a proximal aortic dissection requires urgent surgical intervention, whereas a distal aortic dissection is treated medically, unless the patient has severe, ongoing pain or the patient is hemodynamically unstable.
TTE is a good modality for identifying a proximal aortic dissection. The parasternal view allows one to see the aortic root, the lower third of the ascending aorta, and part of the descending thoracic aorta behind the LA (Fig. 12). The aortic arch, the origin of the supra-aortic trunks, and the proximal third of the descending aorta, can be assessed using the suprasternal view. However, the main limitation of TTE is the difficulty in obtaining good quality images. And the diagnostic errors are frequent when the dissection is small or another type of aortic disease is present. The low negative predictive value of TTE does not permit the diagnosis of dissection to be ruled out, and further tests will be required.46)47)
Because of the proximity between the esophagus and aorta, TEE can acquire the imaging of aorta without disturbance of the chest wall or lung. Because it provides a clearer and more complete view of the entire aorta, TEE is now the most common initial diagnostic imaging modality for diagnosing aortic dissection, with CT. TEE requires greater skill and patient cooperation but is very accurate in identifying dissections of the ascending and descending thoracic aorta, achieving 98% sensitivity and approximately 90% specificity.48)
The surgical approach may differ depending on the location of the entry tear, particularly in retrograde dissections of the ascending aorta (20% of cases).49) Occasionally, two-dimensional echocardiography cannot be visualized the intimal tear in the proximal part of the arch. In these cases, color Doppler may be helpful by showing a turbulent jet flow directed towards the false lumen. Using pulsed-wave Doppler, it can be verified that the flow velocity at the tear is usually below 1.5 m/s and that the flow goes from the true to the false lumen in systole.
In certain cases, false lumen identification is important for the management. For example, when the aortic arch is involved, the surgeon needs to know whether the supraaortic vessels originate from the false lumen. The distinction between a true and false lumen is relatively easy. The false lumen is usually larger and has less blood flow than the true lumen. Partial thrombosis of the false lumen is present frequently and total thrombosis is present occasionally (Fig. 13). Doppler study may be useful for differentiating the true lumen from the false lumen. Furthermore, variants of acute aortic syndromes such as intramural hematoma and atherosclerotic penetrating ulcers can also be identified.
Another advantage of TEE is the capability of detecting the potential complications of acute aortic dissection, including hemopericardium, coronary involvement,50) and aortic insufficiency. When aortic regurgitation is present, its mechanism can be defined by TEE. This is very useful to surgeons for identifying patients who are good candidates for valve repair.
Although TEE is a very sensitive diagnostic tool for diagnosing acute aortic diseases, TEE is a semi-invasive technique. When the probe is inserted into the upper gastrointestinal tract, heart rate and blood pressure can be increased, and arterial oxygen saturation may be decreased. And these situations can make patients dangerous hemodynamically. For this reason, it is important to administer adequate sedation and monitor arterial pressure, with an intravenous line in place for antihypertensive drug administration if needed.
Acute PE is an important clinical problem with 10% mortality.51) Symptoms and signs of PE are often nonspecific or overlap with other acute cardio-pulmonary syndromes. Although there are ECG changes that are more common in PE, including the S1Q3T3 pattern, atrial tachyarrhythmias, incomplete right bundle-branch block, or negative T wave in the right and midprecordial leads, the ECG alone is not sufficiently sensitive or specific to rule out or confirm the diagnosis.52)53)
Although checking of serum D-dimer level and contrast-enhanced CT are the basic diagnostic tools for PE, echocardiography can also play a role in diagnosis and management of PE, especially in evaluating RV function. Bedside echocardiography can usually differentiate reliably among illnesses that have radically different treatment, including acute MI, pericardial tamponade, dissection of the aorta, and PE complicated by right heart failure.
RV dilatation is the most consistent echocardiographic finding in a massive PE.54) Right-to-left septal shifting may occur because of increased pressure of pulmonary artery.55) Echocardiography can play an important role in the management of the patients by identifying the position of the interventricular septum and the degree of pulmonary arterial pressure.
McConnell's sign (i.e.-RV free wall hypokinesis with normal RV apical motion) appears to be helpful for diagnosis of a massive PE.56) On occasion, thrombi-in-transit are detected in the chambers on the right side of the heart.57) These thrombi are highly mobile and appear like a popcorn or a snake. Their presence is a clear marker of high risk because these patients have typically suffered from a massive PE and more often manifest overt RV dysfunction, and greater hemodynamic compromise, and have a worse prognosis.58)
Detection of RV dysfunction due to PE helps to stratify the risk, delineate the prognosis, and plan optimal management. Echocardiographic evidence of RV hypokinesis predicts a two-fold increase in mortality in 2 weeks and 3 months.59)
Transthoracic parasternal short axis view at the base of the heart allows direct visualization of a thrombus inside the pulmonary artery (Fig. 14). In addition, the main pulmonary trunk and its bifurcation are well visualized by TEE. TEE has been shown to have comparable sensitivity (80%-97%) and specificity (86%-100%) as spiral CT for detecting central pulmonary emboli. Thus, TEE may play a role in the diagnosis of central PE, and is perhaps best applied in the setting of hemodynamical instability.
Dyspnea is one of the cardinal symptoms of the heart disease. When a patient visits the ED with a chief complaint of acute onset dyspnea, it is important to identify the cause of dyspnea, and especially to distinguish cardiac from pulmonary origin. When the etiology is in doubt, echocardiography can play a key role in elucidating the origin of dyspnea by documenting or ruling out the common cardiac causes of pulmonary congestion.60) In this regard, echocardiography can be the preferred initial diagnostic modality when the history, physical examination, and routine laboratory tests cannot identify the cause of dyspnea (Table 5).61)
Echocardiography can be useful in diagnosing other conditions, such as peripheral edema and unexplained hypotension. The ACC/AHA committee provided guidelines for clinical application of echocardiography in 1997, and these include the indications in patients with edema, dyspnea, and cardiomyopathy (Table 6).3)
Echocardiography is a valuable, noninvasive diagnostic tool that can provide information about cardiac function and valvular abnormalities, and alternative explanations for causes of chest pain and shortness of breath. In the ED, an early, bedside, echocardiographic examination is a safe and cost-effective technique for evaluating further patients with chest pain, elusive clinical findings, and nondiagnostic ECGs. Stress echocardiography can provide additional information about myocardial ischemia and hemodynamic changes. MCE can evaluate the myocardial perfusion state and assess myocardial viability after ischemic attack. In the acute coronary syndrome, two-dimensional and Doppler echocardiography can play a key role in both diagnosing acute coronary syndrome and detecting complications of MI.
Echocardiography promptly confirms the presence of and hemodynamic changes in acute pericardial emergencies, including pericardial effusion, and cardiac tamponade. Echocardiography guidance allows the clinician to perform urgent pericardiocentesis safely in the same direction as the probe was aimed during the echocardiography. TEE has great value for the rapid identification of acute aortic disease and should be used in the ED to diagnose such unstable patients correctly.
Although echocardiography is widely used in various fields, echocardiography has some limitations. Adequate training and skillful technique are required to obtain a good image. It is difficult to acquire a good image from certain patients, especially those who have chronic obstructive lung disease, morbid obesity, or chest deformity. The immediate availability of a skilled sonographer is essential and may be a problem in clinics.
Ongoing advances in imaging acquisition and analysis in echocardiography will make the echocardiography more popular for evaluating and diagnosing diseases.
Figures and Tables
Fig. 1
Ischemic cascade: In experimental models, left ventricular regional wall motion abnormalities are detected after the onset of ischemic stress, but before the appearance of ECG changes and subjective symptoms.

Fig. 2
17-Segmental analysis of LV walls. 1: basal anterior, 2: basal anteroseptal, 3: basal inferoseptal, 4: basal inferior, 5: basal inferolateral, 6: basal anterolateral, 7: mid anterior, 8: mid anteroseptal, 9: mid inferoseptal, 10: mid inferior, 11: mid inferolateral, 12: mid anterolateral, 13: apical anterior, 14: apical septal, 15: apical inferior, 16: apical lateral, 17: apex.

Fig. 3
Typical distributions of the coronary arteries. Some segments are supplied by variable coronary arteries. RCA: right coronary artery, LAD: left coronary artery, LCX: left circumflex artery.
Fig. 4
This figure illustrates the myocardial responses to a low dose and a peak dose of dobutamine. As the dobutamine dose is increased, coronary blood flow and contractility of the myocardium increase, and these improve the wall motion of myocardium. In ischemia, coronary artery stenosis prevents sufficient coronary blood flow, resulting in worsening of wall motion at a peak dose of dobutamine. If the myocardium that is akinetic at rest, is viable, myocardial contractility increases continuously with a low dose and a peak dose of dobutamine, indicating no further coronary stenosis after acute coronary artery disease. If the myocardium is viable but the coronary artery is still stenotic after acute coronary artery disease, myocardial wall motion improves initially at a low dose of dobutamine, but worsens with a peak dose. This phenomenon is called a biphasic response. When the myocardium is scarred, no improvement of myocardial wall motion occurs with an escalating dose of dobutamine.

Fig. 5
Contrast harmonic imaging from the apical four-chamber view with intravenous microbubbles as the echocardiographic contrast agent. The definition of the left ventricular (LV) endocardial border (dotted line) is more distinguishable by injection of an echocardiographic contrast agent.

Fig. 6
Imaging of ventricular septal rupture after myocardial infarction: A transthoracic apical four-chamber view with color Doppler image demonstrates a shunt from the left ventricle (*) to the right ventricle (RV) in a patient with anterior wall myocardial infarction (arrow).
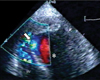
Fig. 7
Transesophageal echocardiographic images show the rupture of a papillary muscle and mitral regurgitation (MR) in a patient with acute myocardial infarction. A: In a multiplane four-chamber view (0 degrees with retroflexion of the transducer tip), a ruptured posteromedial papillary muscle (arrow), which is attached to the posterior mitral leaflet prolapses into the left atrium (LA). B: Color Doppler image demonstrates severe mitral regurgitation caused by a rupture of a papillary muscle. LV: left ventricle.
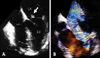
Fig. 8
Transthoracic echocardiography examination of a 72-year-old man who had an acute chest pain appeared about 24 hours ago. Initial images {A: parasternal long axis view (left). B: apical four-chamber view (left)} showed an akinesis of LV anterior wall and apex, indicating an acute anterior wall myocardial infarction. After 3 minutes, the echo-free space appeared suddenly, which represented a hemorrhagic pericardial effusion, caused by a rupture of the ventricular free wall {A: parasternal long axis view (right). B: apical four-chamber view (right)}. Arrowheads indicate the possible site of rupture. LV: left ventricle.
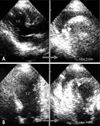
Fig. 9
A: The wall of the true aneurysm is composed of fibrous tissue and contains elements of the cardiac wall. The true aneurysm represents a wide mouth resulting from a gradual bulge of the involved portion of the wall. B: The pseudoaneurysm occurs when a rupture of the ventricular free wall is contained by overlying, adherent pericardium. Abrupt loss of muscular continuity exists at the ostium of the pseudo-aneurysm. Characteristically, the mouth of a false aneurysm is narrow compared with the width of the fundus.
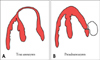
Fig. 10
Transthoracic echocardiographic images show an example of right ventricular (RV) infarction. A: Parasternal views (right: short axis, left: long axis) demonstrates a RV dilatation and RV free wall akinesis. B: Apical four-chamber view shows inferior septal akinesis and right atrial (RA) and RV dilatation. C: The inferior vena cava (IVC) is dilated because of venous congestion.
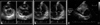
Fig. 11
Two-dimensional echocardiography in an apical four-chamber view shows a mass distinct from the endocardial border and protruding into the left ventricular (LV) cavity (*) in a patient with acute anteroapical wall myocardial infarction.
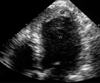
Fig. 12
Transthoracic parasternal long-axis view demonstrates an acute aortic dissection involving the proximal ascending aorta. The image shows intimal flaps (arrow) in the dilated ascending aorta, and not dilated, unaffected descending aorta (*). LV: left ventricle, LA: left atrium.
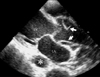
Fig. 13
Transverse transesophageal echocardiographic imaging of descending aorta shows a intimal flap (arrowhead), associated with an acute aortic dissection. An echogenic material, presumed to be a thrombus, is in the false lumen (arrows).
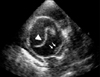
Fig. 14
Transthoracic parasternal short axis view at the base of the heart, obtained by tilting the transducer further superiorly. At this level, the great arteries are sectioned transversely. This picture shows a dilated main pulmonary artery (MPA) and a thrombus (arrow) in the bifurcation of the MPA, which extends into the right main branch. Ao: aorta, LPA: left pulmonary artery.
References
1. de Winter RJ, Koster RW, Sturk A, Sanders GT. Value of myoglobin, troponin T, and CK-Mb mass in ruling out an acute myocardial infarction in the emergency room. Circulation. 1995. 92:3401–3407.

2. Otto CM. Textbook of Clinical Echocardiography. 2004. 3rd edition. Philadelphia: Elsevier Saunders.
3. Cheitlin MD, Alpert JS, Armstrong WF, Aurigemma GP, Beller GA, Bierman FZ, Davidson TW, Davis JL, Douglas PS, Gillam LD. ACC/AHA guidelines for the clinical application of echocardiography. A report of the American college of Cardiology/American heart association task force on practice guidelines (committee on clinical application of echocardiography). Developed in collaboration with the American society of echocardiography. Circulation. 1997. 95:1686–1744.

4. Goodkin GM, Spevack DM, Tunick PA, Kronzon I. How useful is hand carried bedside echocardiography in critically ill patients? J Am Coll Cardiol. 2001. 37:2019–2022.

5. Weston P, Alexander JH, Patel MR, Maynard C, Crawford L, Wagner GS. Hand-held echocardiographic examination of patients with symptoms of acute coronary syndromes in the emergency department: The 30-day outcome associated with normal left ventricular wall motion. Am Heart J. 2004. 148:1096–1101.

6. Hauser AM, Gangadharan V, Ramos RG, Gordon S, Timmis GC. Sequence of mechanical, electrocardiographic and clinical effects of repeated coronary artery occlusion in human beings: Echocardiographic observations during coronary angioplasty. J Am Coll Cardiol. 1985. 5:193–197.

7. Choi YS, Youn HJ, Jung SE, Choi YW, Lee DH, Park CS, Oh YS, Seung KB, Kim JH, Hong SJ. The association between coronary artery calcification on MDCT and angiographic coronary artery stenosis. Korean Circ J. 2007. 37:167–172.

8. Schiller NB, Shah PM, Crawford M, De Maria A, Devereux R, Feigenbaum H, Gutgesell H, Reicheck N, Sahn D, Schnittger I, Silverman NH, Tajik AJ. Recommendations for quantitation of the left ventricle by two-dimensional echocardiography. American society of echocardiography committee on standards, subcommittee on quantitation of two-dimensional echocardiograms. J Am Soc Echocardiogr. 1989. 2:358–367.

9. Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, Picard MH, Roman MJ, Seward J, Shanewise JS, Solomon SD, Spencer KT, Sutton MS, Stewart WJ. Chamber Quantification Writing Group. American Society of Echocardiography's Guidelines and Standards Committee. European Association of Echocardiography. Recommendations for chamber quantification: A report from the American Society of Echocardiography's Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr. 2005. 18:1440–1463.

10. Oh JK, Gibbons RJ, Christian TF, Gersh BJ, Click RL, Sitthisook S, Tajik AJ, Seward JB. Correlation of regional wall motion abnormalities detected by two-dimensional echocardiography with perfusion defect determined by technetium 99m sestamibi imaging in patients treated with reperfusion therapy during acute myocardial infarction. Am Heart J. 1996. 131:32–37.

11. Youn HJ. Direct visualization of coronary artery and flow using transthoracic Doppler echocardiography. J Korean Pediatr Cardiol Soc. 2006. 10:354–366.
12. Youn HJ. Demonstration of pathologic coronary flow dynamics using transthoracic Doppler echocardiography: Its potential role in clinical decision-making. Korean Circ J. 2005. 35:269–281.

13. Opherk D, Mall G, Zebe H, Schwarz F, Weihe E, Manthey J, Kübler W. Reduction of coronary reserve: A mechanism for angina pectoris in patients with arterial hypertension and normal coronary arteries. Circulation. 1984. 69:1–7.

14. Cannon RO 3rd, Watson RM, Rosing DR, Epstein SE. Angina caused by reduced vasodilator reserve of the small coronary arteries. J Am Coll Cardiol. 1983. 1:1359–1373.

15. Marcus ML, Doty DB, Hiratzka LF, Wright CB, Eastham CL. Decreased coronary flow reserve: A mechanism for angina pectoris in patients with aortic stenosis and normal coronary arteries. N Engl J Med. 1982. 307:1362–1366.
16. Youn HJ, Park CS, Cho EJ, Chung HO, Jeon HK, Lee JM, Oh YS, Chung WS, Chae JS, Kim JH, Choi KB, Hong SJ. Clinical significance of slow flow velocity in the distal left anterior descending coronary artery detected by transthoracic Doppler ehocardiography. Korean Circ J. 2002. 32:299–308.

17. Watanabe N, Akasaka T, Yamaura Y, Akiyama M, Koyama Y, Kamiyama N, Neishi Y, Kaji S, Saito Y, Yoshida K. Noninvasive detection of total occlusion of the left anterior descending coronary artery with transthoracic Doppler echocardiography. J Am Coll Cardiol. 2001. 38:1328–1332.

18. Badruddin SM, Ahmad A, Mickelson J, Abukhalil J, Winters WL, Nagueh SF, Zoghbi WA. Supine bicycle versus posttreadmil exercise echocardiography in the detection of myocardial ischemia: A randomized single-blind crossover trial. J Am Coll Cardiol. 1999. 33:1485–1490.

19. Roger VL, Pellikka PA, Oh JK, Miller FA, Seward JB, Tajik AJ. Stress echocardiography. Part I. Exercise echocardiography: Techniques, implementation, clinical applications and correlations. Mayo Clin Proc. 1995. 70:5–15.

20. Quiñones MA, Verani MS, Haichin RM, Mahmarian JJ, Suarez J, Zoghbi WA. Exercise echocardiography versus 201Tl single-photon emission computed tomography in evaluation of coronary artery disease: Analysis of 292 patients. Circulation. 1992. 85:1026–1031.

21. Pellikka PA, Roger VL, Oh JK, Miller FA, Seward JB, Tajik AJ. Stress echocardiography. Part II. Dobutamine stress echocardiography: Techniques, implementation, clinical applications and correlations. Mayo Clin Proc. 1995. 70:16–27.

22. Savonitto S, Ardissino D, Granger CB, Morando G, Prando MD, Mafrici A, Cavallini C, Melandri G, Thompson TD, Vahanian A, Ohman EM, Califf RM, Van de Werf F, Topol EJ. Prognostic value of the admission electrocardiogram in acute coronary syndromes. JAMA. 1999. 281:707–713.

23. Rainbird AJ, Mulvagh SL, Oh JK, McCully RB, Klarich KW, Shub C, Mahoney DW, Pellikka PA. Contrast dobutamine stress echocardiography: Clinical practice assessment in 300 consecutive patients. J Am Soc Echocardiogr. 2001. 14:378–385.

24. Czitrom D, Karila-Cohen D, Brochet E, Juliard JM, Faraggi M, Aumont MC, Assayag P, Steg PG. Acute assessment of microvascular perfusion patterns by myocardial contrast echocardiography during myocardial infarction: Relation to timing and extent of functional recovery. Heart. 1999. 81:12–16.

25. Vançon AC, Fox ER, Chow CM, Hill J, Weyman AE, Picard MH, Scherrer-Crosbie M. Pulse inversion harmonic imaging improves endocardial border visualization in two-dimensional images: Comparison with harmonic imaging. J Am Soc Echocardiogr. 2002. 15:302–308.

27. Kang DH, Kang SJ, Song JM, Choi KJ, Hong MK, Song JK, Park SW, Park SJ. Efficacy of myocardial contrast echocardiography in the diagnosis and risk stratification of acute coronary syndrome. Am J Cardiol. 2005. 96:1498–1502.

28. Kaul S, Senior R, Firschke C, Wang XQ, Lindner J, Villanueva FS, Firozan S, Kontos MC, Taylor A, Nixon IJ, Watson DD, Harrell FE. Incremental value of cardiac imaging in patients presenting to the emergency department with chest pain and without ST-segment elevation: A multicenter study. Am Heart J. 2004. 148:129–136.

29. Hillis GS, Mulvagh SL, Gunda M, Hagen ME, Reeder GS, Oh JK. Contrast echocardiography using intravenous octafluoropropane and real-time perfusion imaging predicts functional recovery after myocardial infarction. J Am Soc Echocardiogr. 2003. 16:638–645.

30. Lepper W, Hoffmann R, Kamp O, Franke A, de Cock CC, Kühl HP, Sieswerda GT, Dahl J, Janssens U, Voci P, Visser CA, Hanrath P. Assessment of myocardial reperfusion by intravenous myocardial contrast echocardiography and coronary flow reverse after primary percutaneous transluminal angioplasty in patients with acute myocardial infarction. Circulation. 2000. 101:2368–2374.

31. Chirillo F, Cavarzerani A, Ius P, Totis O, Bruni A, Valfré C, Stritoni P. Role of transthoracic, transesophageal, and transgastric two-dimensional and color Doppler echocardiography in the evaluation of mechanical complications of acute myocardial infarction. Am J Cardiol. 1995. 76:833–836.

32. Lemery R, Smith HC, Giuliani ER, Gersh BJ. Prognosis in rupture of the ventricular septum after acute myocardial infarction and role of early surgical intervention. Am J Cardiol. 1992. 70:147–151.

33. Menon V, Webb JG, Hillis LD, Sleeper LA, Abboud R, Dzavik V, Slater JN, Forman R, Monrad ES, Talley JD, Hochman JS. Outcome and profile of ventricular septal rupture with cardiogenic shock after myocardial infarction: A report from the SHOCK Trial Registry. Should we emergently revascularize occluded coronaries in cardiogenic shock? J Am Coll Cardiol. 1984. 36:1080–1087.
34. Kishon Y, Iqbal A, Oh JK, Gersh BJ, Freeman WK, Seward JB, Tajik AJ. Evolution of echocardiographic modalities in detection of post myocardial infarction ventricular septal defect and papillary muscle rupture: Study of 62 patients. Am Heart J. 1993. 126:667–675.

35. Purcaro A, Costantini C, Ciampani N, Mazzanti M, Silenzi C, Gili A, Belardinelli R, Astolfi D. Diagnostic criteria and management of subacute ventricular free wall rupture complicating acute myocardial infarction. Am J Cardiol. 1997. 80:397–405.

36. Yeo TC, Malouf JF, Oh JK, Seward JB. Clinical profile and outcome in 52 patients with cardiac pseudoaneurysm. Ann Intern Med. 1998. 128:299–305.

37. Goldstein JA. Pathophysiology and management of right heart ischemia. J Am Coll Cardiol. 2002. 40:841–853.

38. Ozdemir K, Altunkeser BB, Içli A, Ozdil H, Gök H. New parameters in identification of right ventricular myocardial infarction and proximal right coronary artery lesion. Chest. 2003. 124:219–226.

39. Esakof DD, Vannan MA, Pandian NG, Cao QL, Schwartz SL, Bojar RM. Visualization of left ventricular pseudoaneurysm with panoramic transesophageal echocardiography. J Am Soc Echocardiogr. 1994. 7:174–178.

40. Foster E, Schiller NB. The role of transesophageal echocardiography in critical care: UCSF experience. J Am Soc Echocardiogr. 1992. 5:368–374.

41. Widimsky P, Gregor P. Pericardial involvement during the course of myocardial infarction. A long-term clinical and echocardiographic study. Chest. 1995. 1081:89–93.
42. Tsang TS, Enriquez-Sarano M, Freeman WK, Barnes ME, Sinak LJ, Gersh BJ, Bailey KR, Seward JB. Consecutive 1127 therapeutic echocardiographically guided pericardiocentesis: Clinical profile, practice pattern, and outcomes spanning 21 years. Mayo Clin Proc. 2002. 77:429–436.

44. Appleton CP, Hatle LK, Popp RL. Cardiac tamponade and pericardial effusion: Respiratory variation in transvalvular flow velocities by Doppler echocardiography. J Am Coll Cardiol. 1988. 11:1020–1030.

45. O'Sullivan J, Heads A, Hunter S. Microbubble image enhancement and pericardiocentesis. Int J Cardiol. 1993. 42:95–96.
46. Erbel R, Börner N, Steller D, Brunier J, Thelen M, Pfeiffer C, Mohr-Kahaly S, Iversen S, Oelert H, Meyer J. Detection of aortic dissection by transesophageal echocardiography. Br Heart J. 1987. 58:45–51.
47. Khandheria BK, Tajik AJ, Taylor CL, Safford RE, Miller FA Jr, Stanson AW, Sinak LJ, Oh JK, Seward JB. Aortic dissection: Review of value and limitations of two-dimensional echocardiography in a six year experience. J Am Soc Echocardiogr. 1989. 2:17–24.

48. Hagan PG, Nienaber CA, Isselbacher EM, Bruckman D, Karavite DJ, Russman PL, Evangelista A, Fattori R, Suzuki T, Oh JK, Moore AG, Malouf JF, Pape LA, Gaca C, Sechtem U, Lenferink S, Deutsch HJ, Diedrichs H, Marcos y Robles J, Llovet A, Gilon D, Das SK, Armstrong WF, Deeb GM, Eagle KA. The international registry of acute aortic dissection (IRAD): New insights into old disease. JAMA. 2000. 283:897–903.

49. Erbel R, Oelert H, Meyer J, Puth M, Mohr-Katoly S, Hausmann D, Daniel W, Maffei S, Caruso A, Covino FE. Effect of medical and surgical therapy on aortic dissection evaluated by transesophageal echocardiography. Implications for prognosis and therapy. Circulation. 1993. 87:1604–1615.

50. Kwon JA, Youn HJ, Oh YS, Choi SH, Lee JS, Lee MH, Chang JH, Park HS, Park CS, Lee JM, Chung WS, Hong SJ. A case of acute aortic dissection involved left and right coronary arterial ostia diagnosed with transesophageal echocardiography. J Korean Soc Echocardiogr. 2001. 9:141–145.

51. Silverstein MD, Heit JA, Mohr DN, Petterson TM, O'Fallon WM, Melton LJ 3rd. Trends in the incidence of deep vein thrombosis and pulmonary embolism: A 25-year populationbased study. Arch Intern Med. 1998. 158:585–593.

52. Sinha N, Yalamanchili K, Sukhija R, Aronow WS, Fleisher AG, Maguire GP, Lehrman SG. Role of the 12-lead electrocardiogram in diagnosing pulmonary embolism. Cardiol Rev. 2005. 13:46–49.

53. Rodger M, Makropoulos D, Turek M, Quevillon J, Raymond F, Rasuli P, Wells PS. Diagnostic value of the electrocardiogram in suspected pulmonary embolism. Am J Cardiol. 2000. 86:807–809.

54. Kasper W, Geibel A, Tiede N, Bassenge D, Kauder E, Konstantinides S, Meinertz T, Just H. Distinguishing between acute and subacute massive pulmonary embolism by conventional and Doppler echocardiography. Br Heart J. 1993. 70:352–356.

55. Belenkie I, Dani R, Smith ER, Tyberg JV. Effects of volume loading during experimental acute pulmonary embolism. Circulation. 1989. 80:178–188.

56. McConnell MV, Solomon SD, Rayan ME, Come PC, Goldhaber SZ, Lee RT. Regional right ventricular dysfunction detected by echocardiography in acute pulmonary embolism. Am J Cardiol. 1996. 78:469–473.

57. Proano M, Oh JK, Frye RL, Johnson CM, Tajik AJ, Taliercio CP. Successful treatment of pulmonary embolism and associated mobile right atrial thrombus with use of central thrombolytic infusion. Mayo Clin Proc. 1988. 63:1181–1185.

58. Torbicki A, Galié N, Covezzoli A, Rossi E, De Rosa M, Goldhaber SZ. ICOPER study group. Right heart thrombi in pulmonary embolism: Results from the international cooperative pulmonary embolism resistry. J Am Coll Cardiol. 2003. 41:2245–2251.
59. Ribeiro A, Lindmarker P, Juhlin-Dannfelt A, Johnsson H, Jorfeldt L. Echocardiography Doppler in pulmonary embolism: Right ventricular dysfunction as a predictor of mortality rate. Am Heart J. 1997. 134:479–487.

60. Voelkel NF, Quaife RA, Leinwand LA, Barst RJ, McGoon MD, Meldrum DR, Dupuis J, Long CS, Rubin LJ, Smart FW, Suzuki YJ, Gladwin M, Denholm EM, Gail DB. National heart, lung, and blood institute working group on cellular and molecular mechanisms of right heart failure. Right ventricular function and failure. Circulation. 2006. 114:1883–1891.

61. Vannan Mani A. Atlas of echocardiography. 2005. 1st edition. Philadelphia: Current Medicine Group.




 PDF
PDF ePub
ePub Citation
Citation Print
Print








 XML Download
XML Download