Abstract
Danon disease is characterized clinically by the triad of cardiomyopathy, myopathy and mental retardation. It was originally reported as a lysosomal glycogen storage disease with normal acid maltase by Danon. Danon disease results from mutations in lysosome associated membrane protein-2 (LAMP-2) gene. The LAMP-2 gene is located on Xq24-25. We report a case of suspected Danon disease in patient who had hypertrophic cardiomyopathy and mental retardation along with abnormal findings in electromyography.
Hypertrophic cardiomyopathy (HCM) is defined by the presence of unexplained left ventricular hypertrophy (LVH). HCM is usually inherited as an autosomal dominant trait.1-4) Mutations in 10 genes related to the sarcomeric proteins account for approximately 60% of all cases of HCM.1-4) According to a recent gene analysis, abnormalities in many genes were found. Danon disease is one of them.3-5) Danon disease is caused by mutation of LAMP-2 gene and has a unique clinical manifestation of cardiomyopathy, myopathy and mental retardation.5-14) There is no case of Danon disease reported in Korea.
The authors experienced a 30 year-old male patient who had unique clinical features of Danon disease such as cardiomyopathy, myopathy and mental retardation. Therefore, we report this case with review of literature.
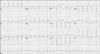
A 30-year-old man with mental retardation had admitted to our hospital complaining of dyspnea and orthopnea in April 2004. After echocardiographic examination, he was diagnosed as HCM at that time. Electromyography (EMG) was performed because his blood test showed an elevated creatinine kinase (CK) level. The patient was diagnosed as a myopathy with typical findings of EMG. After that admission, he was stable for about 2 years. After the hospitalization in 2004, the patient had been stabilized with use of carvediol, ramipril, digoxin and furosemide. However, his symptoms got worse from early 2007. In May 2007, he was rehospitalized for worsening dyspnea and general weakness. Murmur and irregular heart sound were heard from the chest by auscultation. In the abdominal examination, the liver was palpated with 4 fingerbreadths and mild tenderness was induced. However, there was no pretibial pitting edema. His parents had no certain disease other than mental retardation. In complete blood counts, WBC 16,600/mm3, neutrophil 92%, hemoglobin 11.8 g/dL and platelet 101,000/mm3 were found. The serum electrolyte test was within normal range. In the cardiac enzyme test, CK was 739 IU/L, CK-MB was 6.11 ng/mL and myoglobin was increased to 198 ng/dL. The serum level of brain natriuretic peptide was increased to 1468 pg/mL (normal range: 0-100 pg/mL). The chemistry test showed AST 59 IU/L, ALT 38 IU/L, BUN 15.3 mg/dL and Cr 0.9 mg/dL. The chest X-ray showed pulmonary congestion and cardiomegaly. Electrocardiography showed atrial flutter with 133 beats of heart rate (Fig. 1). The echocardiogram revealed global hypokinesia and calculated left ventricular (LV) ejection fraction (EF) was 13%. The inner diameters for the LV end diastole and systole were 56 mm and 52 mm, respectively (Fig. 2A and B). The thickness of LV wall was 9 mm in May 2007. In comparison with echocardiogram in 2004, the LVEF and LV inner diameters are similar, but the LV wall thickness decreased from 14 mm to 9 mm (Fig. 2). These results of echocardiography showed the typical pattern of progression to the dilated cardiomyopathy. Electromyographic findings were positive sharp waves and fibrillation potentials on muscles. In spite of supportive management, cardiogenic shock was not resolved and he died at the day of admission.
HCM is a common inherited cardiac disease with remarkable clinical and genetic heterogenecity.2) In the majority of HCM, it is caused by mutations in genes that encode different cardiac sarcomeric proteins.1-4) However, HCM population without sarcomere gene mutations could have one of several diseases that mimic the phenotypic expression of HCM.3) Hypertrophy caused by non-sarcomeric gene mutation includes various type of metabolic disease, such as Danon, Pompe or Fabry disease and AMP activated protein kinase γ2 (PRKAG2) mutation, in which the myocardium is infiltrated with atypical cellular byproducts. Collectively, these disorders are defined as a subset of unexplained hypertrophy such as storage cardiomyopathies, rather than HCM.1)4-6)14)15)
Danon disease is caused by mutation of LAMP-2 gene, which encodes the lysosomal membrane protein.5-12) It is an X-linked dominant disorder but de novo mutations are possible.7)8) The natural history of Danon disease is characterized in male patients by an early onset and a very poor prognosis.8) Male patients with Danon disease usually display the typical clinical triad. However, female patients usually present with isolated cardiomyopathy.6)10) The reasons for the difference are that the lack of LAMP-2 varies according to the mutation types of LAMP-2 gene.9)
The pathologic hallmark of sarcomeric gene mutations is myocyte hypertrophy, myocyte disarray, and increased interstitial fibrosis, whereas myocytes in storage cardiomyopathies show hypertrophy with vacuoles or aggregates of disease-associated material.1)13)15) In addition to these microscopic differences, there are clinical distinctions between HCM and storage cardiomyopathies. There is high prevalence of electrical abnormalities including ventricular pre-excitation and progressive conduction system defects in patients with storage cardiomyopathies. The rate of progression from hypertrophy to dilation and severe heart failure is also higher in storage cardiomyopathies than in HCM.1)3)4)14)15) The distinct clues of Danon disease from other storage cardiomyopathies are the accumulation of autophagic material and the lack of LAMP-2 in muscle.3-5) The onset age of Danon disease is younger than other diseases. Also there is no specific treatment for Danon disease and poor prognosis because of fast progress to end- stage heart failure.3)4)8)14)
Because this case showed mental retardation, HCM, myopathy, and rapid progression toward end-stage heart failure early in adulthood, we doubted Danon disease as a diagnosis. However, the patient was not confirmed as Danon disease because his family did not consent the biopsy of the skeletal muscle to diagnosis and we did not identify the LAMP-2 gene mutation through a genetic analysis.
The echocardiographic features of Danon disease are similar to these of HCM. However, they have very different clinical manifestations, mode of inheritance, prognosis and subsequently different genetic counseling. A careful clinical evaluation for this disease should be taken into consideration for all patients with HCM. Especially, for young male patients with HCM under 30-year old, the possibility of Danon disease should be considered.
Figures and Tables
References
1. Alcalai R, Seidman JG, Seidman CE. Genetic basis of hypertrophic cardiomyopaty: from bench to the clinics. J Cardiovasc Electrophysiol. 2008. 19:104–110.
2. Maron BJ. Hypertrophic cardiomyopathy: a systematic review. JAMA. 2002. 287:1308–1320.
3. Keren A, Syrris P, McKenna WJ. Hypertrophic cardiomyopathy: the genetic determiants of clinical disease expression. Nat Clin Pract Cardiovasc Med. 2008. 51:158–168.
4. Lind JM, Chiu C, Semsarian C. Genetic basis of hypertrophic cardiomyopathy. Expert Rev Cardiovasc Ther. 2006. 4:927–934.

5. Arad M, Maron BJ, Gorham JM, Johnson WH, Saul JP, Perez-Atayde AR, Spirito P, Wright GB, Kanter RJ, Seidman CE, Seidman JG. Glycogen storage diseases presenting as hypertrophic cardiomyopathy. N Engl J Med. 2005. 352:362–372.

6. Danon MJ, Oh SH, Di Mauro S, Manaligod JR, Eastwood A, Naidu S. Lysosomal glycogen storage disease with normal acid maltase. Neurology. 1981. 31:51–57.

7. Charron P, Villard E, Sébillon P, Laforêt P, Maisonobe T, Duboscq-Bidot L, Romero N, Drouin-Garraud V, Frébourg T, Richard P, Eymard B, Komajda M. Danon's disease as a cause of hypertrophic cardiomyopathy: a systematic survey. Heart. 2004. 90:842–846.

8. Sugie K, Yamamoto A, Murayama BS, Oh SJ, Takahashi M, Mora M, Riggs JE, Colomer J, Iturriaga C, Meloni A, Lamperti C, Saitoh S, Byrne E, DiMauro S, Nonaka I, Hirano M, Nishino I. Clinicopathological feature of genetically confirmed Danon disease. Neurology. 2002. 58:1773–1778.

9. Fanin M, Nascimbeni AC, Fulizio L, Spinazzi M, Melacini P, Anqelini C. Generalized Lysosome-associated membrane protein-2 defect explains multisystem clinical involvement and allows leukocyte diagnostic screening in Danon disease. Am J Pathol. 2006. 168:1309–1320.

10. Nishino I, Fu J, Tanji K, Yamada T, Shimojo S, Koori T, Mora M, Riggs JE, Oh SJ, Koga Y, Sue CM, Yamamoto A, Murakami N, Shanske S, Byme E, Bonilla E, Nonaka I, DiMauro S, Hirano M. Primary LAMP-2 deficiency causes X-linked vacuolar cardiomyopathy and myopathy (Danon disease). Nature. 2000. 406:906–910.

11. Sugimoto S, Shiomi K, Yamamoto A, Nishino I, Nonaka I, Ohi T. LAMP-2 positive vacuolar myopahty with dilated cardiomyopathy. Intern Med. 2007. 46:757–760.
12. Taylor MR, Ku L, Slavov D, Cavanaugh j, Boucek M, Zhu X, Carniel E, Barnes C, Quan D, Prall R, Lovell MA, Mierau G, Ruegg P, Mandava N, Bristow MR, Towbin JA, Mestroni L. Danon disease presenting with dilated cardiomyopathy and a complex phenotype. J Hum Genet. 2007. 52:830–835.

13. Christine S. Genetic cause of inherited cardiac hypertrophy. Mayo Clin Proc. 2002. 77:1315–1319.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download