Abstract
Acute pulmonary thromboembolism (PTE) is a common problem. In the emergency room, a substantial number of patients with chest discomfort have had an acute PTE. Presently, accepted diagnostic modalities for the confirmation of PTE include ventilation/perfusion (V/Q) scanning, chest computed tomography (CT), and standard angiography. However, each modality has important limitations. Although chest CT scanning has for the most part replaced lung scanning as the main diagnostic test for PTE, transthoracic echocardiography (TTE) is a noninvasive modality providing rapid results at the bedside. As well as being noninvasive modality, TTE avoids the contrast and radiation hazards of chest CT or conventional angiography. Thus echocardiography is an attractive imaging modality to diagnose PTE. TTE allows visualization of the aorta and the LV to evaluate for other etiologies of chest pain. Besides giving important diagnostic information, TTE can aid prediction of prognosis. Presence of RV dilatation and dysfunction is a poor prognostic sign and is the indicator for thrombolytic therapy. However, TTE has some limitations including poor imaging quality depending on the acoustic window and has a low sensitivity in detecting PTE.
Acute pulmonary thromboembolism (PTE) is a common problem. There are at least 600,000 estimated cases of newly diagnosed PTE and more than 50,000 related deaths in the United States each year.1)2) The overall 3-month mortality rate for all patients who present with PTE is reported to be about 15% and the reported mortality rate for those in shock is nearly 50%.3) This fatality rate for PTE exceeds the mortality rate for acute myocardial infarction. In spite of increasing knowledge about PTE and imaging technology, the antemortem diagnosis of fatal PTE has not changed appreciably over the last 40 years and remains fixed at approximately 30%.1)3)
Acute coronary syndromes, acute aortic syndromes and acute PTE are the three major disease entities in patients presenting with chest pain in the emergency room. The problem is the similarity in clinical presentation between acute coronary syndromes and PTE. The most common symptoms in PTE include dyspnea in 80% and chest discomfort in 65% of patients. However, these symptoms are nonspecific in the differentiation of PTE and acute coronary syndromes.4-6) Also, features of PTE on physical examination are nondiscriminatory in the differentiation. Given the significant overlap of signs and symptoms between PTE and acute coronary syndromes, clinicians, especially cardiologists, should be familiar with this disease entity. PTE should be considered as one of the possible causes.
Presently, accepted diagnostic modalities for the confirmation of PTE include ventilation/perfusion (V/Q) scanning, chest computed tomography (CT), and standard angiography. Although chest CT scanning has for the most part replaced lung scanning as the main diagnostic test for PTE, transthoracic echocardiography (TTE) is a noninvasive modality providing rapid results at the bedside. This modality is one of the most useful tests in aiding diagnosis and risk stratification in patients with PTE. In this review, we will discuss the role of echocardiography in patients with PTE.
PTE can have various pathophysiological effects. Increased pulmonary vascular resistance primarily caused by vascular obstruction. The size of the pulmonary embolus determines the initial hemodynamic compromise. If the RV systolic pressure acutely increases to more than 40 to 50 mmHg, the RV may fail. Loss of RV function then leads to an acute drop in forward cardiac output and subsequent hemodynamic collapse.7) The acute RV distension may shift the interventricular septum to the left and impair the left ventricular function.8) Further pressure increase is evoked by neurohumoral agents or pulmonary baroreceptors. The release of serotonin, histamine and other inflammatory platelet-related molecules during the episode impair hemodynamic collapse. Airway resistance is increased due to bronchoconstriction and decreased pulmonary compliance is induced by lung edema, lung hemorrhage and loss of surfactant.9)
Although the diagnosis of PTE is difficult, noninvasive diagnostic approaches have become increasingly reliable. The most important point is to remember to consider the possible diagnosis of PTE. The most useful approach is the clinical assessment of likelihood, based on presenting signs and symptoms, in conjunction with other diagnostic testing.
Plasma D-dimer assay plays an important role in the diagnosis of acute PTE. The traditional enzyme-linked immunosorbent assays (ELISAs) are generally available and have a very high sensitivity (98%). Although elevated plasma concentrations of D-dimer are sensitive for the presence of PTE, it is not specific. When PTE is not highly suspected, plasma D-dimer by ELISA usually suffices to rule out this condition.
Conventional electrocardiography has some additional roles besides presentation of diagnostic clues. On electrocardiogram, classic findings for an acute PTE include acute right heart overload such as S1Q3T3, a newly developed right bundle branch block, right axis deviation and P pulmonale.8) Electrocardiograms can be used to exclude acute myocardial infarction. Also, T wave inversion in leads V1 to V4 has the greatest accuracy for identifying RV dysfunction in patients with acute PTE.10)
V/Q scanning was the most used test before the introduction of chest CT scanning to the clinical field. The PIOPED (Prospective Investigation of Pulmonary Embolism Diagnosis) investigators reported the sensitivity of V/Q scanning in the diagnosis of acute PTE.11) They reported that high probability results are very helpful in the diagnosis of acute PTE and a high clinical probability yielded a diagnostic accuracy of 96%. However, with a high-probability scan, patients with a low probability of PTE according to clinical screening criteria had documentation of a PTE by angiography in around 50% of cases. Although V/Q scanning has been the first line test for evaluation of acute PTE in past years, it has several limitations. V/Q scans are relatively expensive, not readily available, and usually require further diagnostic tests.11)12)
Noninvasive imaging modalities, including chest CT and magnetic resonance imaging (MR), are commonly used clinically. In particular, chest CT has been replacing the inconvenient V/Q scanning and the invasiveness of pulmonary angiography in recent years, especially in patients with normal renal function. Also, CT scans provide the images of other adjacent structures including lung parenchyma, pleura and mediastinum besides pulmonary vasculatures.13) Perrier et al.14) reported the diagnostic accuracy with helical chest CT imaging of 229 patients in a total 1108 consecutive PTE patients. They demonstrated that the sensitivity and specificity of a normal CT pulmonary angiography scan was approximately that of a low-probability V/Q scan (70 and 90%, respectively).14) More recent data showed a comparison of CT pulmonary angiography with V/Q scan.15) The study revealed that CT pulmonary angiography was not inferior to V/Q scanning in ruling out PTE. However, significantly more patients were diagnosed with PTE using CT angiography. Recently, chest multi-detector CT scanning has been used in the differential diagnosis of acute chest pain.16) Moreover, it can detect RV dysfunction and provide important treatment information in patients with acute PTE.17) Although CT scanning is relatively expensive, unavailable at the bedside, requires intravenous contrast agents and gives a relatively high dose of radiation, it is noninvasive, widely available, sensitive for the diagnosis of PTE, and produces quick results. Thus helical CT is rapidly becoming the imaging test of choice for the evaluation of acute PTE.
MR is emerging noninvasive modality. In a recent study by Kluge et al.,18) multitechnique thoracic MR followed by MR venography was used successfully to search for both deep venous thrombosis (DVT) and PTE. Among 207 combined examinations, PTE was diagnosed in 76 and DVT in 78 examinations. Thirteen patients without PTE showed DVT; thus, MR venography detected 17% additional cases of thromboembolism. With using MR imaging technique, evaluation of PE and DVT was routinely feasible as one-stop-shopping. MRI can be considered a second-line technique to avoid contraindications to CT.
Pulmonary angiography used to be regarded as the gold standard for the evaluation of PTE. Typical findings described for acute PTE during angiography include an intraluminal filling defect, vessel cutoff, areas of oligemia, and asymmetry of blood flow.19) However, this technique is invasive and may be associated with serious complications. Current indications for the diagnosis of PTE reserve conventional angiography for patients in whom noninvasive and laboratory testing is inconclusive, and who need some interventions.8)
Because transthoracic echocardiography (TTE) is noninvasive, provides rapid bedside results, and avoids the contrast and radiation hazards of chest CT, echocardiography is a potentially attractive imaging modality to diagnose PTE. Also, it is usually available in most hospitals.
Most common clinical indications of the echocardiography are evaluation of presence of RV dysfunction and grade of pulmonary hypertension. With this technique, clinicians can understand hemodynamic status of the PTE. Also, TTE allows visualization of the aorta and the left ventricle (LV) to evaluate for other etiologies of chest pain, thus making it an attractive test. However, TTE is limited by the acoustic windows available.
The echocardiography uses indirect signs of the hemodynamic consequences of acute RV pressure overload to diagnose acute PTE.20) TTE can aid in the diagnosis of PTE and demonstrate other etiologies for chest pain by investigation for the acute hemodynamic consequences of PTE. In patients with acute PTE, RV dilatation, RV dysfunction and hypokinesis of the RV wall can be identified (Fig. 1).8) In normal subjects, an obstruction of more than 30% of the pulmonary vasculature can produce RV dysfunction. A way to evaluate RV dilatation and dysfunction is from the ratio of the RV end-diastolic area to LV end-diastolic area. The normal ratio is about 0.6. A ratio of more than 1.0 is consistent with severe RV dysfunction.21) Other echocardiographic findings consistent with PTE (specifically pulmonary hypertension) are flattening of the interventricular septum and LV diastolic dysfunction. Flattening of the interventricular septum occurs when the transseptal pressure gradient reverses caused by RV pressure overload during diastolic period (Fig. 2). In these patients, low cardiac output with shock and a prominent A-wave in pulsed wave Doppler recording of the mitral flow can be seen.22-25) LV impairment occurs when there is a reduced LV dimension associated with RV dilatation. Other proposed markers of RV dysfunction including RV/LV end-diastolic area ratio were listed on the Table 1.26)
The McConnell sign is one of the features to look for in patients with acute PTE with TTE (Fig. 3).27) This sign shows normal contraction or sparing of the RV apex with hypokinesis of the midportion of the RV free wall. With this sign, acute PTE can be diagnosed with a sensitivity of 77% and a specificity of 94%. It can be used to differentiate acute PTE from primary pulmonary hypertension. However, in one study reported by Miniati et al.,28) the hypokinesis of the RV wall was diagnosed in about 44% of those with acute PTE.
TTE allows for the evaluation of tricuspid regurgitation (TR) and an indirect assessment of systolic pulmonary arterial (PA) pressure. It can be obtained by evaluating the continuous wave Doppler analysis. With normal pulmonic valvular function, the systolic PA pressure can be estimated by the maximal velocity of the TR jet with the Bernoulli equation (systolic PA pressure=RA pressure+4 ×TR Vmax2). Right atrial (RA) pressure can be estimated from the size of inferior vena cava and its respiratory motion (caval index).29) The estimation of RA pressure is listed in Table 2. Patients with acute pulmonary hypertension typically have TR Vmax more than 3.0 m/s corresponding to a systolic PA pressure of about 40 mmHg. The estimation of PA pressure with TR velocity is feasible and has high sensitivity and specificity.
The pulsed wave Doppler analysis of RV outflow tract can give another method for evaluation of pulmonary hypertension.30) The acceleration phase becomes shorter with increased PA pressure. Mean PA pressure can be estimated using Mahan's equation (Mean PA pressure=79-0.45×RV outflow tract acceleration time). Mean PA pressure can be estimated with late pulmonic valve regurgitation velocity (Mean PA pressure=4×Peak PR velocity2).31)
With TTE, a free-floating thrombus or a clot transiting through the right heart can be found. Although these findings are the most diagnostic, their visualization occurs only in about 7% of all TTE studies for PTE (Fig. 4). In addition to imaging an embolus-in-transit,32) TTE gives the ability to directly visualize the main and right pulmonary artery (PA) and proximal portion of left PA.33)34)
The diagnostic utility of TTE has been evaluated. Grifoni et al.35) reported the diagnostic probability of TTE as a tool for triage in the emergency room in 117 consecutive patients with suspected acute PTE. In this study, TTE had a sensitivity of 51% and a specificity of 87%. The positive predictive value was 82% and the negative predictive value was 60%. In another study by Jackson et al.,36) similar results were shown in 124 consecutive patients with suspected acute PTE. TTE yielded a sensitivity of 41% and a specificity of 91%. The conclusion of the study was that TTE does not have sufficient sensitivity to exclude the diagnosis of acute PTE. Therefore, outside the intensive care unit (ICU), as a stand-alone imaging technique for the diagnosis of PTE, echocardiography should not be used alone.
However, in the ICU, echocardiography is attractive since it is noninvasive and does not require the transport of an unstable patient. In one study performed with the ICU patients, the common TTE findings of acute PTE were worsening tricuspid regurgitation (90%), pulmonary hypertension (77%), dilated RV (74%) and right heart strain (61%).37) Moreover, 80% of patients with shock and PTE show evidence of right-heart dysfunction on echocardiography.38)
However, in the patient with unexplained shock, the differential diagnosis is broad and includes a number of processes that can cause both RV dilation and dysfunction. Thus, findings from TTE, even in patients with shock suspected to be due to a PTE, are nonspecific. Even if echocardiography findings are nonspecific for PTE, these may still provide important information by helping to exclude other causes of shock, such as acute LV dysfunction, tamponade, acute valvular disease, and aortic dissection.38)
Myocardial strain is a dimensionless index of change in myocardial length in response to the applied force and is expressed as a fraction or percent change.39) Peak systolic strain analyzed by tissue Doppler imaging has been used to evaluate ventricular function. It correlated well with changes in stroke volume and was more closely related to changes in global hemodynamics.40) Recently, tissue Doppler imaging has been used to assess RV function objectively in the patient with an acute PTE.41)42) Kjaergaard et al.41) reported RV strain changes during the acute and recovery phase of PTE demonstrating the hypokinesis of RV free wall with strain analysis. Park et al.42) showed that decreased peak systolic strain of RV midventricle was related to decreased RV systolic function; this value was improved after treatment (Fig. 5). In this study, the negative value of the RV midventricular strain may be from the outward dilatation of the RV free wall during the systole in the acute period and suggests decreased RV free wall motion. However, the value became normalized after normalization of pulmonary arterial pressure after successful treatment. In other study by Hsiao et al.,43) they reported the usefulness of regional myocardial performance index of RV (RV MPI) by tissue Doppler to diagnose PTE. They concluded that the RV MPI >0.55 identified PE with a sensitivity of 85% and a specificity of 78%.
Transesophageal echocardiography (TEE) provides an excellent image quality compared with TTE. It can be used in the patients with a poor acoustic window. For detection of PTE, TEE has 60-80% sensitivity and a 95-100% specificity. In particular, for the detection of central PTE, TEE has 90-95% sensitivity and a 100% specificity.44) TEE, like TTE, also allows visualization of secondary changes in cardiac chamber size and function due to the hemodynamic effects of the PTE. In one study by Rosenberger et al.,45) TEE can give direct visualization of pulmonary thromboemboli (46%). However, the sensitivity for direct visualization of thromboemboli at any specific location was only 26%. Other TEE evidence of RV dysfunction was observed in 96%, and TR in 50%.
However, TEE is not as widely available as TTE and requires special training in performance and interpretation. TEE also requires some sedation of the patient, which may be difficult if the patient is hemodynamically unstable or is respiratory compromised.44)
In patients with acute PTE, the presence of RV dysfunction has been found to have a significantly poor prognosis. So, RV dilatation and dysfunction judged by echocardiography can be used in the risk stratification for PTE management. The presence of RV dysfunction and shock are the indicator for the use of thrombolytic agents such as tissue plasminogen activator.46) Toosi et al.47) reported their data associated with risk stratification of 159 patients with acute PTE. In their article, moderate to severe RV hypokinesis and RV/LV end-diastolic area ratio >1.0 were significantly associated with higher in-hospital mortality and demonstrated the best predictive values for short-term outcomes. Moreover, they reported that the sensitivity and negative predictive value of diastolic LV impairment, moderate to severe RV hypokinesis and RV dilatation (RV/LV end-diastolic area ratio >1.0 and RV end-diastolic area >30 mm) for in-hospital mortality were 100%.
In one prospective study of 209 consecutive patients diagnosed with an acute PTE without hemodynamic compromise, 65 patients (31%) had RV dysfunction.48) In these patients, the mortality rate was much higher than those patients without RV dysfunction (5% with RV dysfunction vs. 1.2% without RV dysfunction). In the ICOPER registry (International Cooperative Pulmonary Embolism Registry) including 2454 consecutive patients with acute PTE, 40% of the patients had RV dysfunction, and 4% had free-floating cardiac thrombi.49) The overall 90-day mortality was 15.3%. Multiple-regression modeling revealed that RV dysfunction was a strong and significant predictor of death.4)
Combining echocardiographic data and laboratory tests can give additional prognostic implications. In 124 consecutive patients with acute PTE, Binder et al.50) reported their result about the combination of echocardiographic data with either NT-pro B-type natriuretic peptide (BNP) or cardiac troponin T (cTnT) in the prediction of complicated in-hospital course or death. With a positive echocardiographic results and either NT-proBNP >1000 pg/mL or cTnT >0.04 ng/mL, the risk of severe complication or death was approximately 38%. In other study evaluating the association of RV enlargement, elevated cardiac troponin I (cTnI) and 30 day mortality in patients with acute PTE, the mortality rate for patients with cTnI >0.1 ng/mL was 32%, with RV enlargement 28% and for patients with both findings 38%.51)
PTE remains a life-threatening disease and one of three major disease entities with chest discomfort seen in the emergency room. Despite progress in imaging techniques and knowledge of this disease, its medical diagnosis is one of the most difficult to achieve. The clinical assessment of PTE probability remains central to the diagnosis and evaluation. Presently, accepted diagnostic modalities for the confirmation of PTE include V/Q scanning, chest CT, and standard angiography. All approaches have limitations and doctors must aware their cons and pros.
Because echocardiography is noninvasive, provides rapid bedside results, and avoids the contrast and radiation hazards of chest CT or conventional angiography, echocardiography is an attractive imaging modality to diagnose PTE. Also, echocardiography allows visualization of the aorta and the LV to evaluate for other etiologies of chest pain, thus making it a more attractive test. Recently, many researchers tried to evaluate RV function more objectively with tricuspid annular plane systolic excursion, systolic velocity of tricuspid valvular annulus, RV myocardial performance index or myocardial strain. However, it is uncertain as yet as to the role of these indices in the detection of PTE or in the prediction of prognosis. Further evaluation should be needed.
Besides giving important diagnostic information, echocardiography can aid prediction of prognosis. Presence of RV dilatation and dysfunction is a poor prognostic sign and is the indicator for thrombolytic therapy. Moreover, using echocardiographic data with laboratory data (i.e. NT-proBNP or cTnT and cTnI) can give additional prognostic information in these patients. A proposed flow chart for the evaluation of PTE is pictured in Fig. 6. Echocardiography is optional in the clinical suspicion of pulmonary embolism. However, it can give information about possible causes of shock or presence of pulmonary hypertension in that step. Though echocardiography showed poor sensitivity in the detection of PTE, it can be also used in the diagnostic step. However, the definitive diagnosis should be made by other diagnostic modality. Echocardiography is mandatory in the determination of prognosis. Presence of RV dysfunction or shock are associated with poor prognosis, thrombolysis or thrombectomy should be considered.
Figures and Tables
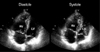 | Fig. 1Characteristic features of acute pulmonary thromboembolism identified by transthoracic echocardiography. Right ventricular dilatation and dysfunction were seen in a patient with acute pulmonary thromboembolism. In the diastole, the ratio of the right ventricular end-diastolic area to left ventricular end-diastolic area was more than 1.0 which is consistent with severe right ventricular dysfunction (normal value is less than 0.6). |
 | Fig. 2Systolic and diastolic images with a parasternal short axis view from a patient with an acute pulmonary thromboembolism revealing septal flattening (D-shaped left ventricle) consistent with right ventricular pressure overload. |
 | Fig. 3McConnell's sign. Regional wall thickening fractions (mean±standard deviation) were presented in normal patients, patients with primary pulmonary hypertension (PPH), and those with a PTE. While apical wall motion was reduced in the PPH patients, it was intact in PTE patients. (Figure from McConnell MV, et al. Regional right ventricular dysfunction detected by echocardiography in acute pulmonary embolism. Am J Cardiol 1996;78:469-73). |
 | Fig. 4Free-floating thrombi were found by transthoracic echocardiographic exam in a patient with acute pulmonary thromboembolism. A. A huge thrombus (arrows) was seen in the right atrium extended to right ventricle and crossing the tricuspid valve. B. After the opening of the right atrial wall, a worm like thrombus was found in the right atrium. |
 | Fig. 5Doppler tissue images of the right ventricular (RV) free wall were obtained using a low parasternal view, and three segments (apex, midventricle and base) were evaluated with an offset length of 12 mm. Baseline analysis showed decreased strain of the midventricle of the RV (A). Follow-up analysis showed increased strain of the midventricle with treatment (B). (Park JH, et al. Int J Cardiol 2008;125:319-24.) |
 | Fig. 6A proposed algorithm of diagnosis and care for patient with an acute pulmonary thromboembolism (PTE). Imaging modality in the bracket is optional according to the clinical situation. PEx: physical examination, CXR: chest X-ray, ECG: electrocardiography, VQ: ventilation/perfusion scan, MR: magnetic resonance imaging, PAgram: pulmonary ateriogram, RV: right ventricle, pro-BNP: pro B type natriuretic peptide, cTnT: cardiac troponin T, cTnI: cardiac troponin I. |
References
1. Wood KE. Major pulmonary embolism: review of a pathophysiologic approach to the golden hour of hemodynamically significant pulmonary embolism. Chest. 2002. 121:877–905.
2. Lilienfeld DE, Chan E, Ehland J, Godbold JH, Landrigan PJ, Marsh G. Mortality from pulmonary embolism in the United States: 1962 to 1984. Chest. 1990. 98:1067–1072.

3. Stein PD, Henry JW. Prevalence of acute pulmonary embolism among patients in a general hospital and at autopsy. Chest. 1995. 108:978–981.

4. Simonneau G, Azarian R, Brenot F, Dartevelle PG, Musset D, Duroux P. Surgical management of unresolved pulmonary embolism. A personal series of 72 patients. Chest. 1995. 107:52S–55S.
5. Stein PD, Henry JW. Clinical characteristics of patients with acute pulmonary embolism stratified according to their presenting syndromes. Chest. 1997. 112:974–979.

6. Stein PD, Terrin ML, Hales CA, Palevsky HI, Saltzman HA, Thompson BT, Weg JG. Clinical, laboratory, roentgenographic, and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest. 1991. 100:598–603.

7. Konstantinides S. Pulmonary embolism: impact of right ventricular dysfunction. Curr Opin Cardiol. 2005. 20:496–501.

8. Rahimtoola A, Bergin JD. Acute pulmonary embolism: an update on diagnosis and management. Curr Probl Cardiol. 2005. 30:61–114.

9. Goldhaber SZ, Elliott CG. Acute pulmonary embolism: part I: epidemiology, pathophysiology, and diagnosis. Circulation. 2003. 108:2726–2729.
10. Punukollu G, Gowda RM, Vasavada BC, Khan IA. Role of electrocardiography in identifying right ventricular dysfunction in acute pulmonary embolism. Am J Cardiol. 2005. 96:450–452.

11. The PIOPED Investigators. Value of the ventilation/perfusion scan in acute pulmonary embolism. Results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA. 1990. 263:2753–2759.
12. Bone RC. Ventilation/perfusion scan in pulmonary embolism. 'The Emperor is incompletely attired'. JAMA. 1990. 263:2794–2795.

13. Kim KI, Muller NL, Mayo JR. Clinically suspected pulmonary embolism: utility of spiral CT. Radiology. 1999. 210:693–697.

14. Perrier A, Howarth N, Didier D, Loubeyre P, Unger PF, de Moerloose P, Slosman D, Junod A, Bounameaux H. Performance of helical computed tomography in unselected outpatients with suspected pulmonary embolism. Ann Intern Med. 2001. 135:88–97.

15. Anderson DR, Kahn SR, Rodger MA, Kovacs MJ, Morris T, Hirsch A, Lang E, Stiell I, Kovacs G, Dreyer J, Dennie C, Cartier Y, Barnes D, Burton E, Pleasance S, Skedgel C, O'Rouke K, Wells PS. Computed tomographic pulmonary angiography vs ventilation-perfusion lung scanning in patients with suspected pulmonary embolism: a randomized controlled trial. JAMA. 2007. 298:2743–2753.

16. Johnson TR, Nikolaou K, Wintersperger BJ, Knez A, Boekstegers P, Reiser MF, Becker CR. ECG-gated 64-MDCT angiography in the differential diagnosis of acute chest pain. AJR Am J Roentgenol. 2007. 188:76–82.

17. Dogan H, Kroft LJ, Huisman MV, van der Geest RJ, de Roos A. Right ventricular function in patients with acute pulmonary embolism: analysis with electrocardiography-synchronized multi-detector row CT. Radiology. 2007. 242:78–84.

18. Kluge A, Mueller C, Strunk J, Lange U, Bachmann G. Experience in 207 combined MRI examinations for acute pulmonary embolism and deep vein thrombosis. AJR Am J Roentgenol. 2006. 186:1686–1696.

19. Dalen JE, Brooks HL, Johnson LW, Meister SG, Szucs MM Jr, Dexter L. Pulmonary angiography in acute pulmonary embolism: indications, techniques, and results in 367 patients. Am Heart J. 1971. 81:175–185.

20. Kim EM, Park JH, Jang WI, Ahn KT, Kim MS, Kim KS, Jeong IS, Lee JH, Na KR, Seong IW. A case with acute pulmonary thromboembolism associated wtih nephrotic syndrome. J Cardiovasc Ultrasound. 2007. 15:101–104.

22. Cheriex EC, Sreeram N, Eussen YF, Pieters FA, Wellens HJ. Cross sectional Doppler echocardiography as the initial technique for the diagnosis of acute pulmonary embolism. Br Heart J. 1994. 72:52–57.

23. Come PC. Echocardiographic evaluation of pulmonary embolism and its response to therapeutic interventions. Chest. 1992. 101:151S–162S.

24. Jardin F, Dubourg O, Gueret P, Delorme G, Bourdarias JP. Quantitative two-dimensional echocardiography in massive pulmonary embolism: emphasis on ventricular interdependence and leftward septal displacement. J Am Coll Cardiol. 1987. 10:1201–1206.

25. Kasper W, Geibel A, Tiede N, Bassenge D, Kauder E, Konstantinides S, Meinertz T, Just H. Distinguishing between acute and subacute massive pulmonary embolism by conventional and Doppler echocardiography. Br Heart J. 1993. 70:352–356.

26. Kreit JW. The impact of right ventricular dysfunction on the prognosis and therapy of normotensive patients with pulmonary embolism. Chest. 2004. 125:1539–1545.

27. McConnell MV, Solomon SD, Rayan ME, Come PC, Goldhaber SZ, Lee RT. Regional right ventricular dysfunction detected by echocardiography in acute pulmonary embolism. Am J Cardiol. 1996. 78:469–473.

28. Miniati M, Monti S, Pratali L, Di Ricco G, Marini C, Formichi B, Prediletto R, Michelassi C, Di Lorenzo M, Tonelli L, Pistolesi M. Value of transthoracic echocardiography in the diagnosis of pulmonary embolism: results of a prospective study in unselected patients. Am J Med. 2001. 110:528–535.

29. Kircher BJ, Himelman RB, Schiller NB. Noninvasive estimation of right atrial pressure from the inspiratory collapse of the inferior vena cava. Am J Cardiol. 1990. 66:493–496.

30. Chan KL, Currie PJ, Seward JB, Hagler DJ, Mair DD, Tajik AJ. Comparison of three Doppler ultrasound methods in the prediction of pulmonary artery pressure. J Am Coll Cardiol. 1987. 9:549–554.

31. Masuyama T, Kodama K, Kitabatake A, Sato H, Nanto S, Inoue M. Continuous-wave Doppler echocardiographic detection of pulmonary regurgitation and its application to noninvasive estimation of pulmonary artery pressure. Circulation. 1986. 74:484–492.

32. Lee JH, Park JH, Kim EM, Jang WI, Ahn KT, Kim MS, Kim KS, Jeong IS, Jeong JO, Seong IW. Prelude of tragedy: Entrapped huge thrombi into a patent foramen ovale impending paradoxical embolism. J Cardiovasc Ultrasound. 2007. 15:121–123.

33. lee KH, Hwang SO, Kim YS, Ahn ME, Cho YK, Yoon JH, Park KS, Choe KH. Thrombi in main pulmonary artery detected by echocardiography. J Kor Soc Echo. 1994. 2:225–228.

34. Lee BH, Son IS, Kim SJ, Moon JY, Kang HS, Choue CW, Kim KS, Song JS, Bae JH. A case of pulmonary artery sarcoma with cancer pulmonary embolism. J Kor Soc Echo. 2003. 11:124–127.
35. Grifoni S, Olivotto I, Cecchini P, Pieralli F, Camaiti A, Santoro G, Pieri A, Toccafondi S, Magazzini S, Berni G, Agnelli G. Utility of an integrated clinical, echocardiographic, and venous ultrasonographic approach for triage of patients with suspected pulmonary embolism. Am J Cardiol. 1998. 82:1230–1235.

36. Jackson RE, Rudoni RR, Hauser AM, Pascual RG, Hussey ME. Prospective evaluation of two-dimensional transthoracic echocardiography in emergency department patients with suspected pulmonary embolism. Acad Emerg Med. 2000. 7:994–998.

37. Stawicki SP, Seamon MJ, Kim PK, Meredith DM, Chovanes J, Schwab CW, Gracias VH. Transthoracic echocardiography for pulmonary embolism in the ICU: finding the "right" findings. J Am Coll Surg. 2008. 206:42–47.

38. Arcasoy SM, Vachani A. Local and systemic thrombolytic therapy for acute venous thromboembolism. Clin Chest Med. 2003. 24:73–91.

39. Yip G, Abraham T, Belohlavek M, Khandheria BK. Clinical applications of strain rate imaging. J Am Soc Echocardiogr. 2003. 16:1334–1342.

40. Sutherland GR, Di Salvo G, Claus P, D'Hooge J, Bijnens B. Strain and strain rate imaging: a new clinical approach to quantifying regional myocardial function. J Am Soc Echocardiogr. 2004. 17:788–802.

41. Kjaergaard J, Sogaard P, Hassager C. Right ventricular strain in pulmonary embolism by Doppler tissue echocardiography. J Am Soc Echocardiogr. 2004. 17:1210–1212.

42. Park JH, Park YS, Park SJ, Lee JH, Choi SW, Jeong JO, Seong IW. Midventricular peak systolic strain and Tei index of the right ventricle correlated with decreased right ventricular systolic function in patients with acute pulmonary thromboembolism. Int J Cardiol. 2008. 125:319–324.

43. Hsiao SH, Chang SM, Lee CY, Yang SH, Lin SK, Chiou KR. Usefulness of tissue Doppler parameters for identifying pulmonary embolism in patients with signs of pulmonary hypertension. Am J Cardiol. 2006. 98:685–690.

44. Leibowitz D. Role of echocardiography in the diagnosis and treatment of acute pulmonary thromboembolism. J Am Soc Echocardiogr. 2001. 14:921–926.

45. Rosenberger P, Shernan SK, Body SC, Eltzschig HK. Utility of intraoperative transesophageal echocardiography for diagnosis of pulmonary embolism. Anesth Analg. 2004. 99:12–16.

46. Dalen JE, Alpert JS, Hirsh J. Thrombolytic therapy for pulmonary embolism: is it effective? Is it safe? When is it indicated? Arch Intern Med. 1997. 157:2550–2556.

47. Toosi MS, Merlino JD, Leeper KV. Prognostic value of the shock index along with transthoracic echocardiography in risk stratification of patients with acute pulmonary embolism. Am J Cardiol. 2008. 101:700–705.

48. Grifoni S, Olivotto I, Cecchini P, Pieralli F, Camaiti A, Santoro G, Conti A, Agnelli G, Berni G. Short-term clinical outcome of patients with acute pulmonary embolism, normal blood pressure, and echocardiographic right ventricular dysfunction. Circulation. 2000. 101:2817–2822.

49. Goldhaber SZ, Visani L, De Rosa M. Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER). Lancet. 1999. 353:1386–1389.

50. Binder L, Pieske B, Olschewski M, Geibel A, Klostermann B, Reiner C, Konstantinides S. N-terminal pro-brain natriuretic peptide or troponin testing followed by echocardiography for risk stratification of acute pulmonary embolism. Circulation. 2005. 112:1573–1579.

51. Scridon T, Scridon C, Skali H, Alvarez A, Goldhaber SZ, Solomon SD. Prognostic significance of troponin elevation and right ventricular enlargement in acute pulmonary embolism. Am J Cardiol. 2005. 96:303–305.

52. Goldhaber SZ. Echocardiography in the management of pulmonary embolism. Ann Intern Med. 2002. 136:691–700.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download