Abstract
Primary cardiac angiosarcoma is a rare and rapidly progressing tumor characterized by high predilection for right-sided heart involvement and a very poor prognosis. In this report, we present a case of angiosarcoma involving right atrium and ventricle in 61-year old woman with clinical presentation of repeated bloody pericardial effusion. Right atrial perforation was confirmed by contrast echocardiography using hand-agitated saline contrast. Primary cardiac angiosarcoma was confirmed by histological and immunohistochemical studies.
Primary cardiac tumors are rare, and approximately 25% of which are malignant.1) Primary cardiac angiosarcoma, which is a rapidly progressing tumor, is the most common malignant tumor of cardiac origin. It is associated with a poor prognosis and median survival time of 3 to 6 months.2)3) We report a case of 61-year-old woman who had a history of unknown cause of pericardial effusion 7 months ago. Cardiac angiosarcoma with right atrial perforation was demonstrated by two-dimensional and contrast echocardiography.
A 61-year-old woman was admitted to our hospital complaining of progressive exertional dyspnea developed two months ago. A chest X-ray showed moderate cardiomegaly. An electrocardiogram showed normal sinus rhythm with a heart rate of 76/min. Transthoracic echocardiography (TTE) showed massive pericardial effusion with swinging heart and hemodynamic significance. Computed tomography (CT) of chest revealed a large amount of pericardial effusion without any evidence of abnormal mass in chest structure including heart. We conducted pericardial window operation with pericardial biopsy using video-associated thoracoscopic surgery (VATS) in order to find the causality and relieve the symptom. Operative finding showed a thickened but relatively normal looking of pericardium, and then the 800 cc of dark brown colored pericardial effusion was drained. White cell count of the drained fluid was 3,800/mm3 (neutrophils 5%, lymphocytes 57%). The drained fluid was negative for malignant cells and bacterial cultures including culture for acid fast bacilli. Pericardial biopsy showed fibrosis with chronic inflammation and there was no evidence of granuloma or malignant cell infiltration. Positron-emission tomography (PET) showed no focal hypermetabolic lesion in whole body. Although the cause of pericardial effusion remained unknown at that time, the patient felt relief of symptom after operation, and she was discharged and regular follow-up was recommended.
Seven months after the discharge, dyspnea redeveloped. Chest X-ray showed a right pleural effusion, with the liquid level at the fifth rib. The pleural effusion was bloody and 800 cc of pleural fluid was removed at thoracentesis. Red blood cell count of the drained fluid was 2,670,000/mm3 and white cell count of the drained fluid was 1,400/mm3 (neutrophils 22%, lymphocytes 10%).
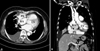
Two-dimensional echocardiography revealed the massive pericardial effusion over the right heart and a redundant motion of right atrial free wall. There were multiple mobile right ventricular masses below tricuspid valve which involved tricuspid valve. The subcostal view showed a suspicious 8-mm tissue defect in the right atrial free wall. Contrast echocardiography using hand-agitated saline revealed microbubble passage from right atrium to pericardial effusion through the defect (Fig. 1). Chest CT revealed multiple low density nodules around loculated hemopericardium in right pericardial space and suspicious of direct communication from right atrium to hemopericardium with multiple metastatic nodules in lung and liver (Fig. 2).
Emergency operation was undertaken. Through a median sternotomy, hemopericardium with severe adhesion was detected. There were multiple masses along the right atrium, right ventricle and pericardium. Histologic examination of a frozen section was sarcoma. Atrial perforation was 2×1 cm sized and the atrium was repaired with a bovine pericardial patch. There was severe bleeding with massive erosion from mass and it didn't stop. Pericardial adhesiolysis was done and invased pericardium was also resected. Complete resection of tumor was impossible due to its diffuse spread. She died one day after operation due to massive bleeding.
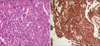
Microscopic and immunohistochemical studies confirmed cardiac angiosarcoma. The tumor cells were atypical, pleomorphic and lined the vascular channels with more than one layer. Cells were stained well by CD31 (Fig. 3).
Primary cardiac tumors are rare with an incidence ranging from 0.001 to 0.030% in autopsy cases, and mostly benign.4) In adults, approximately 75% of primary cardiac tumors are benign, with myxoma accounting for up to half of the cases. Primary cardiac sarcoma is the most common primary cardiac malignancy, and one third of those are angiosarcoma.1)5) Angiosarcoma is most commonly located in the right side of the heart, especially in the right atrium. Angiosarcomas typically occur in adults in their 30s to 50s but can occur in almost any age group from 2 to 80. These are more common in males than females.6) With the exception of a single report of familial occurrence of cardiac angiosarcomas, all others appear to occur sporadically. There is little relationship among the oncogeneses of angiosarcoma; complex cryptogenetic changes and mutation in p53 have been identified in some angiosarcomas.7)
Angiosarcoma has such features as rapid progression, local invasion and distant metastasis, with 66 to 89% of patients already demonstrating evidence of metastatic disease at initial presentation. The most frequent sites of metastases include lung, liver, brain, and bone, although metastases to lymph nodes, adrenal glands, spleen, and skin have also been reported.6)8) Initial findings may include: dyspnea, chest pain, superior vena cava syndrome, heart murmur, constitutional symptoms, arrhythmias, pericardial effusion, pericardial tamponade,9)10) non-specific ST changes, arrhythmias, and AV block on EKG. It is difficult to diagnose cardiac angiosarcoma because there is no specific symptom. Spontaneous right atrial perforation with angiosarcoma is rare. More rare is the spontaneous right atrial perforation diagnosed by two-dimensional echocardiography and contrast echocardiography. Less than 10 cases, including our own, have been reported in total.11-18)
There are several diagnostic modalities for cardiac angiosarcoma. Echocardiography is the initial imaging modality of choice, while CT and Magnetic resonance imaging (MRI) are complementary imaging for a better characterization of the tumor growth and involvement. One report demonstrated the advantage of MRI and transesophageal echocardiography (TEE) over TTE and CT for primary cardiac angiosarcoma detection.19)
Grossly, angiosarcomas typically appear as large, multilobular, dark brown intramural masses. Histologically, cardiac angiosarcomas usually exhibit evidence of endothelial differentiation with formation of vascular channels. The lining cells are atypical, pleomorphic and form regular anastomosing sinusoid structure. In poorly differentiated cases, there is little evidence of endothelial derivation and vascular channel formation. Immunohistochemical study can be used to further support evidence of endothelial differentiation by demonstrating CD31, CD34, and von Willebrand factor-positive immunophenotype in the tumor cells.20)
Median survival in cardiac angiosarcomas is 3 to 6 months.2)3) This is partially because there are no specific symptoms (most patients presented with advanced-stage diseases). Neither consensus on treatment nor treatment proven more effective exists; however, a multidisciplinary approach to the treatment is advocated.6)9)12)21) It consists of combination of treatments such as surgery, radiotherapy, adjuvant/neoadjuvant chemotherapy and immunotherapy (IL-12).22) Surgical resection is usually indicated when there is no evidence of metastasis and when myocardial resection is reparative.12) Neoadjuvant chemotherapy may be administered to enhance the rate of complete resection.23) Recently, heart transplantation has become an alternative therapy for patients with localized disease, of which outcome is not improved much.12)
In our case, she already had extracardiac metastases (lung and liver) as well as atrial perforation at the diagnosis, and the period from initial presentation to death was short (about 7 months). Moreover, there is no evidence of angiosarcoma even if she underwent TTE, CT, PET and window operation biopsy at initial presentation. There is no consensus on treatment. Nevertheless, early diagnosis and combination of treatments remain the most efficient approach. Although it is rare, cardiac angiosarcoma is a cause of unexplained pericardial effusion and has a very poor prognosis. 1) suspicion of cardiac angiosarcoma, 2) delicate work up and frequent follow up (e.g., echocardiography) for early diagnosis of cardiac angiosarcoma are therefore recommended for unexplained recurrent pericardial effusion.
Figures and Tables
Fig. 1
Two-dimensional and contrast echocardiographic findings. In right ventricle beneath the tricuspid valve, multiple nodular masses were found (A) and contrast passage from right atrium to pericardial effusion through suspicious tissue defect in right atrial free wall was documented (B and C). RA: right atrium, PE: pericardial effusion. Arrow: masses, arrowhead : contrast passage.

Fig. 2
Computed tomographic images. Multiple nodular masses were found in the pericardial space and right atrial free wall (A). A suspicious of direct communication between right atrium and pericardial effusion was demonstrated (B). LV: left ventricle, PE: pericardial effusion, RA: right atrium. Arrow: mass, arrowhead: direct communication.
References
1. Burke A, Virmani R. Tumors of the heart and the great vessels: atlas of tumor pathology. 1996. Washington D.C.: Armed Forces Institute of Pathology.
2. Bear PA, Moodie DS. Malignant primary cardiac tumors. The Cleveland Clinic experience, 1956 to 1986. Chest. 1987. 92:860–862.
3. Putnam JB Jr., Sweeney MS, Colon R, Lanza LA, Frazier OH, Cooley DA. Primary cardiac sarcomas. Ann Thorac Surg. 1991. 51:906–910.

4. Butany J, Nair V, Naseemuddin A, Nair GM, Catton C, Yau T. Cardiac tumours: diagnosis and management. Lancet Oncol. 2005. 6:219–228.

5. Best AK, Dobson RL, Ahmad AR. Best cases from the AFIP: cardiac angiosarcoma. Radiographics. 2003. 23 Spec No:S141–S145.
6. Sinatra R, Brancaccio G, di Gioia CR, De Santis M, Sbraga F, Gallo P. Integrated approach for cardiac angiosarcoma. Int J Cardiol. 2003. 88:301–304.

7. Zu Y, Perle MA, Yan Z, Liu J, Kumar A, Waisman J. Chromosomal abnormalities and p53 gene mutation in a cardiac angiosarcoma. Appl Immunohistochem Mol Morphol. 2001. 9:24–28.

8. Brandt RR, Arnold R, Bohle RM, Dill T, Hamm CW. Cardiac angiosarcoma: case report and review of the literature. Z Kardiol. 2005. 94:824–828.

9. Kim JS, Song SG, Ko WS, Park YH, Kim JH, Chun KJ, Hong TJ, Shin YW. A Case of Primary Right Atrial Angiosarcoma Manifested with Cardiac Tamponade. J Korean Soc Echocardiogr. 2004. 12:36–38.

10. Rha SW, Shim WJ, Park SM, Park SW, Lim DS, Kim YH, Ro YM. A Case of Right Atrial Sarcoma Complicated with Hemopericardium and Cardiac Tamponade. J Korean Soc Echocardiogr. 2002. 10:69–73.

11. Satou Y, Nakagawa Y, Miki H, Suzuki H, Takahashi M. Cardiac angiosarcoma with ruptured right atrium diagnosed by echocardiography. Chest. 1991. 100:274–275.

12. Sakaguchi M, Minato N, Katayama Y, Nakashima A. Cardiac angiosarcoma with right atrial perforation and cardiac tamponade. Ann Thorac Cardiovasc Surg. 2006. 12:145–148.
13. Oshima K, Ohtaki A, Kano M, Tange S, Hasegawa Y, Ikeda F, Ishikawa S, Morishita Y. Primary cardiac angiosarcoma associated with cardiac tamponade: case report. Jpn Circ J. 1999. 63:822–824.

14. Ohri SK, Nihoyannopoulos P, Taylor KM, Keogh BE. Angiosarcoma of the heart causing cardiac rupture: a rare cause of hemopericardium. Ann Thorac Surg. 1993. 55:525–528.

15. Mukohara N, Tobe S, Azami T. Angiosarcoma causing cardiac rupture. Jpn J Thorac Cardiovasc Surg. 2001. 49:516–518.

16. Lobo AJ, Butland RJ, Stewart S, Shneerson JM. Primary cardiac angiosarcoma causing rupture of the heart and spontaneous bilateral pneumothorax. Thorax. 1989. 44:78–79.

17. Lantz DA, Dougherty TH, Lucca MJ. Primary angiosarcoma of the heart causing cardiac rupture. Am Heart J. 1989. 118:186–188.

18. Corso RB, Kraychete N, Nardeli S, Moitinho R, Ourives C, Silva RM, Pereira RE. Spontaneous rupture of a right atrial angiosarcoma and cardiac tamponade. Arq Bras Cardiol. 2003. 81:611–613.

19. Inoko M, Iga K, Kyo K, Kondo H, Tamura T, Izumi C, Kitagichi S, Hirozane T, Himura Y, Gen H, Konishi T. Primary cardiac angiosarcoma detected by magnetic resonance imaging but not by computed tomography. Intern Med. 2001. 40:391–395.

20. Weiss SW, Goldblum JR. Enzinger and Weiss's Soft Tissue Tumors. 2001. 4th ed. St. Louis: Mosby-Harcourt Brace.
22. McManus B, Lee C-H. Primary tumors of the heart, in Braunwald's Heart Disease. 2008. 8th ed. Saunders;1825.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download