Abstract
This phase II study evaluated the efficacy and safety of combination chemotherapy with irinotecan, high-dose 5-fluorouracil (5-FU), and leucovorin as a second line of treatment in metastatic gastric cancer refractory to taxane and cisplatin-based chemotherapy. Eligible criteria were as follows: histologically confirmed adenocarcinoma of the stomach previously treated with taxane and cisplatin, age≥18 years, Eastern Clinical Oncology Group (ECOG) performance status of 1 or less, and adequate organ function. Irinotecan (150 mg/m2) and leucovorin (200 mg/m2) were given on day 1, followed by a 400-mg/m2 bolus infusion of 5-FU and then 2,400 mg/m2 5-FU as a 48-hour continuous infusion. This cycle was repeated every 2 weeks until disease progression or unacceptable toxicities. Thirty-seven patients were enrolled. The patients' median age was 55 years (range, 27~73 years), and the ECOG performance status of all patients was 1. All patients were evaluable for safety and survival, and 29 patients (78.3%) were evaluable for tumor response. The overall response rate was 17.2% [95% confidence interval (CI): 3.5~30.9%]. The median progression-free survival and overall survival were 4.3 months (95% CI: 2.1~6.6) and 10.6 months (95% CI: 7.1~14.1). Greater than grade 3 hematological toxicities were neutropenia in 9 (24.3%), febrile neutropenia in 1 (2.7%), and thrombocytopenia in 1 (2.7%) patient. The major nonhematological toxicity was asthenia, but most patients showed grade 1 or 2. Greater than grade 3 nonhematological toxicities were elevated AST/ALT in 4 (10.8%), hyperbilirubinemia in 2 (5.4%), and nausea in 2 (5.4%) patients. This study showed that combination chemotherapy with irinotecan, high dose 5-FU, and leucovorin is well tolerated and active in patients with cancer refractory to taxane and cisplatin.
Although the incidence and mortality of gastric cancer have decreased worldwide, gastric cancer remains the second leading cause of cancer mortality behind lung cancer.1 Early detection and radical surgery have improved treatment outcomes of localized gastric cancer. In cases of unresectable advanced or recurrent gastric cancer, systemic chemotherapy has been recommended as a standard treatment, and its prolongation of survival or increase in the quality of life is well established.2
During the past decade, new chemotherapeutic agents, including taxanes (paclitaxel, docetaxel), irinotecan, oxaliplatin, and oral 5-fluouracil (5-FU), have been intensively investigated throughout the world. In comparison with old chemotherapeutic agents, including cisplatin, 5-FU, doxorubicin, and mitomycin C, prolongation of survival was observed in several clinical trials with new chemotherapeutic agents.3,4 However, only the triple combination chemotherapy with docetaxel, cisplatin, and 5-FU (DCF) regimen showed significant prolongation of survival in comparison with traditional combination chemotherapy with cisplatin and 5-FU (CF).5 Nevertheless, in the clinical setting, the DCF regimen has not been accepted globally as a standard therapy because of its substantial toxicity and small survival advantage. Recently, other combination chemotherapies with new chemotherapeutic agents showed improvement of survival and toxicity, but median progression-free survival (PFS) and overall survival (OS) were limited to within 7 and 13 months, respectively.6 Taking all these considerations into account, there is an urgent need for second-line chemotherapy to prolong survival in metastatic colorectal cancer. For second-line chemotherapy, phase II studies have shown an overall response rate (RR) of 4~32.4% and a median PFS and OS of 2.2~4.5 and 3.5~10.7 months, respectively.7-11 This means that second-line chemotherapy may prolong survival in advanced gastric cancer patients in good general condition after first-line chemotherapy. However, until now, there has been no standard second-line chemotherapy, although various combination regimens have been tried.12
Irinotecan is a water-soluble, semisynthetic analogue of camptothecin that has been shown to exhibit anti-tumor activity against gastric cancer.13,14 The combination of irinotecan and 5-FU/leucovorin (LV) was shown to be superior to irinotecan alone or in combination with cisplatin.15,16 Also, the combination of irinotecan with continuous 5-FU infusion/LV showed superior survival and response rates compared with the combination of cisplatin and continuous 5-FU infusion/LV or the combination of irinotecan and bolus 5-FU infusion/LV for chemotherapy-naive metastatic gastric cancer.17 Therefore, irinotecan has been widely used in metastatic gastric cancer. Variable biweekly regimens of irinotecan have been used; the most common difference in those regimens was the dose of irinotecan, such as 150 mg to 180 mg, and 5-FU, usually as a 400-mg bolus infusion followed by a 600-mg continuous infusion for 2 days. Actually, pretreated gastric cancer has been shown to more chemoresistant than treatment-naïve gastric cancer. To overcome this point, another option is augmentation of the chemotherapeutic agents if this is tolerated by the patient.
Therefore, this study was conducted to evaluate the efficacy and safety of combination chemotherapy with irinotecan, high-dose infusional 5-FU, and LV in gastric cancer refractory to taxane and cisplatin-based chemotherapy as the first line of treatment.
Patients had histologically confirmed metastatic or recurrent gastric adenocarcinoma. Eligibility criteria were as follows: (i) age ≥18 years; (ii) Eastern Cooperative Oncology Group (ECOG) performance scale ≤1; (iii) a life expectancy ≥ 3 months; (iv) evaluable disease with or without measurable lesions; (v) disease progression to prior palliative chemotherapy with taxane and cisplatin; (vi) adequate baseline hematologic function [absolute neutrophil count (ANC) ≥ 1,500/mm3, platelets ≥ 100,000/mm3], hepatic function [serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) ≤ 2.5 times the ULN (upper limit of normal) and serum bilirubin ≤ 2 ULN], and renal function (serum creatinine ≤ 2 ULN); (vii) no concomitant uncontrolled medical illness; and (viii) no history or evidence of brain or meningeal metastases. All patients provided written informed consent according to the guideline provided by the institutional review board.
Patients received irinotecan (150 mg/m2) infusion simultaneously to leucovorin (200 mg/m2) as a 2-h infusion on day 1, followed by a 400-mg/m2 bolus infusion of 5-FU and then 2,400 mg/m2 5-FU as a 48-hour continuous infusion for 46 hours. This cycle was repeated every 2 weeks. Atropine 0.25 mg was given subcutaneously to prevent cholinergic syndrome before irinotecan infusion. Dexamethasone 20 mg and a 5-hydroxytryptamine type 3 receptor antagonist were given as antiemetic prophylaxis before chemotherapy. Patients were advised to commence loperamide with the onset of diarrhea (4 mg after the first loose stool and then 2 mg every 2 hours up to 12 hours after the last loose stool). If the diarrhea persisted for more than 24 hours despite appropriate loperamide therapy, a 5-day prophylactic oral fluoroquinolone treatment was initiated. Treatment was continued in the absence of disease progression, unacceptable toxicity, patients' refusal, or physicians' decision against further treatment.
The subsequent cycle of chemotherapy was withheld if ANC <1,500/mm3 or platelets <100,000/mm3 was noted on the due day. Patients presenting with grade 3 or 4 hematologic toxicity restarted the treatment with a 25% dose reduction of all drugs. During the study, granulocyte colony-stimulating factor was allowed in patients with grade 4 neutropenia or febrile neutropenia. In patients developing grade 2 diarrhea, treatment was restarted with a 25% dose reduction of irinotecan; in cases of grade 3 or 4 diarrhea, all drugs were reduced by 50%. Patients developing grade 2 mucositis received a 25% dose reduction of leucovorin and 5-FU; in case of grade 3 or 4 mucositis, leucovorin and 5-FU were restarted at a 50% dose reduction and irinotecan at 75%. If drug administration was delayed more than 2 weeks, the patient was excluded from the study.
Baseline evaluations included a full medical history, physical examination, ECOG performance status, CBC, serum chemistries, electrolytes, urinalysis, chest X-ray, electrocardiogram, and computed tomography (CT) scans. Baseline evaluations were completed within 3 weeks before treatment. For females with child-bearing potential, a negative serum pregnancy test was also required. Gastroduodenoscopy and positron emission tomography were planned for the evaluation of complete responders of all evaluable lesions.
History taking, physical examination, CBC, electrolytes, serum chemistry, urinalysis, and chest X-ray were repeated before the initiation of each subsequent cycle. CT scans for evaluating response to treatment were performed to every six cycles or when disease progression was suspected clinically. Treatment response was evaluated according to the guidelines of the Response Evaluation Criteria in Solid Tumors Committee.18 The definitions of response were as follows: (i) complete response (CR), the disappearance of all target lesions; (ii) partial response (PR), at least a 30% decrease in the sum of the longest diameters of target lesions, taking as a reference the baseline sum of the longest diameters; (iii) progressive disease (PD), at least a 20% increase in the sum of the longest diameters of targeted lesions, taking as a reference the smallest sum of the longest diameters since the treatment started or the appearance of one or more new lesions; and (iv) [jen1]stable disease (SD), neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD. Patients were considered to be evaluable for response when they had clinical evidence of disease progression. All toxicity was graded according to the National Cancer Institute Common Toxicity Criteria (version 3.0.). The dose intensity (DI) was calculated as the ratio of the total dose per square meter of the patient, divided by the total treatment duration (mg/m2/week). In this calculation, the end of treatment was considered to be 14 days after day 1 of the last cycle of chemotherapy. The relative DI was calculated as the ratio of the DI actually delivered to the DI planned by the protocol.
This study was conducted by using a Simon two-stage design. A sample size of 30 was required to accept the hypothesis that the true RR was greater than 25% with 85% power, and to reject the hypothesis that the RR was less than 5% with 5% significance. Initially, we planned to enroll 9 patients in the first stage. If one or more responses were observed, we planned to continue the second stage for a total of 30 evaluable patients. If we assume the dropout rate to be 10%, the total number of enrolled patients will be 33. All enrolled patients were included in the intent-to-treat analysis. The primary endpoint was the RR and the secondary endpoints were PFS and OS. The duration of response was calculated from the date of response confirmation to the date when disease progression was first observed. PFS was calculated from the first day of treatment to the date on which progressive disease was first observed. OS was calculated from the first day of treatment to the date of death or last follow-up. The Kaplan- Meier method was used in all survival analysis. The Statistics Package for the Social Sciences (SPSS) program for Windows (SPSS Inc., Chicago, IL, version 16.0) was applied for analysis.
Thirty-seven patients were enrolled. Patient characteristics are summarized in Table 1. The median age was 55 years, with a range of 27 to 73 years. Thirty-one patients were male (83.8%) and 6 were female (16.2%). The ECOG performance status of all patients was one. All patients had histologically confirmed adenocarcinoma. Twenty patients (54.1%) had prior gastrectomy. Fifteen patients (40.5%) had recurrent disease after undergoing an operation and 22 patients (59.5%) had no prior operation because of initially distant metastasis. The most common metastatic site was the abdominal lymph nodes, followed by the peritoneum and liver.
The median number of treatment cycles was 6, with a range from 1 to 19 cycles. Twenty patients received more than 6 cycles (54.1%). The median duration of treatment for all patients was 3.1 months, with a range of 0.5 to 12.8 months. The total number of cycles delivered was 242. A total of 22 (9.1%) cycles were delayed in 11 patients due to asthenia, infection, mucositis, neutropenia, or by patients' preference. Seventeen patients (56 cycles) were subjected to dose reduction due to asthenia, old age, creatinine elevation, liver enzyme elevation, mucositis, febrile neutropenia, and nausea. The target doses for irinotecan, 5-FU, and LV were 75, 1400, and 100 mg/m2/week, respectively, and the achieved doses for irinotecan, 5-FU, and LV were 61.2, 1151.3, and 100 mg/m2/week, respectively. Therefore, the relative DIs of irinotecan and 5-FU were 81.6% and 82.2%, respectively.
Among enrolled patients, 29 were evaluable for treatment response. Response could not be evaluated in 8 patients because of poor general condition (n=5), loss of follow-up (n=2), or in course of chemotherapy (n=1). Among 29 evaluable patients, 1 (3.4%) showed CR and 4 (13.8%) showed PR, resulting in an ORR of 17.2% (95% CI, 3.5~30.9). Eight patients (27.6%) showed SD, resulting in a disease control rate (DCR) of 44.8% (95% CI, 26.7~62.9) (Table 2). The median duration of response was 4.8 months (range, 3.0~6.0). Differences in response rates were not observed according to the patients' baseline characteristics (age, sex, involved organs, disease status, and previous chemotherapy regimen). Of 25 patients with measurable disease, 1 showed CR and 4 showed PR, giving an overall response rate of 20% in the intention-to-treat analysis. Of 4 patients with non-measurable disease, 2 patients showed SD and 2 patients showed PD.
Of the 37 patients who were evaluated for survival outcome, 19 (51.4%) were alive at the time of analysis, and the median duration of follow-up was 6.8 months (95% CI, 3.9~9.6). The median PFS and OS were 4.3 (95% CI: 2.1~6.6) and 10.6 (95% CI, 7.1~14.1) months, respectively (Fig. 1). Survival was also calculated from the first day of first-line chemotherapy (diagnostic date of metastatic gastric cancer or recurred gastric cancer) to the date of death or last follow-up. The median survival was 18.3 months (95% CI, 13.0~23.6).
All patients were evaluable for safety. Chemotherapy-related toxicities are summarized in Table 3. The main hematological toxicity was neutropenia. Greater than grade 3 neutropenia developed in 9 patients (24.3%). Febrile neutropenia developed in 1 patient (2.7%) and the patient recovered after treatment. Greater than grade 3 thrombocytopenia developed in 1 patient (2.7%). The major nonhematological toxicity was asthenia, which spontaneously resolved after 1 to 2 weeks of rest. Greater than grade 3 nonhematological toxicities were elevated AST/ALT in 4 (10.8%), hyper-bilirubinemia in 2 (5.4%), and nausea in 2 (5.4%) patients. No treatment-related mortality occurred.
Taxane and cisplatin have been used as first-line chemotherapy with various schedules and doses since the late 1990s. Taxane and cisplatin chemotherapy has produced a response rate of 30~50% as a first-line treatment. The median PFS and OS were limited to within 5~6 and 10 months, respectively. Therefore, responding patients eventually experience progressive disease.19-22 Recently, the role of second-line chemotherapy in advanced gastric cancer has gained interest, because of improvement of survival in patients in good general condition after first-line chemotherapy. However, a standard second-line chemotherapy has not been established yet and this aim will be continued into prospective trials.
As mentioned above, irinotecan has been shown to exhibit anti-tumor activity against gastric cancer, and the best combination drugs were 5-FU and LV.15~17 In cases previously treated with taxane and cisplatin in particular, irinotecan lacks cross-resistance or similar toxicity with these agents. This point is a merit of irinotecan as second-line chemotherapy in metastatic gastric cancer refractory to taxane and cisplatin-based chemotherapy. In a phase II study for patients with taxane and cisplatin-refractory metastatic gastric cancer, the combination of irinotecan, 5-FU, and LV showed moderate activity with an ORR of 21%.11 The median PFS and OS were 2.5 months and 7.6 months, respectively. Therefore, survival was not satisfactory in comparison with the results obtained with other second-line chemotherapies.
Combination chemotherapy with irinotecan and 5-FU/LV is currently the standard first-line chemotherapy for metastatic colorectal cancer. Despite higher doses of 5-FU/LV than that used in gastric cancer, it showed the tolerable toxicity as well as excellent efficacy.23,24 Therefore, we applied high-dose 5-FU/LV as in metastatic colorectal cancer to metastatic gastric cancer. Compared with the previously used dosage11, the doses of 5-FU and LV in our study were increased by 16.6% and 50%, respectively. Recently, a randomized phase II study was performed to compare efficacy according to the doses of irinotecan and 5-FU/LV in metastatic gastric cancer previously treated with platinum, taxane, and fluoropyrimidine.25 That study showed no difference in survival between low-dose and high-dose 5-FU/LV chemotherapy.
Following this study, the response rate was similar to a previous trial, but the duration of response, median PFS, and OS were prolonged. After PD on this regimen, 16 patients (43.2%) were subsequently treated to third-line chemotherapy: oxaliplatin based chemotherapy (n=12) and oral 5-FU agents (n=4). The prolongation of median OS in this study is most likely the effect of third-line chemotherapy and this point suggested some benefit in subsequent chemotherapy in selected patients. In terms of toxicity, this regimen was fairly well tolerated.
In conclusion, based on comparable activity and a favorable safety profile to other second-line chemotherapies, combination chemotherapy with irinotecan, 5-FU, and LV may be considered as second-line chemotherapy in metastatic gastric cancer refractory to taxane and cisplatin-based chemotherapy.
Figures and Tables
Fig. 1
Survival probability of the 37 patients. The overall survival (OS, A) and progression-free survival (PFS, B) in the 37 patients were 10.6 months and 4.3 months, respectively.
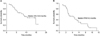
References
1. Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005. 55:74–108.

2. Glimelius B, Ekstrom K, Hoffman K, Graf W, Sjoden PO, Haglund U, et al. Randomized comparison between chemotherapy plus best supportive care with best supportive care in advanced gastric cancer. Ann Oncol. 1997. 8:163–168.

3. Cunningham D, Starling N, Rao S, Iveson T, Nicolson M, Coxon F, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med. 2008. 358:36–46.

4. Koizumi W, Narahara H, Hara T, Takagane A, Akiya T, Takagi M, et al. S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol. 2008. 9:215–221.

5. Van Cutsem E, Moiseyenko VM, Tjulandin S, Majlis A, Constenla M, Boni C, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 study group. J Clin Oncol. 2006. 24:4991–4997.

6. Ohtsu A. Chemotherapy for metastatic gastric cancer: past, present, and future. J Gastroenterol. 2008. 43:256–264.

7. Jeong J, Jeung HC, Rha SY, Im CK, Shin SJ, Ahn JB, et al. Phase II study of combination chemotherapy of 5-fluorouracil, low-dose leucovorin, and oxaliplatin (FLOX regimen) in pretreated advanced gastric cancer. Ann Oncol. 2008. 19:1135–1140.

8. Rosati G, Bilancia D, Germano D, Dinota A, Romano R, Reggiardo G, et al. Reduced dose intensity of docetaxel plus capecitabine as second-line palliative chemotherapy in patients with metastatic gastric cancer: a phase II study. Ann Oncol. 2007. 18:Suppl 6. vi128–vi132.

9. Takiuchi H, Goto M, Imamura H, Furukawa H, Imano M, Imamoto H, et al. Multi-center phase II study for combination therapy with paclitaxel/doxifluridine to treat advanced/recurrent gastric cancer showing resistance to S-1 (OGSG 0302). Jpn J Clin Oncol. 2008. 38:176–181.

10. Jo JC, Lee JL, Ryu MH, Sym SJ, Lee SS, Chang HM, et al. Docetaxel monotherapy as a second-line treatment after failure of fluoropyrimidine and platinum in advanced gastric cancer: experience of 154 patients with prognostic factor analysis. Jpn J Clin Oncol. 2007. 37:936–941.

11. Kim ST, Kang WK, Kang JH, Park KW, Lee J, Lee SH, et al. Salvage chemotherapy with irinotecan, 5-fluorouracil and leucovorin for taxane- and cisplatin-refractory, metastatic gastric cancer. Br J Cancer. 2005. 92:1850–1854.

12. Wilson D, Hiller L, Geh JI. Review of second-line chemotherapy for advanced gastric adenocarcinoma. Clin Oncol (R Coll Radiol). 2005. 17:81–90.

13. Kohne CH, Catane R, Klein B, Ducreux M, Thuss-Patience P, Niederle N, et al. Irinotecan is active in chemonaive patients with metastatic gastric cancer: a phase II multicentric trial. Br J Cancer. 2003. 89:997–1001.

14. Ajani JA, Baker J, Pisters PW, Ho L, Mansfield PF, Feig BW, et al. CPT-11 plus cisplatin in patients with advanced, untreated gastric or gastroesophageal junction carcinoma: results of a phase II study. Cancer. 2002. 94:641–646.

15. Blanke CD, Haller DG, Benson AB, Rothenberg ML, Berlin J, Mori M, et al. A phase II study of irinotecan with 5-fluorouracil and leucovorin in patients with previously untreated gastric adenocarcinoma. Ann Oncol. 2001. 12:1575–1580.

16. Pozzo C, Barone C, Szanto J, Padi E, Peschel C, Bukki J, et al. Irinotecan in combination with 5-fluorouracil and folinic acid or with cisplatin in patients with advanced gastric or esophageal-gastric junction adenocarcinoma: results of a randomized phase II study. Ann Oncol. 2004. 15:1773–1781.

17. Bouche O, Raoul JL, Bonnetain F, Giovannini M, Etienne PL, Lledo G, et al. Randomized multicenter phase II trial of a biweekly regimen of fluorouracil and leucovorin (LV5FU2), LV5FU2 plus cisplatin, or LV5FU2 plus irinotecan in patients with previously untreated metastatic gastric cancer: a Federation Francophone de Cancerologie Digestive Group Study--FFCD 9803. J Clin Oncol. 2004. 22:4319–4328.
18. Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States National Cancer Institute of Canada. J Natl Cancer Inst. 2000. 92:205–216.
19. Roth AD, Maibach R, Martinelli G, Fazio N, Aapro MS, Pagani O, et al. Docetaxel (Taxotere)-cisplatin (TC): an effective drug combination in gastric carcinoma. Swiss Group for Clinical Cancer Research (SAKK), and the European Institute of Oncology (EIO). Ann Oncol. 2000. 11:301–306.

20. Ridwelski K, Gebauer T, Fahlke J, Kroning H, Kettner E, Meyer F, et al. Combination chemotherapy with docetaxel and cisplatin for locally advanced and metastatic gastric cancer. Ann Oncol. 2001. 12:47–51.

21. Kornek GV, Raderer M, Schull B, Fiebiger W, Gedlicka C, Lenauer A, et al. Effective combination chemotherapy with paclitaxel and cisplatin with or without human granulocyte colony-stimulating factor and/or erythropoietin in patients with advanced gastric cancer. Br J Cancer. 2002. 86:1858–1863.

22. Roth AD, Maibach R, Fazio N, Sessa C, Stupp R, Morant R, et al. 5-Fluorouracil as protracted continuous intravenous infusion can be added to full-dose docetaxel (Taxotere)-cisplatin in advanced gastric carcinoma: a phase I-II trial. Ann Oncol. 2004. 15:759–764.

23. Douillard JY, Cunningham D, Roth AD, Navarro M, James RD, Karasek P, et al. Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet. 2000. 355:1041–1047.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download