Abstract
Cardiovascular disease is one of the most frequent causes of death in both males and females throughout the world. However, women exhibit a greater symptom burden, more functional disability, and a higher prevalence of nonobstructive coronary artery disease (CAD) compared to men when evaluated for signs and symptoms of myocardial ischemia. This paradoxical sex difference appears to be linked to a sex-specific pathophysiology of myocardial ischemia including coronary microvascular dysfunction, a component of the 'Yentl Syndrome'. Accordingly, the term ischemic heart disease (IHD) is more appropriate for a discussion specific to women rather than CAD or coronary heart disease. Following the National Heart, Lung, and Blood Institute Heart Truth/American Heart Association, Women's Ischemia Syndrome Evaluation and guideline campaigns, the cardiovascular mortality in women has been decreased, although significant gender gaps in clinical outcomes still exist. Women less likely undergo testing, yet guidelines indicate that symptomatic women at intermediate to high IHD risk should have further test (e.g. exercise treadmill test or stress imaging) for myocardial ischemia and prognosis. Further, women have suboptimal use of evidence-based guideline therapies compared with men with and without obstructive CAD. Anti-anginal and anti-atherosclerotic strategies are effective for symptom and ischemia management in women with evidence of ischemia and nonobstructive CAD, although more female-specific study is needed. IHD guidelines are not "cardiac catheterization" based but related to evidence of "myocardial ischemia and angina". A simplified approach to IHD management with ABCs (aspirin, angiotensin-converting enzyme inhibitors/angiotensin-renin blockers, beta blockers, cholesterol management and statin) should be used and can help to increases adherence to guidelines.
Cardiovascular disease (CVD) is a leading cause of death in both females and males throughout the world. Over the past 30 years, considerable progress of diagnosis and treatment has been achieved in this area. Although there have been significant declines in CVD mortality for women, these reductions lag behind those seen in men. In addition, where there has been a decrease in mortality from CVD across all age groups over time in men, in the young women (age≤55 years) there has been a remarkable increase in mortality from CVD.1)2) Moreover, for many decades, the diagnosis of the underlying burden of obstructive coronary artery disease (CAD) has been the focus of diagnostic evaluation algorithms for women and men presenting with chest pain.
Published studies, including the National Heart, Lung, and Blood Institute-sponsored Coronary Artery Surgery Study and the Women's Ischemia Syndrome Evaluation (WISE) study have reported that as many as 50% of women undergoing coronary angiography for suspected IHD are found to have normal or nonobstructed epicardial coronary arteries.3)4)5)
Moreover, women exhibit a greater symptom burden, more functional disability, and a higher prevalence of no obstructive CAD compared to men when evaluated for signs and symptoms of myocardial ischemia.6)7)8)9)10) Among many clinical cohorts, paradoxical sex differences have been observed in patients with symptoms and outcomes of CAD. Women have a two-fold increase in "normal" coronary arteries in the setting of acute coronary syndrome (ACS), non ST-segment elevation and ST-segment elevation myocardial infarction (MI) (Table 1).11) Women have less anatomical obstructive CAD and relatively more preserved left ventricular function despite higher rates of myocardial ischemia and mortality compared with men, even when controlling for age.7)8)9)10)11)12) This paradoxical sex difference appears to be linked to a sex-specific pathophysiology of myocardial ischemia including coronary microvascular dysfunction (CMD), a component of the 'Yentl Syndrome'. Accordingly, the term IHD is more appropriate for a discussion specific to women rather than CAD or coronary heart disease (CHD).
In this paper, we review IHD in women regarding the recognition, diagnosis and management, particularly focusing on microvascular angina and we examine the sex-specific issues related to myocardial ischemia in women in terms of recognition and detection, diagnostic testing, as well as therapeutic management strategies for IHD.
A 40-year-old woman was hospitalized for acute shortness of breath and chest tightness. Electrocardiography (EKG) showed ST-segment elevations and she had an elevated troponin 0.48 ng/mL (normal<0.04 ng/mL). A chest computed tomographic (CT) scan showed no evidence of pulmonary embolism. A coronary computed tomographic angiographic (CCTA) showed no obstructive CAD. The patient was discharged from the hospital without specific diagnosis. Five years prior, she was diagnosed with a positive anti-nucleotide antibody (ANA) which appeared to be a borderline. She was later tested and was diagnosed with systemic lupus erythematosus with a positive ANA. She also had a history of pregnancy induced hypertension. Follow up outpatient EKG had non-significant ST changes and echocardiography showed normal left ventricular wall motion and ejection fraction. She currently takes buspirone HCl orally 10 mg b.i.d, enteric coated aspirin 81 mg daily, esomeprazole 40 mg once daily, hydroxychloroquine 100 mg b.i.d, pravastatin 40 mg at bedtime and verapamil 120 mg daily with sedative-hypnotics as needed.
What is the diagnosis for cardiac symptom? Should she have any further evaluations? Or to further evaluate the basis for her symptoms, what should we do for her as a next step? How should she be treated?
The initial step to improve the outcome of women's IHD is recognition of at-risk women. In general, the greater number of traditional cardiovascular risk factors is given, the higher CVD risk is expected in any cohort. Fig. 1 showed CVD mortality trends for males and females in United States from the years 1979 to 1997, and demonstrate increasing mortality and a new female majority8) in contrast to decreased mortality in men.
Other work indicates that the adverse IHD gender gap is the widest in relatively young women, where MI mortality is 2-fold higher in women under 50 years compared with age-matched men.7) Further, it was recognized in 1991 that women with CAD were less likely to undergo coronary angiography or percutaneous coronary intervention,13) including when admission symptoms were more prominent in women than in men.14) Unlike men, women were submitted less frequently to any diagnostic or therapeutic intervention. This alarming fact was described by Healy,15) the first woman director of National Health Institute in United States, as the 'Yentl Syndrome' named after the Jewish heroine of Isaac Singer, who was masqueraded as a boy in order to be educated in the Talmud philosophy. Healy15) concluded that when a woman has been shown to have obstructive CAD, like men, only then she gets the appropriate treatment. More recently, Johnston et al.16) identified inappropriate and inadequate cardiovascular medications and coronary angiography in women with chest pain compared with men and suggested that when women look like men (with 'male-pattern' obstructive CAD), they are more likely to be diagnosed and treated like men. We have revived the use of 'Yentl Syndrome' terminology to the paradox of adverse outcomes of women with IHD, as well as the under-diagnosis and under-treatment of women.17)
What relevance does this have to the adverse IHD gender gaps for women? While it is now recognized that there are many gender differences in IHD characteristics and outcomes, including more frequent angina diagnosis, more emergency room visits, higher MI mortality, and higher rates of heart failure in women compared with men,8)18) the etiologies contributing to these differences are less well elucidated. Specifically, not only diagnostic evaluation of women may be misleading, but also the appropriate treatment selection can be difficult.
Therefore, specific studies examining gender differences to understand these gaps and paradoxical difference are needed. Two decades ago, the WISE study was commissioned as a National Heart, Lung and Blood Institute (NHLBI)-sponsored four-center study with the aims to improve diagnostic testing for the evaluation of IHD in women, and to improve understanding of the pathophysiology of myocardial ischemia in women with and without obstructive CAD.19)
The WISE study has shown that up to half of women undergoing clinically-indicated coronary angiography have either no or nonobstructive CAD,7)8)20)21) which is associated with a 2.5% yearly risk of major adverse cardiovascular events, including death, nonfatal MI, nonfatal stroke, and congestive heart failure.22)23) Further WISE follow-up in the women with nonobstructive CAD and evidence of myocardial ischemia demonstrates a relatively poor prognosis compared with women with nonobstructive CAD and no myocardial ischemia.24) Data from the WISE and related studies implicate abnormal coronary reactivity,25) including CMD,26) and plaque erosion/distal microembolization27)28) as contributory to a female-pattern IHD pathophysiology.
Women are less likely to receive guidelines-indicated IHD therapy. Novack et al.29) demonstrated that systemic use of the 2000 European Society of Cardiology and American College of Cardiology (ACC) guidelines30) was associated with a reduction in adverse sex-related ACS mortality in women, bringing female mortality close to that of men. Notably, a persistent adverse female mortality sex gap existed, suggesting more work is needed to understand sex-specific pathophysiology to improve outcomes for women.
The symptoms and signs of ischemia with "normal" coronary angiography can be similar to typical angina, although women have a relatively higher prevalence of atypical symptoms compared to men. Women tend to present with less typical symptoms, such as fatigue (70.7%), sleep disturbance (47.8%) and shortness of breath (42.1%),31) back pain, indigestion, weakness, nausea/vomiting, dyspnea and weakness.7)32)
Data from the WISE study indicate that typical vs atypical angina does not discriminate between obstructive and nonobstructive CAD in a population of women undergoing coronary angiography.33) Therefore angina classification does not adequately stratify obstructive CAD in women and most symptomatic women over 50 years are at intermediate risk (20-50%) and should be tested for myocardial ischemia (Table 2).34)
For diagnostic testing referral, it is important to categorize symptomatic women as being at low, intermediate, and high risk for IHD. For the symptomatic woman, use of risk scores weighted for the general population of asymptomatic individuals such as the Atherosclerotic Cardiovascular Disease (ASCVD), Reynolds or Framingham risk scores is not validated or recommended. For symptomatic subjects, based on age, a woman generally does not approach intermediate risk of IHD until the fifth decade of life, whereas women in their 70s and those who are older may be categorized as high risk. Symptomatic women who are at intermediate risk should have diagnostic tests for myocardial ischemia and prognosis because characterization of their symptoms does not appear to stratify risk of obstructive CAD. The past decades provided abundant evidence to guide clinical decision making regarding diagnostic testing for suspected IHD. In 2014, the American Heart Association (AHA) updated a consensus statement regarding role of noninvasive testing in the clinical evaluation of women with suspected IHD.35)
Low-risk women are generally not candidates for further diagnostic testing. The low-intermediate–risk or intermediate-risk woman is a candidate for an exercise treadmill test if she is functionally capable and has a normal or interpretable rest electrocardiography (ECG). Women with intermediate high IHD risk with an abnormal 12-lead rest ECG with ST-segment abnormalities may be referred for stress imaging (myocardial perfusion imaging, echocardiography, or cardiac magnetic resonance imaging) or CCTA. Women at high IHD risk with stable symptoms may be referred for a stress imaging modality for functional assessment of their ischemic burden and to guide posttest, anti-ischemic therapeutic decision making. Fig. 2 provides a synopsis of this evaluation algorithm.35)
Alexander et al.36) tested the prognostic value of the Duke Treadmill Score (DTS) in 976 women and 2249 men referred for evaluation of chest pain who underwent exercise treadmill testing and subsequent diagnostic cardiac catheterization. The DTS is calculated as follows: duration of exercise in minutes - (5x the maximal ST-segment deviation during or after exercise, in millimeters) - (4x the treadmill angina index). Treadmill scores: ranged from -25 (indicating the highest risk) to +15 (indicating the lowest risk). They found that the DTS effectively stratifies women into diagnostic and prognostic risk categories.36)
Among DTS variables, exercise duration was the strongest predictive parameter.37) The WISE study evaluated the prognostic value of functional capacity in asymptomatic (n=8715) and symptomatic (n=8214) women. Shaw et al.8) synthesized data from various reports and noted that women who have generally worse functional capacity, engage less often in physical exercise programs, and have more functional decline during their menopausal years as compared to age-matched men. Women who achieve <5 METs are at increased risk of cardiac event.
A recent comparative effectiveness trial randomized 824 symptomatic women at low to intermediate risk to an index exercise treadmill test compared with stress myocardial perfusion single-photon emission computed tomography (SPECT)37) and found similar 2-year rates of major adverse cardiac outcomes (p=0.59). Therefore, imaging is not appropriate in intermediate risk women with a normal ECG who can exercise.
Adverse cardiac event rates increase with the number of inducible wall motion abnormalities during stress echocardiography and are independently predictive of death from CAD or nonfatal MI above and beyond presenting symptoms, clinical risk factors, and a patient's medical history.38) Women with multiple inducible wall motion abnormalities, especially if they encumber varying vascular territories or are in the anterior wall, are at particularly high risk, with annual event rates of 5% or higher and even as high as 10% per year for those undergoing dobutamine stress echocardiography.39)40)41)
Exercise and pharmacologic stress gated myocardial perfusion imaging with SPECT is commonly used diagnostic imaging procedure and useful for risk stratification in higher risk women.42)43)44) Recently, the prognostic value of resting/stress 82Rb Positron Emission Tomography (PET) was published from a large multicenter cohort of women and men.45) From this report, 5-year CAD mortality ranged from 0.9-13% for women with 0-≥15% abnormal myocardium at stress PET (p<0.0001), which comparable to death rates were reported for men.
Coronary artery anatomy and presence of obstructive CAD can also be assessed by noninvasive coronary CCTA and invasive CAG. Min et al.46) evaluated a consecutive cohort of 24775 patients undergoing CCTA between 2005 and 2009 without known CAD who met inclusion criteria. At a 2.3±1.1-year follow-up, the mean annualized death rate was 0.3% for patients without obstructive CAD. By comparison, for women the relative hazard for death was elevated 1.7-fold for patients with mild nonobstructive CAD (p=0.016). In addition, multivessel CAD was associated with relative risk ratios that were in the range of 3-4 fold higher for women (p<0.0001). Women were at higher prognostic risk for CAD burden (Fig. 3).35) Another recent report suggests that the overall prognostic relationship is similar for women and men based on CCTA findings of the extent and severity of obstructive CAD.47)
CMD is proposed to be a contributor to the signs and symptoms of ischemia. CMD can be diagnosed using invasive angiography techniques, and novel noninvasive techniques (such as PET, transthoracic Doppler echocardiography, and cardiac magnetic resonance imaging) are increasingly available. The relationship between major adverse outcomes and baseline coronary flow reserve (CFR) was assessed in 189 women with suspected ischemia and risk factors for atherosclerosis, who were referred for coronary angiography as part of WISE.48) The majority (81%) of the women had either normal findings at angiography or evidence of <50% stenosis; during mean 5.4 years follow-up, a CFR<2.32 was the best predictor of major adverse outcomes—defined as death, nonfatal MI, nonfatal stroke, and heart failure requiring hospitalization—with a sensitivity of 62% and specificity of 65%.48)
Women remain the majority of victims and still receive fewer therapies to prevent and treat IHD despite guidelines noting their benefit. Women have less cholesterol screening, fewer lipid-lowering therapies, less use of heparin, beta-blockers and aspirin during MI, less antiplatelet therapy for secondary prevention, fewer referrals to cardiac rehabilitation, fewer implantable cardioverter-defibrillators and heart transplants compared to men with the same recognized indications.49)50)51)52)53)54)55)
To improve IHD outcomes in women, understanding female specific IHD pattern may help due to the relatively high frequency of CMD in symptomatic women with and without obstructive CAD18)56)57) which we and others have linked with the symptoms, signs, and IHD adverse outcomes.22)48)
Under-treatment of women may in part be attributed to the lower prevalence of obstructive CAD and a lack of recognition of IHD. Of note, IHD guidelines related to ACS and angina are not "cardiac catheterization" based but indicate appropriate treatment for evidence of ACS and angina. Further, a simplified approach to IHD management helps to increase adherence to guidelines. The AHA 'Get With The Guidelines'58) and the ACC 'Guidelines Applied in Practice'29) appear to have the largest impact on narrowing therapeutic gender gaps that disadvantage women.
Application of the four evidence-based ACS medication strategies (angiotensin-converting enzyme [ACE] inhibitors/angiotensin-renin blockers [ARBs], ß-blockers, aspirin, and statins) regardless of coronary angiography preferentially saves women's lives.29) Use of this strategy in stable angina patients was associated with equivalent mortality between the sexes,16) although event rates were predictably lower in this stable lower risk population and therefore the sensitivity to detect differences may be limited compared with an ACS population.
Regarding specific treatment of CMD, therapeutic lifestyle changes as well as anti-atherosclerotic and anti-anginal medications may be beneficial, but clinical outcome trials are lacking and are needed to guide treatment. Specifically, beta-blockers are effective for improving chest pain symptoms.59) No controlled studies are available on the effects of nitrates on clinical outcomes in women. Statins and ACE inhibitors improve endothelial dysfunction60)61) and may be beneficial in patients with nonobstructive CAD.60)61)62) Benefits of statins on the coronary microcirculation have been documented in clinical studies.63) Combinations of drugs, specifically statins and ACE inhibitors, may amplify these benefits.60) The proven benefit of exercise training in this population64) suggests that mechanisms of adrenergic modulation play a role.
Evidence-based therapeutic strategy or algorithm for IHD in women can be summarized as in Table 3, using an A-B-C format to use the four evidence-based medications associated with a significant reduction in recurrent major adverse cardiac events following an ACS.65)66)67)68)69)
Novel therapies with ranolazine and ivaburadine have been evaluated in women without obstructive CAD. Particularly, ranolazine, a piperazine derivative,70) is increasingly becoming an adjunctive or first-line antianginal medication in patients with refractory angina or when traditional antianginal agents are ineffective or are contraindicated (such as in patients with a low blood pressure or heart rate).70)71)72) The mechanism of action of this drug is uncertain, but is thought to involve inhibition of the late sodium current of the cardiac action potential,73) which in turn causes homeostatic improvement in sodium and calcium concentrations. Specifically, ranolazine reduces calcium overload in ischemic myocytes.
Is there an evidence of progress in improving IHD outcomes and closing these gender gaps for women when increasing recognition of IHD and treatment with guidelines? After NHLBI Heart Truth/AHA awareness, WISE sex-specific IHD research, and ACC/AHA guideline campaigns, the US cardiovascular mortality in women has decreased by 43% (Fig. 4), although women remain with an adverse mortality gap, and ongoing work is needed.
Returning to our case, the female patient had persistent outpatient symptoms of chest pain, signs of ST-segment changes on resting ECG following a ST-segment elevation with troponin elevation but no obstructive CAD by CCTA episode. Therefore, she can be diagnosed as IHD with no obstructive CAD with prior ST-segment elevation myocardial infarction (STEMI). She is at high risk for recurrent adverse events, and stress testing with imaging can be conducted to estimate prognosis and guide management. Based on recommended guidelines, she should be treated with guidelines post-STEMI therapy, including adding or replacing calcium channel blocker with a beta-blocker and adding ACE/ARB, intensifying statin and maintaining her low dose aspirin therapy.
The recognition, diagnosis, and management of IHD in women currently is suboptimal, as women are often under-diagnosed and under-treated, contributing to the adverse mortality gap experienced by women compared to men. Women exhibit a greater symptom burden, more functional disability, and a higher prevalence of nonobstructive CAD compared to men when evaluated for signs and symptoms of myocardial ischemia. This paradoxical sex difference appears to be linked to a sex-specific pathophysiology of myocardial ischemia including CMD. Accordingly, the term IHD is more appropriate for a discussion specific to women rather than CAD or CHD. As per current guidelines, symptomatic women at intermediate risk can have further testing (e.g. exercise treadmill test or stress imaging) for myocardial ischemia and prognosis. Women diagnosed with IHD should be prescribed evidence-based guideline therapies. Anti-anginal and anti-atherosclerotic strategies may be effective for symptom and ischemia management in symptomatic women with evidence of ischemia and nonobstructive CAD, although more prospective gender specific studies for prevention and improving prognosis are required. ACS and angina guidelines are not "cardiac catheterization" based, but suggest that evidence of myocardial ischemia and angina be treated. A simplified approach to IHD management with ABC (aspirin, ACE inhibitors/ARBs, beta blockers, cholesterol management with statin) can help to increase use of and adherence to existing guidelines. Further investigation of female-specific IHD is warranted to close adverse CVD gender gaps for women.
Figures and Tables
Fig. 1
Cardiovascular disease mortality trends for males and females in United States from the years 1979 to 1997. This demonstrates increasing mortality for females as a new female majority in contrast to decreased mortality in men. This alarming fact of gender gap was recognized in 1991 as the 'Yentl Syndrome' that when a woman has been shown to have obstructive coronary artery disease, like men, only then she gets the appropriate treatment. Then National Heart, Lung and Blood Institute (NHLBI)-sponsored Women's Ischemia Syndrome Evaluation (WISE) study was commissioned and American Heart Association (AHA) began to take an action.

Fig. 2
Diagnostic evaluation algorithm for women presenting with suspected ischemic heart disease (IHD) symptoms and intermediate IHD risk and intermediate-high IHD risk. ETT: exercise treadmill testing, ADL: activities of daily living, DASI: Duke Activity Status Index, CCTA: coronary computed tomography angiography, ECG: electrocardiography, SIHD: stable ischemic heart disease, angio: angiography. Reprinted from Mieres et al.35) with permission. Copyright © 2014, American Heart Association, Inc.
Fig. 3
Relative hazard for all-cause mortality from CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: An International Multicenter Registry) in younger (aged<65 years) and older (aged≥65 years) women (n=12128) and men (n=11726). Women were at higher prognostic risk for coronary artery disease burden. 1VD indicates 1-vessel disease; 2VD, 2-vessel disease; and 3VD, 3-vessel disease. Reprinted from Mieres et al.35) with permission. Copyright © 2014, American Heart Association, Inc.
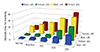
Fig. 4
Cardiovascular disease mortality trends for males and females in United States from the years 1979 to 2004. After National Heart, Lung, and Blood Institute (NHLBI) Heart Truth/American Heart Association (AHA) awareness, Women's Ischemia Syndrome Evaluation (WISE) sex-specific ischemic heart disease research, and American College of Cardiology/AHA guideline campaigns, the US cardiovascular mortality in women has decreased by 43%.

Table 1
Prevalence of "normal" and nonobstructive coronary arteries in women compared with men

| No./total (%) | p | ||
|---|---|---|---|
| Women | Men | ||
| Acute coronary syndrome | |||
| GUSTO74) | 343/1768 (19.4) | 394/4638 (8.4) | <0.001 |
| TIMI 1875) | 95/555 (17.0) | 99/1091 (9.0) | <0.001 |
| Unstable angina74) | 252/826 (30.5) | 220/1580 (13.9) | <0.001 |
| TIMI IIIa76) | 30/113 (26.5) | 27/278 (8.3) | <0.001 |
| MI without ST-segment elevation74) | 41/450 (9.1) | 55/1299 (4.2) | 0.001 |
| MI with ST-segment elevation74) | 50/492 (10.2) | 119/1759 (6.8) | 0.020 |
GUSTO: global utilization of streptokinase and t-PA for occluded coronary arteries, TIMI: thrombosis in myocardial infarction, MI: myocardial infarction. Reprinted from Bugiardini et al.11) with permission. Copyright © 2005, American Medical Association
Table 2
American College of Cardiology/American Heart Association practice guidelines on exercise testing: pretest probability of coronary artery disease by age, sex, and symptoms

Reprinted from Mieres et al.34) with permission. Copyright © 2005, American Heart Association, Inc.
Acknowledgments
This work was supported by contracts from the National Heart, Lung and Blood Institutes, nos. N01-HV-68161, N01-HV-68162, N01-HV-68163, N01-HV-68164, RO1-HL-073412-01, grants U0164829,U01 HL649141, U01 HL649241, and grants from the Gustavus and Louis Pfeiffer Research Foundation, Danville, New Jersey, The Women's Guild of Cedars-Sinai Medical Center, Los Angeles, California, The Ladies Hospital Aid Society of Western Pennsylvania, Pittsburgh, Pennsylvania, and QMED, Inc., Laurence Harbor, New Jersey, and the Edythe L. Broad Endowment, the Barbra Streisand Women's Cardiovascular Research and Education Program, the Linda Joy Pollin Women's Heart Health Program, and the Constance Austin Fellowship Endowment, Cedars-Sinai Medical Center, Los Angeles and the Erika Glazer Women's Heart Health Project, Cedars-Sinai Medical Center, Los Angeles, California.
References
1. Ford ES, Capewell S. Coronary heart disease mortality among young adults in the U.S. from 1980 through 2002: concealed leveling ofmortality rates. J Am Coll Cardiol. 2007; 50:2128–2132.
2. Dreyer RP, Wang Y, Strait KM, et al. Gender differences in the trajectory of recovery in health status among young patients with acute myocardial infarction: results from the variation in recovery: role of gender on outcomes of young AMI patients (VIRGO) study. Circulation. 2015; 131:1971–1980.
3. Proudfit WL, Shirey EK, Sones FM Jr. Selective cine coronary arteriography Correlation with clinical findings in 1,000 patients. Circulation. 1966; 33:901–910.
4. Kemp HG, Kronmal RA, Vliestra RE, Frye RL. Seven year survival of patients with normal or near normal coronary arteriograms: a CASS registry study. J Am Coll Cardiol. 1986; 7:479–483.
5. Sharaf BL, Pepine CJ, Kerensky RA, et al. Detailed angiographic analysis of women with suspected ischemic chest pain (pilot phase data from the NHLBI-sponsored Women's Ischemia Syndrome Evaluation [WISE] Study Angiographic Core Laboratory). Am J Cardiol. 2001; 87:937–941. A3
6. Moriel M, Rozanski A, Klein J, Berman DS, Merz CN. The limited efficacy of exercise radionuclide ventriculography in assessing prognosis of women with coronary artery disease. Am J Cardiol. 1995; 76:1030–1035.
7. Bairey Merz CN, Shaw LJ, Reis SE, et al. Insights from the NHLBI-Sponsored Women's Ischemia Syndrome Evaluation (WISE) Study: Part II: gender differences in presentation, diagnosis, and outcome with regard to gender-based pathophysiology of atherosclerosis and macrovascular and microvascular coronary disease. J Am Coll Cardiol. 2006; 47:3 Suppl. S21–S29.
8. Shaw LJ, Bairey Merz CN, Pepine CJ, et al. Insights from the NHLBI-Sponsored Women's Ischemia Syndrome Evaluation (WISE) Study: Part I: gender differences in traditional and novel risk factors, symptom evaluation, and gender-optimized diagnostic strategies. J Am Coll Cardiol. 2006; 47:3 Suppl. S4–S20.
9. Shaw LJ, Shaw RE, Bairey Merz CN, et al. Impact of ethnicity and gender differences on angiographic coronary artery disease prevalence and in-hospital mortality in the American College of Cardiology-National Cardiovascular Data Registry. Circulation. 2008; 117:1787–1801.
10. Glaser R, Selzer F, Jacobs AK, et al. Effect of gender on prognosis following percutaneous coronary intervention for stable angina pectoris and acute coronary syndromes. Am J Cardiol. 2006; 98:1446–1450.
11. Bugiardini R, Bairey Merz CN. Angina with "normal" coronary arteries: a changing philosophy. JAMA. 2005; 293:477–484.
12. Smilowitz NR, Sampson BA, Abrecht CR, Siegfried JS, Hochman JS, Reynolds HR. Women have less severe and extensive coronary atherosclerosis in fatal cases of ischemic heart disease: an autopsy study. Am Heart J. 2011; 161:681–688.
13. Ayanian JZ, Epstein AM. Differences in the use of procedures between women and men hospitalized for coronary heart disease. N Engl J Med. 1991; 325:221–225.
14. Steingart RM, Packer M, Hamm P, et al. Sex differences in the management of coronary artery disease. Survival and Ventricular Enlargement Investigators. N Engl J Med. 1991; 325:226–230.
15. Healy B. The Yentl syndrome. N Engl J Med. 1991; 325:274–276.
16. Johnston N, Schenck-Gustafsson K, Lagerqvist B. Are we using cardiovascular medications and coronary angiography appropriately in men and women with chest pain? Eur Heart J. 2011; 32:1331–1336.
17. Merz CN. The Yentl syndrome is alive and well. Eur Heart J. 2011; 32:1313–1315.
18. Lee DH, Jeong MH, Rhee JA, et al. Predictors of long-term survival in acute coronary syndrome patients with left ventricular dysfunction after percutaneous coronary intervention. Korean Circ J. 2012; 42:692–697.
19. Merz CN, Kelsey SF, Pepine CJ, et al. The Women's Ischemia Syndrome Evaluation (WISE) study: protocol design, methodology and feasibility report. J Am Coll Cardiol. 1999; 33:1453–1461.
20. Johnson BD, Shaw LJ, Pepine CJ, et al. Persistent chest pain predicts cardiovascular events in women without obstructive coronary artery disease: results from the NIH-NHLBI-sponsored Women's Ischaemia Syndrome Evaluation (WISE) study. Eur Heart J. 2006; 27:1408–1415.
21. Reis SE, Holubkov R, Conrad Smith AJ, et al. Coronary microvascular dysfunction is highly prevalent in women with chest pain in the absence of coronary artery disease: results from the NHLBI WISE study. Am Heart J. 2001; 141:735–741.
22. Gulati M, Cooper-DeHoff RM, McClure C, et al. Adverse cardiovascular outcomes in women with nonobstructive coronary artery disease: a report from the Women's Ischemia Syndrome Evaluation Study and the St James Women Take Heart Project. Arch Intern Med. 2009; 169:843–850.
23. Kothawade K, Bairey Merz CN. Microvascular coronary dysfunction in women: pathophysiology, diagnosis, and management. Curr Probl Cardiol. 2011; 36:291–318.
24. Sharaf B, Wood T, Shaw L, et al. Adverse outcomes among women presenting with signs and symptoms of ischemia and no obstructive coronary artery disease: findings from the National Heart, Lung, and Blood Institute-sponsored Women's Ischemia Syndrome Evaluation (WISE) angiographic core laboratory. Am Heart J. 2013; 166:134–141.
25. von Mering GO, Arant CB, Wessel TR. Abnormal coronary vasomotion as a prognostic indicator of cardiovascular events in women: results from the National Heart, Lung, and Blood Institute-Sponsored Women's Ischemia Syndrome Evaluation (WISE). Circulation. 2004; 109:722–725.
26. Wong TY, Klein R, Sharrett AR, et al. Retinal arteriolar narrowing and risk of coronary heart disease in men and women. The Atherosclerosis Risk in Communities Study. JAMA. 2002; 287:1153–1159.
27. Burke AP, Farb A, Malcolm GT, Liang Y, Smialek J, Virmani R. Effects of risk factors on the mechanism of acute thrombosis and sudden coronary death in women. Circulation. 1998; 97:2110–2116.
28. Burke AP, Virmani R, Galis Z, Haudenschild CC, Muller JE. 34th Bethesda Conference: Task force #2--What is the pathologic basis for new atherosclerosis imaging techniques? J Am Coll Cardiol. 2003; 41:1874–1886.
29. Novack V, Cutlip DE, Jotkowitz A, Lieberman N, Porath A. Reduction in sex-based mortality difference with implementation of new cardiology guidelines. Am J Med. 2008; 121:597–603.
30. Myocardial infarction redefined--a consensus document of The Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction. Eur Heart J. 2000; 21:1502–1513.
31. McSweeney JC, Cody M, O'Sullivan P, Elberson K, Moser DK, Garvin BJ. Women's early warning symptoms of acute myocardial infarction. Circulation. 2003; 108:2619–2623.
32. DeVon HA, Ryan CJ, Ochs AL, Shapiro M. Symptoms across the continuum of acute coronary syndromes: differences between women and men. Am J Crit Care. 2008; 17:14–24. quiz 25.
33. Johnson BD, Kelsey SF, Bairey Merz CN. Chapter 10. Clinical risk assessment in women: chest discomfort. Report from the WISE study. In : Shaw LJ, Redberg RF, editors. Coronary Disease in Women: Evidence-Based Diagnosis and Treatment. Totowa, NJ: Humana Press;2003. p. 129–141.
34. Mieres JH, Shaw LJ, Arai A, et al. Role of noninvasive testing in the clinical evaluation of women with suspected coronary artery disease: Consensus statement from the Cardiac Imaging Committee, Council on Clinical Cardiology, and the Cardiovascular Imaging and Intervention Committee, Council on Cardiovascular Radiology and Intervention, American Heart Association. Circulation. 2005; 111:682–696.
35. Mieres JH, Gulati M, Bairey Merz N, et al. Role of noninvasive testing in the clinical evaluation of women with suspected ischemic heart disease: a consensus statement from the American Heart Association. Circulation. 2014; 130:350–379.
36. Alexander KP, Shaw LJ, Shaw LK, Delong ER, Mark DB, Peterson ED. Value of exercise treadmill testing in women. J Am Coll Cardiol. 1998; 32:1657–1664.
37. Shaw LJ, Mieres JH, Hendel RH, et al. Comparative effectiveness of exercise electrocardiography with or without myocardial perfusion single photon emission computed tomography in women with suspected coronary artery disease: results from the What Is the Optimal Method for Ischemia Evaluation in Women (WOMEN) trial. Circulation. 2011; 124:1239–1249.
38. Mieres JH, Shaw LJ, Arai A, et al. Role of noninvasive testing in the clinical evaluation of women with suspected coronary artery disease: consensus statement from the Cardiac Imaging Committee, Council on Clinical Cardiology, and the Cardiovascular Imaging and Intervention Committee, Council on Cardiovascular Radiology and Intervention, American Heart Association. Circulation. 2005; 111:682–696.
39. Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2012; 60:e44–e164.
40. Shaw LJ, Vasey C, Sawada S, Rimmerman C, Marwick TH. Impact of gender on risk stratification by exercise and dobutamine stress echocardiography: long-term mortality in 4234 women and 6898 men. Eur Heart J. 2005; 26:447–456.
41. Arruda-Olson AM, Juracan EM, Mahoney DW, McCully RB, Roger VL, Pellikka PA. Prognostic value of exercise echocardiography in 5,798 patients: is there a gender difference. J Am Coll Cardiol. 2002; 39:625–631.
42. Shaw LJ, Hage FG, Berman DS, Hachamovitch R, Iskandrian A. Prognosis in the era of comparative effectiveness research: where is nuclear cardiology now and where should it be? J Nucl Cardiol. 2012; 19:1026–1043.
43. Gargiulo P, Petretta M, Bruzzese D, et al. Myocardial perfusion scintigraphy and echocardiography for detecting coronary artery disease in hypertensive patients: a meta-analysis. Eur J Nucl Med Mol Imaging. 2011; 38:2040–2049.
44. Bateman TM, Heller GV, McGhie AI, et al. Diagnostic accuracy of rest/stress ECG-gated Rb-82 myocardial perfusion PET: comparison with ECG-gated Tc-99m sestamibi SPECT. J Nucl Cardiol. 2006; 13:24–33.
45. Kay J, Dorbala S, Goyal A, et al. Influence of sex on risk stratification with stress myocardial perfusion Rb-82 positron emission tomography: results from the PET (Positron Emission Tomography) Prognosis Multicenter Registry. J Am Coll Cardiol. 2013; 62:1866–1876.
46. Min JK, Dunning A, Lin FY, et al. Age- and sex-related differences in all-cause mortality risk based on coronary computed tomography angiography findings results from the International Multicenter CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: An International Multicenter Registry) of 23,854 patients without known coronary artery disease. J Am Coll Cardiol. 2011; 58:849–860.
47. Lin FY, Shaw LJ, Dunning AM, et al. Mortality risk in symptomatic patients with nonobstructive coronary artery disease: a prospective 2-center study of 2,583 patients undergoing 64-detector row coronary computed tomographic angiography. J Am Coll Cardiol. 2011; 58:510–519.
48. Pepine CJ, Anderson RD, Sharaf BL, et al. Coronary microvascular reactivity to adenosine predicts adverse outcome in women evaluated for suspected ischemia results from the National Heart, Lung and Blood Institute WISE (Women's Ischemia Syndrome Evaluation) study. J Am Coll Cardiol. 2010; 55:2825–2832.
49. Chandra NC, Ziegelstein RC, Rogers WJ, et al. Observations of the treatment of women in the United States with myocardial infarction: a report from the National Registry of Myocardial Infarction-I. Arch Intern Med. 1998; 158:981–988.
50. Nohria A, Vaccarino V, Krumholz HM. Gender differences in mortality after myocardial infarction. Why women fare worse than men. Cardiol Clin. 1998; 16:45–57.
51. Scott LB, Allen JK. Providers' perceptions of factors affecting women's referral to outpatient cardiac rehabilitation programs: an exploratory study. J Cardiopulm Rehabil. 2004; 24:387–391.
52. O'Meara JG, Kardia SL, Armon JJ, Brown CA, Boerwinkle E, Turner ST. Ethnic and sex differences in the prevalence, treatment, and control of dyslipidemia among hypertensive adults in the GENOA study. Arch Intern Med. 2004; 164:1313–1318.
53. Hendrix KH, Riehle JE, Egan BM. Ethnic, gender, and age-related differences in treatment and control of dyslipidemia in hypertensive patients. Ethn Dis. 2005; 15:11–16.
54. Cho L, Hoogwerf B, Huang J, Brennan DM, Hazen SL. Gender differences in utilization of effective cardiovascular secondary prevention: a Cleveland clinic prevention database study. J Womens Health (Larchmt). 2008; 17:515–521.
55. Chou AF, Scholle SH, Weisman CS, Bierman AS, Correa-de-Araujo R, Mosca L. Gender disparities in the quality of cardiovascular disease care in private managed care plans. Womens Health Issues. 2007; 17:120–130.
56. Reis SE, Holubkov R, Lee JS, et al. Coronary flow velocity response to adenosine characterizes coronary microvascular function in women with chest pain and no obstructive coronary disease. Results from the pilot phase of the Women's Ischemia Syndrome Evaluation (WISE) study. J Am Coll Cardiol. 1999; 33:1469–1475.
57. Wessel TR, Arant CB, McGorray SP, et al. Coronary microvascular reactivity is only partially predicted by atherosclerosis risk factors or coronary artery disease in women evaluated for suspected ischemia: results from the NHLBI Women's Ischemia Syndrome Evaluation (WISE). Clin Cardiol. 2007; 30:69–74.
58. LaBresh KA, Fonarow G, Ellrodt AG, et al. Get with the guidelines improves cardiovascular care in hospitalized patients with CAD. Circulation. 2003; 108:Suppl 4. 722. (Abstract).
59. Lanza GA, Colonna G, Pasceri V, Maseri A. Atenolol versus amlodipine versus isosorbide-5-mononitrate on anginal symptoms in syndrome X. Am J Cardiol. 1999; 84:854–856. A8
60. Pizzi C, Manfrini O, Fontana F, Bugiardini R. Angiotensin-converting enzyme inhibitors and 3-hydroxy-3-methylglutaryl coenzyme A reductase in cardiac Syndrome X: role of superoxide dismutase activity. Circulation. 2004; 109:53–58.
61. Kayikcioglu M, Payzin S, Yavuzgil O, Kultursay H, Can LH, Soydan I. Benefits of statin treatment in cardiac syndrome-X1. Eur Heart J. 2003; 24:1999–2005.
62. Chen JW, Hsu NW, Wu TC, Lin SJ, Chang MS. Long-term angiotensin-converting enzyme inhibition reduces plasma asymmetric dimethylarginine and improves endothelial nitric oxide bioavailability and coronary microvascular function in patients with syndrome X. Am J Cardiol. 2002; 90:974–982.
63. Manfrini O, Pizzi C, Morgagni G, Fontana F, Bugiardini R. Effects of pravastatin on myocardial perfusion after percutaneous transluminal coronary angioplasty. Am J Cardiol. 2004; 93:1391–1393. A6
64. Eriksson BE, Tyni-Lennè R, Svedenhag J, et al. Physical training in Syndrome X: physical training counteracts deconditioning and pain in Syndrome X. J Am Coll Cardiol. 2000; 36:1619–1625.
65. Alexander KP, Chen AY, Newby LK, et al. Sex differences in major bleeding with glycoprotein IIb/IIIa inhibitors: results from the CRUSADE (Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines) initiative. Circulation. 2006; 114:1380–1387.
66. Ambrosioni E, Borghi C, Magnani B. The effect of the angiotensin-converting-enzyme inhibitor zofenopril on mortality and morbidity after anterior myocardial infarction. The Survival of Myocardial Infarction Long-Term Evaluation (SMILE) Study Investigators. N Engl J Med. 1995; 332:80–85.
67. Hjalmarson A, Elmfeldt D, Herlitz J, et al. Effect on mortality of metoprolol in acute myocardial infarction. A double-blind randomised trial. Lancet. 1981; 2:823–827.
68. Gluckman TJ, Sachdev M, Schulman SP, Blumenthal RS. A simplified approach to the management of non-ST-segment elevation acute coronary syndromes. JAMA. 2005; 293:349–357.
69. Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014; 130:e344–e426.
70. Timmis AD, Chaitman BR, Crager M. Effects of ranolazine on exercise tolerance and HbA1c in patients with chronic angina and diabetes. Eur Heart J. 2006; 27:42–48.
71. Chaitman BR, Pepine CJ, Parker JO, et al. Effects of ranolazine with atenolol, amlodipine, or diltiazem on exercise tolerance and angina frequency in patients with severe chronic angina: a randomized controlled trial. JAMA. 2004; 291:309–316.
72. Chaitman BR, Skettino SL, Parker JO, et al. Anti-ischemic effects and long-term survival during ranolazine monotherapy in patients with chronic severe angina. J Am Coll Cardiol. 2004; 43:1375–1382.
73. Belardinelli L, Shryock JC, Fraser H. Inhibition of the late sodium current as a potential cardioprotective principle: effects of the late sodium current inhibitor ranolazine. Heart. 2006; 92:Suppl 4. iv6–iv14.
74. Hochman JS, Tamis JE, Thompson TD, et al. Sex, clinical presentation, and outcome in patients with acute coronary syndromes. N Engl J Med. 1999; 341:226–232.
75. Glaser R, Herrmann HC, Murphy SA, et al. Benefit of an early invasive management strategy in women with acute coronary syndromes. JAMA. 2002; 288:3124–3129.
76. Diver DJ, Bier JD, Ferreira PE, et al. Clinical and arteriographic characterization of patients with unstable angina without critical coronary arterial narrowing (from the TIMI-IIIA Trial). Am J Cardiol. 1994; 74:531–537.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download