Abstract
Transcatheter aortic valve implantation (TAVI) has shown favorable outcomes in patients with severe symptomatic aortic valve stenosis who are at high surgical risk or are unsuitable candidates for open heart surgery. However, concerns exist over treating patients who have previously undergone mitral valve surgery due to the potential interference between the mitral prosthetic valve or ring and the TAVI device. In this case report, we present a patient with symptomatic severe aortic stenosis and previous mechanical mitral valve replacement who was successfully treated with TAVI using a CoreValve.
Transcatheter aortic valve implantation (TAVI) has become an alternative therapeutic approach leading to favorable outcomes in patients with symptomatic severe aortic valvular stenosis who are at high surgical risk or are unsuitable candidates for surgical aortic valve replacement.1)2) In general, re-operations after a previous cardiac surgery are associated with increased mortality and high risk for adverse events. Therefore, a history of previous cardiac surgery in patients with symptomatic severe aortic stenosis is generally considered an indication for TAVI. However, concerns still exist over treating patients who have previously undergone mitral valve surgery due to the proximity and the potential mechanical interference between the TAVI device and the previously implanted mitral prosthesis. There have only been a limited number of reports on the feasibility of TAVI using a CoreValve (Medtronic, Minneapolis, MI, USA) in patients with a prosthetic mitral valve. To our knowledge, this is the first case regarding this problem in Korea.
In this case report, we present a patient with symptomatic severe aortic stenosis and previous mechanical mitral valve replacement who was successfully treated with TAVI using a CoreValve.
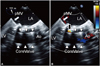
A 75-year-old female was referred to our institution due to heart failure {New York Heart Association (NYHA) class III} as a consequence of severe aortic valve stenosis, which was diagnosed at a local hospital. The patient had a medical history of mitral valve replacement with a mechanical prosthetic valve (St. Jude #29) in 1988. Other comorbidities included hypertension, bronchial asthma, and chronic otitis media. The electrocardiogram (ECG) on admission showed sinus rhythm and findings suggestive of left ventricular hypertrophy. Transthoracic and transesophageal echocardiography showed a heavily calcified aortic valve with an aortic valve area of 0.41 cm2 and peak and mean pressure gradients of 54 and 30 mm Hg, respectively (Figs. 1 and 4A). The bileaflet mechanical mitral valve was intact without any morphological abnormalities. The left ventricle showed normal systolic function and dimensions with mild concentric hypertrophy. There was mild pulmonary hypertension with a right ventricular systolic pressure of 47 mm Hg and mild tricuspidal regurgitation. Computed tomography (CT) images demonstrated a heavily calcified aortic valve with an annulus diameter ranging from 18.6 to 23.3 mm, an annulus perimeter of 68.6 mm, and an annulus area of 350 mm2. The distance between the prosthetic mitral valve and the aortic valve annulus was 4.5 mm. CT angiography demonstrated moderately tortuous iliac arteries with a diameter greater than 5.6 mm and common femoral arteries with a diameter greater than 6.4 mm on both sides. There was no significant stenosis or calcification in the iliac or common femoral artery. Coronary angiography revealed no significant stenosis. The logistic Euroscore II was calculated to be 13.2%. A multidisciplinary heart team consisting of interventional cardiologists, cardiac surgeons, anesthesiologists, and imaging specialists evaluated the case and approved TAVI for this patient.
The procedure was carried out in a hybrid operating room under general anesthesia. The vascular access for the CoreValve delivery catheter was obtained in the left common femoral artery with standard percutaneous access techniques. The "Preclose" technique using two suture-based closure devices (Abbott Vascular, Redwood City, CA, USA) was applied before insertion of the sheath in order to prepare pre-tied knots for closure of the puncture site after the procedure. A temporary pacemaker was placed in the right ventricle via the right femoral vein. A 5 Fr pigtail catheter was inserted into the ascending aorta via the right common femoral artery for angiographic guidance. A 0.032 inch wire was passed through the aortic valve using a 5 Fr AL1 catheter and exchanged with a pre-shaped 0.035 inch superstiff wire. A delivery sheath loaded with a 26 mm CoreValve was inserted through the aortic valve without predilation. The position of the CoreValve was adjusted within the aortic valve, avoiding contact with the prosthetic mitral valve, and was slowly deployed under angiographic and transesophageal echocardiography probe (TEE) guidance (Fig. 2). TEE after the valve deployment revealed a moderate degree of paravalvular leak. Therefore, postdilation using a 22 mm balloon (Maxi LD™, Cordis Corporation, Johnson & Johnson Ltd., Bridgewater, NJ, USA) was performed under rapid pacing. Subsequent TEE showed a decreased paravalvular leak (mild AR) and an adequate position of the implanted CoreValve (Fig. 3). The prosthetic mitral valve showed intact valve function. The peak and mean pressure gradients over the aortic valve as measured by echocardiography were decreased from 54 to 18 mm Hg and from 30 to 8 mm Hg, respectively (Fig. 4). The puncture sites were closed without vascular complications. The patient was monitored in an intensive care unit for two days. No conduction abnormalities were observed on ECG. The patient's symptoms subsequently improved from NYHA class III to class I. On day 8 post-procedure, the patient was discharged without any significant complications.
Patients with an estimated mortality risk >20% by logistic EuroSCORE or >10% by the Society of Thoracic Surgeons score system are generally considered candidates for the TAVI procedure. In addition, combined respiratory failure; pulmonary hypertension; previous cardiac surgery; right ventricular failure; hostile thorax caused by occurrences such as radiation, burns, previous thoracic pleurodesis, or multiple thoracotomies; severe connective tissue disease; liver cirrhosis; cachexia; or porcelain aorta are further indications for TAVI.3) Also, a history of cardiac surgery is generally associated with increased mortality (5-26%) due to postoperative adhesion and the patient's impaired general condition.4)5)
The technical concerns regarding TAVI in patients with prosthetic mitral valves are due to the potential interactions between the aortic and mitral prostheses at the anatomic aorto-mitral continuity. The significant reduction or even absence of themitroaortic space to accommodate the balloon-expanded valve and the presence of a mechanical structure instead of fibrous tissue can limit the expansion of the percutaneous valve.6) Therefore, a pre-existing prosthetic valve or ring had been considered an exclusion criterion in clinical trials such as the PARTNER II trial (Edwards Sapien valve) and the Medtronic CoreValve U.S Pivotal Trial.7)8) Several reports have since demonstrated that TAVI can be successfully performed in patients with mechanical or biological mitral valves or annuloplasty mitral rings.6)9)10)11) However, there have been a limited number of reports on TAVI using CoreValve, a self-expandable stent with a longer stent frame compared to balloon-expandable valves, in patients with a mechanical mitral valve. This is the first Korean case report on this issue.
In the present case, we avoided any displacement or deformation of the CoreValve and distortion of the preexisting mitral prosthesis by meticulous positioning of the TAVI device under TEE guidance. Also, post-procedural echocardiography revealed intact mitral valve function. Pre-procedural assessment using echocardiography and CT images are important to avoid potential interference between the aortic annulus and the mitral prosthesis. The intraprocedural imaging guidance by TEE is critical to adjust the extent of the prosthetic valve strut protrusion into the LVOT.
Our case demonstrates that TAVI using a CoreValve can be performed safely in patients with previously implanted prosthetic mitral valves.
Figures and Tables
Fig. 1
Pre-procedural transthoracic echocardiogram with a parasternal long axis view of the heavily calcified aortic valve with valve area of 0.41 cm2. A: without color Doppler. B: with color Doppler. AV: aortic valve, LA: left atrium, LV: left ventricle, RV: right ventricle, pMV: prosthetic mitral valve.

Fig. 2
Fluoroscopic images of transcatheter aortic valve implantation using a CoreValve. A: partially deployed CoreValve. B: completely deployed CoreValve. TEE: transesophageal echocardiography probe, pMV: bileaflet mechanical prosthetic mitral valve (dotted circle), W: superstiff wire inserted into the left ventricle, TP: temporary pacemaker inserted into the right ventricle.

Fig. 3
Post-procedural images of transesophageal echocardiography (135 degree long axis view). A: without color Doppler. B: with color Doppler. The implanted CoreValve (arrowheads) did not interfere with the mechanical mitral valve (white arrow). The black arrows indicate a mild degree of paravalvular leak. Ao: aorta, LA: left atrium, LV: left ventricle, pMV: prosthetic mitral valve.
References
1. Cribier A, Eltchaninoff H, Tron C, et al. Treatment of calcific aortic stenosis with the percutaneous heart valve: mid-term follow-up from the initial feasibility studies: the French experience. J Am Coll Cardiol. 2006; 47:1214–1223.
2. Grube E, Buellesfeld L, Mueller R, et al. Progress and current status of percutaneous aortic valve replacement: results of three device generations of the CoreValve Revalving system. Circ Cardiovasc Interv. 2008; 1:167–175.
3. Kim IS, Ko YG, Shin S, et al. The first case of successful transcatheter aortic valve implantation using CoreValve in Korea. Korean Circ J. 2012; 42:788–791.
4. Akins CW, Buckley MJ, Daggett WM, et al. Risk of reoperative valve replacement for failed mitral and aortic bioprostheses. Ann Thorac Surg. 1998; 65:1545–1551. discussion 1551-2.
5. Jamieson WR, Burr LH, Miyagishima RT, et al. Re-operation for bioprosthetic aortic structural failure - risk assessment. Eur J Cardiothorac Surg. 2003; 24:873–878.
6. Soon JL, Ye J, Lichtenstein SV, Wood D, Webb JG, Cheung A. Transapical transcatheter aortic valve implantation in the presence of a mitral prosthesis. J Am Coll Cardiol. 2011; 58:715–721.
7. Petronio AS, De Carlo M, Bedogni F, et al. Safety and efficacy of the subclavian approach for transcatheter aortic valve implantation with the CoreValve revalving system. Circ Cardiovasc Interv. 2010; 3:359–366.
8. Antman EM. Implications of contemporary clinical trials series: the Placement of Aortic Transcatheter Valve trial (PARTNER). Circulation. 2012; 125:3054.
9. Rodés-Cabau J, Dumont E, Miró S, et al. Apical aortic valve implantation in a patient with a mechanical valve prosthesis in mitral position. Circ Cardiovasc Interv. 2008; 1:233.
10. Bruschi G, De Marco F, Oreglia J, et al. Percutaneous implantation of CoreValve aortic prostheses in patients with a mechanical mitral valve. Ann Thorac Surg. 2009; 88:e50–e52.
11. Bruschi G, De Marco F, Barosi A, et al. Self-expandable transcatheter aortic valve implantation for aortic stenosis after mitral valve surgery. Interact Cardiovasc Thorac Surg. 2013; 17:90–95.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download