Introduction
Pilsicainide is a pure sodium channel blocker with slow recovery kinetics, and it is known to be effective in converting recent-onset atrial fibrillation to sinus rhythm.1)2) On the other hand, high plasma concentrations of class IC antiarrhythmic drugs, including pilsicainide, have been known to induce life-threatening tachyarrhythmias, such as ventricular tachycardia, torsades de pointes (TdP) and ventricular fibrillation.3)
We report a case of sudden cardiac death; the patient received only three low oral doses of pilsicainide (100 mg/day) to convert paroxysmal atrial fibrillation to sinus rhythm and developed TdP only two days later during Holter electrocardiogram (ECG) monitoring. This case may present a concern about the proarrhythmic effects of an oral pilsicainide for treatment of atrial fibrillation in octogenarian patients.
Case
An 84-year-old male visited our hospital due to palpitations and shortness of breath that began two days previously. He underwent physical examination, 12-lead ECG, chest X-ray, and blood tests. The patient's heart rate was 150 beats/min, and his blood pressure was 126/76 mm Hg. Chest X-ray revealed no cardiomegaly (cardiothoracic index=46%) and no congestion. An ECG examination showed atrial fibrillation, which was considered to be of recent onset (within 48 hours). Although ECG showed a tendency for poor progression of R in V 1-3, the patient had no chest pain and no history of coronary artery disease. In addition, an echocardiography, which was performed in a private clinic two months before the patient visited our hospital, showed normal left ventricular wall motion; during the past two months, there were no significant differences of ST-T changes in ECG. Routine laboratory tests revealed serum sodium of 142 mEq/L, potassium of 4.8 mEq/L (no hypokalemia and hyperkalemia) and creatinine of 1.0 mg/dL, which were indicative of mild renal dysfunction. Thus, oral low-dose pilsicainide at 50 mg twice daily was prescribed to convert atrial fibrillation to a sinus rhythm, and a Holter ECG was ordered in the outpatient clinic. In addition, he started anticoagulant treatment with warfarin but did not receive any other drugs, including any other antiarrhythmic drugs.
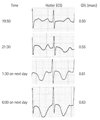
One day after receiving an oral administration of pilsicainide, he visited our hospital again, and a Holter monitor was fitted to evaluate the efficacy of pilsicainide and initiated at 13:30 in the afternoon. At this time, he felt no palpitations and showed a sinus rhythm with normal QT and QTc intervals (320 msec and 400 msec, respectively) in the CM5 lead of the Holter ECG (Fig. 1). However, the patient died during his sleep 18 hours after the Holter ECG was started. He reported no discomfort during the day or before his sudden death.
The analysis of the Holter ECG recording revealed that the cause of sudden death was an episode of TdP (Fig. 2). The Holter ECG showed that the patient developed atrial fibrillation followed by repetitive non-sustained and sustained TdP (Figs. 2 and 3). As shown in Fig. 4, the QTc interval in the Holter ECG showed gradual elongation from 13:30 at the start until 6:00 the next day. At 7:00 the next day, TdP was observed, and it continued for approximately one hour; at 8:00, cardiac arrest was detected in the Holter ECG. The electrophysiological parameters, QRS complexes, RR, and QTc interval were measured. The segments with excessive noise, ectopic activity and atrial fibrillation were excluded from the analysis. Fig. 5 illustrates the trend of the electrophysiological parameters. The QTc interval was clearly prolonged compared with that of initial Holter ECG. At the same time, the width of the QRS complexes was slightly prolonged compared with that of the initial Holter ECG.
Discussion
Pilsicainide, one of the class IC antiarrhythmic drugs, is very popular in Japan and is also available in Korea for terminating paroxysmal atrial fibrillation partially due to the favorable effects reported in the Pilsicainide Suppression Trial on Atrial Fibrillation clinical study.2) An increase in the rate of sudden death in patients taking a class IC drug after myocardial infarction was reported in the Cardiac Arrhythmia Suppression Trial study;4) however, it is unknown whether pilsicainide also increases the rate of sudden death for patients with atrial fibrillation.
Pilsicainide is rapidly absorbed from the gastrointestinal tract, and most of it is excreted from the kidney; therefore, the half-life of elimination is prolonged in patients with severe renal failure. In fact, previous studies reported arrhythmic events in patients after taking pilsicainide, and almost all these patients had renal function failure. In our case, the level of serum creatinine was in the normal range, but the patient was 84 years old, and his body weight was 65 kg. Thus, the estimated creatinine clearance was 46 mL/min, and the estimated glomerular filtration rate was 54.4 mL/min/1.73 m2, which were considered to represent mild renal dysfunction even though the serum creatinine was 1.0 mg/dL. Although the terminal electrical events leading to sudden cardiac death are rarely recorded on Holter ECG monitoring, in this case the patient happened to be wearing the Holter ECG by chance during the cardiac event. The Holter ECG monitor revealed a gradually prolonged QTc interval just before TdP, which was considered to indicate an elevated plasma concentration of pilsicainide, although the actual level was not measured.
While orally administered pilsicainide is an effective drug for terminating atrial fibrillation, the dose should be adjusted cautiously in octogenarian patients even if their serum creatinine levels are within normal ranges. In addition, when pilsicainide is prescribed for the octogenarian, the patient should be admitted to the hospital for monitoring.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download