Abstract
Clinical features of acute myocarditis range from a subclinical state to a fulminant state. Fulminant myocarditis with ventricular arrhythmia or atrioventricular block is associated with a high mortality rate. In cases in which aggressive medical therapy for fulminant myocarditis is not likely to be successful, intensive and emergency mechanical circulatory support, such as extracorporeal membrane oxygenation (ECMO) or intra-aortic balloon pump, should be considered. We report life salvage of acute fulminant myocarditis in a 53-year-old woman presented with malignant arrhythmia and cardiogenic shock supported by ECMO.
Acute myocarditis is associated with various electrocardiogram (ECG) alterations, including ST-T changes, Q-waves, atrioventricular- and intraventricular-conduction delays, atrial and ventricular tachyarrhythmia, and low voltage. Fulminant myocarditis with ventricular arrhythmia or atrioventricular block is associated with a high mortality rate. Extracorporeal membrane oxygenation (ECMO) is instituted for the management of life threatening pulmonary or cardiac failure, when no other forms of treatment are likely to be successful. We describe life salvage of ECMO in a patient with cardiogenic shock and malignant arrhythmia caused by acute fulminant myocarditis.
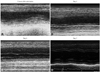
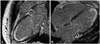
A 53-year-old Korean-Chinese woman was brought into the emergency department of our hospital by an ambulance, with complaints of progressive chest pain and dyspnea. She had a high fever, cough, and general fatigue over the three days. Upon arrival, she was fully conscious and had signs of cold sweating, cyanosis and general pallor. The initial creatine kinase (CK), CK-MB and Troponin-I levels were 626 U/L, 47.27 ng/mL, >50 ng/mL, respecitvely. Emergent coronary angiogram was performed because of low blood pressure (88/56 mm Hg), ST-segment depression in the precordial leads of ECG (Fig. 1A), and systolic dysfunction with left ventricular wall motion abnormality on echocardiography. Coronary angiographic findings were normal, and an intra-aortic balloon pump (IABP) was immediately inserted for hemodynamic support (Fig. 2). On the basis of prodromal symptoms, elevated cardiac enzymes and normal coronary angiographic findings with ventricular wall motion abnormality, we contemplated acute decompensated heart failure due to clinical suspicion acute myocarditis. She was admitted to the coronary care unit, and mechanical ventilation and inotropic agents were started. Although aggressive supportive therapies were provided, her blood pressure continued to decline. We decided to support the patient with ECMO, which was inserted in a veno-arterial configuration by cannulation on both femoral vessels. We were able to reduce the amount of inotropic agents using mechanical circulatory support. However, ECG showed wide QRS tachycardia with left bundle branch block (Fig. 1B and C), and echocardiography showed severe global hypokinesia (Fig. 3A). The serum creatinine level was elevated, and the amount of urination was decreased. Soon after, pulmonary edema developed. Because of low blood pressure and the lack of diuretic effect, continuous renal replacement therapy was started using the ECMO circuit on day 1. Ventricular wall movement was gradually decreased, and ECG showed little or no electrical activity (Figs. 1D and 3B). On day 2, IABP had to be removed because of its malfunction. We continued intensive supportive care. However, the laboratory data and general medical conditions gradually deteriorated. ECG showed little or no electrical activity. On day 5, ECG revealed weak electrical activities (Fig. 1E), and echocardiography showed a slight improvement of systolic function (Fig. 3C). She gradually recovered, and her ECG showed sinus rhythm (Figs. 1F and 3D). Since then, her condition started to gradually recover. Mechanical ventilator was weaned off on day 18, and ECMO was weaned off on day 20. She underwent cardiac magnetic resonance imaging after making sure that she was medically stable, which showed diffuse wall thinning, decreased wall motion and delayed enhancement of diffuse ventricular wall sparing of the infero-septal wall (Fig. 4). During hospitalization, she was complicated with ischemic insult in the lower extremities, hepatitis with jaundice, and acute tubular necrosis. On day 76, she was discharged from the hospital. The ejection fraction on the echocardiogram before discharge was approximately 30%. She receives wound dressing once a day and maintains hemodialysis every other day.
Fulminant myocarditis is an inflammatory process that occurs in the myocardium and has a fatal course due to the rapid development into acute heart failure or cardiogenic shock.1) Fulminant myocarditis is associated with various ECG alterations, including ST-T changes, Q-waves, atrioventricular- and intraventricular-conduction delays, atrial and ventricular tachyarrhythmia, and low voltage.2)
Our case demonstrated various alterations of ECG, which are some of the characteristics that are observed in the fulminant myocarditis. At first, ECG showed ST segmental changes associated with myocardial ischemia, then ECG showed various manifestations, such as ventricular tachycardia, atrial fibrillation with left bundle branch block, and complete atrioventricular block. It also displayed very little electrical activity on the whole leads, resembling an ECG after arrest. Low voltage on ECG can be observed in obesity, lung emphysema, pericardial effusion, pulmonary edema, hypothyroidism and amyloidosis, however low voltage on precordial leads, which is less common than on limb leads, implies cardiac tamponade or post-defibrillation state.3) Low voltage ECG in our case was probably due to pulmonary edema, pericardial effusion and edema of the ventricular wall.
Patients presented with syncope, new ventricular arrhythmia or atrioventricular block, ejection fraction of less than 40 percent, and lack of response to usual care within 1 to 2 weeks are associated with a high mortality rate in acute myocarditis.4)5)
Extracorporeal membrane oxygenation is instituted for providing emergent circulatory support to patients with cardiogenic shock.
Extracorporeal membrane oxygenation is not only useful for acute decompensated heart failure with low cardiac output, but also for life-threatening arrhythmia.6) It provides a bridge-to-recovery or decision regarding either heart transplantation or ventricular assist device. Its adaptation can physiologically reduce wall stress, decrease cytokine activation, and improve myocardial contractility. However, its use is limited by complications such as thromboembolic events.7) It is reported that patients with ischemic insult in the lower extremities or multi-organ failure due to thromboembolic events have poorer prognosis.6)
Long term mechanical circulatory support such as implantable left ventricular assist devices is better treatment modality for advanced heart failure patients with NYHA functional class III B or IV, INTERMACS level 1 to 5 as a bridge to recovery or bridge to transplantation.8) Unfortunately the device is not yet commercially abailable in Korea.
In summary, this patient had multiple factors associated with high mortality, such as low cardiac output and malignant arrhythmia. In addition, ischemic insults in the lower extremities and multi-organ failure (lung, kidney and liver) occurred during intensive therapy. These conditions further aggravated the anticipated poor prognosis. However, early adaptation of mechanical circulatory support, such as IABP and ECMO, combined with intensive medical therapy could save our patient with high-risk fulminant myocarditis.
Figures and Tables
Fig. 1
The electrocardiogram findings. A: incomplete right bundle branch block with ST-segment depression in diffuse precordial leads. B: wide QRS tachycardia with left bundle branch block (LBBB) pattern. C: atrial fibrillation with LBBB pattern of QRS. D: little or no electrical activities. E: junctional rhythm with low voltage and atrioventricular dissociation. F: sinus rhythm with Q-wave in V 1-3, negative T wave in II, III, aVF, V 5, V 6.
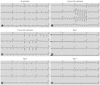
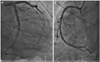
Fig. 2
Coronary angiography shows a near-normal coronary artery. A: left coronary artery. B: right coronary artery.
References
1. Feldman AM, McNamara D. Myocarditis. N Engl J Med. 2000; 343:1388–1398.
2. Morgera T, Di Lenarda A, Dreas L, et al. Electrocardiography of myocarditis revisited: clinical and prognostic significance of electrocardiographic changes. Am Heart J. 1992; 124:455–467.
3. Bruch C, Schmermund A, Dagres N, et al. Changes in QRS voltage in cardiac tamponade and pericardial effusion: reversibility after pericardiocentesis and after anti-inflammatory drug treatment. J Am Coll Cardiol. 2001; 38:219–226.
4. Hare JM, Baughman KL. Fulminant and acute lymphocytic myocarditis: the prognostic value of clinicopathological classification. Eur Heart J. 2001; 22:269–270.
5. McCarthy RE 3rd, Boehmer JP, Hruban RH, et al. Long-term outcome of fulminant myocarditis as compared with acute (nonfulminant) myocarditis. N Engl J Med. 2000; 342:690–695.
6. Aoyama N, Izumi T, Hiramori K, et al. National survey of fulminant myocarditis in Japan: therapeutic guidelines and long-term prognosis of using percutaneous cardiopulmonary support for fulminant myocarditis (special report from a scientific committee). Circ J. 2002; 66:133–144.
7. Marasco SF, Lukas G, McDonald M, McMillan J, Ihle B. Review of ECMO (extra corporeal membrane oxygenation) support in critically ill adult patients. Heart Lung Circ. 2008; 17:Suppl 4. S41–S47.
8. Slaughter MS, Rogers JG, Milano CA, et al. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med. 2009; 361:2241–2251.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download