Abstract
Intramural hematoma formation is not a well-studied complication of percutaneous coronary intervention. We describe a patient with stable angina who developed an intramural hematoma during elective percutaneous coronary intervention (PCI) in the right coronary artery (RCA). Total occlusion with dense dye staining developed a long way from the distal RCA, near the posterior descending artery bifurcation site. The true lumen was compressed by the enlarged, tense, false lumen. The patient was successfully treating with intravascular ultrasound-guided fenestration using a cutting balloon, and a stent was implanted in the distal RCA.
The rate of periprocedural coronary occlusion has been reported to be 6.7% in the setting of balloon angioplasty.1) Acute closure of a coronary artery associated with dissection, coronary spasm, and/or thrombus formation usually occurs within 6 hours after balloon angioplasty.2) Several case reports have shown a different type of dissection morphology suggestive of intramural hematoma formation.3-6) Post-percutaneous coronary intervention (PCI) intramural hematomas have not been well studied, and their prevalence is unknown. Intramural hematomas are difficult to diagnose angiographically, and they may be missed when they do not cause severe luminal narrowing. Treatment may be conservative or may entail stenting or surgery.
We present an interesting case of an intramural hematoma complicating elective PCI. The patient was successfully treated with cutting balloon fenestration and stent implantation.
A 72-year-old woman was admitted to our hospital to be evaluated for a 5-year history of exertional chest pain. She had suffered from diabetes and hypertension for 25 years. On admission, her blood pressure was 126/75 mmHg, and her pulse rate was 92 bpm. There were no specific findings on electrocardiography or echocardiography. Cardiac serum markers were within normal range, and the N-terminal proB-type natriuretic peptide level was 706.9 pg/mL. Coronary angiography revealed focal stenosis leading to 90% luminal narrowing of the midright coronary artery (RCA), 99% narrowing of the posterior descending artery (PDA) ostium, and 95% narrowing of the distal left circumflex artery (LCX) (Fig. 1). One-stage elective PCI was performed for mid-RCA, PDA ostial, and distal LCX lesions. We started PCI with a Runthrough guidewire (Terumo, Japan) and a 6 Fr Judkins right guiding catheter (Cordis, Florida, USA). The simple 90% mid-RCA focal lesion was dilated with a 3.0×15 mm balloon (12 atm) and treated with a 3.5×24 mm zotarolimus-eluting stent (Endeavor, Medtronic, Minnesota, USA) (12 atm). Unexpected total occlusion and dye staining developed near the PDA bifurcation site after ballooning was complete (Fig. 2). The patient experienced significant chest pain, and her blood pressure decreased to 85/50 mmHg. We attempted catheter thrombectomy with a Thrombuster aspiration catheter (Kaneka, Japan) and attempted another dilatation using a 3.0×15 mm balloon, but we were unsuccessful. Intravascular ultrasound (IVUS) study revealed a totally compressed lumen stretching from the stented segment to the far distal RCA, which was caused by a huge hematoma (Fig. 3). We planned for fenestration between the true lumen and the hematoma. A 2.75×10 mm cutting balloon was placed at the RCA-PDA bifurcation site and dilated gently up to 4 atm, after which it burst (Fig. 4). A 2.5×10 mm cutting balloon was dilated in the posterolateral branch (4 atm) and PDA ostium (1.5 atm), in succession. Angiography showed restored thrombolysis in myocardial infarction (TIMI)-3 flow with a huge, long dissection spanning from the middle of the stented segment to the far distal RCA. This dissection was further confirmed by an additional IVUS study. In the interest of minimizing the dissected segment, a 3.0×30 mm Endeavor stent (12 atm) was implanted in the distal RCA. The final angiography was acceptable. After the procedure, the patient's symptoms subsided, and her blood pressure returned to normal range. She was discharged after 4 days and followed in the outpatient department for more than 6 months, with no symptoms.
Intramural hematoma formation is a rare but potentially life-threatening complication occurring after angioplasty.7) It is defined as an accumulation of blood within the medial space. It is a variant of dissection characterized by no direct communication between the true and false lumens.8) In a study of 905 patients undergoing PCI, intramural hematomas were detected in 6.7% using IVUS.1) The hematoma was proximal to the lesion in 36 percent, confined to the lesion in 18 percent, and distal to the lesion in 46 percent.1) Patients with intramural hematomas had a greater than 3-fold increase in their target vessel revascularization rate within 1 month.1)
Intramural hematoma formation occurs through subintimal dissection into the media, with extension into the medial space of the contiguous normal arterial segments where blood accumulates without a flap or identifiable entry or exit points. Murphy et al.3) studied patients who underwent emergent coronary artery bypass surgery (CABG) after PCI and found that all had false lumens with bloody fluid, suggesting dissecting hematomas. Block et al.5) reported a patient who died 90 hours after balloon angioplasty and who had a dissecting hematoma originating at the angioplasty site and extending distally. One common location for dissection after angioplasty is at the junction of the thinnest plaque and adjacent normal arterial wall.9)10) During PCI, the stiffer atherosclerotic plaque resists circumferential expansion more than the normal site does, and high shear stresses are generated at their interface, promoting dissection.11) Waller et al.12) reported that the thickness of the media behind the atherosclerotic plaque is less than half that of the normal wall. Similarly, after stent implantation, dissection may occur at the edge of a stent; that is, the transition point between the rigid stent and the unstented segment.13)
Angiographic findings of a intramural hematoma include tapering encroachment of the lumen, absence of a dissection flap, and refractoriness to intracoronary vasodilators.6)14) One third of all intramural hematomas show no angiographic abnormalities. In these cases, IVUS is the only diagnostic tool that can identify post-PCI intramural hematoma formation.1) Because IVUS permits detailed high-quality cross-sectional imaging of the coronary arteries, it allows for detection of intramural hematomas with or without luminal compression.2) IVUS can identify the entry site and expansion of the hematoma into normal segments of the arterial wall. On IVUS, an intramural hematoma appears as a homogeneous, hyperechoic, crescent-shaped area.15-17) Echogenicity depends on the flow rate, red cell aggregation, and fibrin content. Intramural hematomas can also contain distinct echolucent zones within the hyperechoic areas.18) These echolucent areas represent accumulation of radiographic contrast or saline within the hematoma space.1)
Proper management of post-PCI intramural hematomas has not been well-defined. However, short-term complications (non-Q-wave myocardial infarction and need for repeat revascularization within 1 month) and long-term outcomes (death and need for repeat revascularization at 1 year) are serious.1) Bailout therapies to relieve the luminal obstruction include stenting or redilatation and creation of a typical dissection.15) Massive hematomas may require stent implantation to optimize blood flow. If the hematoma is extensive, long lesion stent deployment may be neccessary.19) The use of angioplasty alone, without stenting, is associated with a potential risk of dissection propagation.20) IVUS may be useful in determining and controlling the bailout therapy, which includes stenting of the entry point at the initial dilatation site. Surgery must be considered in patients who have hematomas extending into the left main coronary artery.15)
We reported a case of acute vessel obstruction after coronary stenting, due to intramural hematoma formation. IVUS offers advantages in determining the underlying mechanism of luminal narrowing.
Figures and Tables
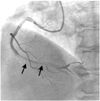
Fig. 1
Elective coronary angiography. The arrow indicates focal stenosis leading to 90% luminal narrowing in the mid-right coronary artery. The arrowhead indicates 99% luminal narrowing of the posterior descending artery ostium.

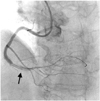
Fig. 2
Angiographic view of new narrowing that formed after stenting. Arrows indicate total occlusion and dye staining far from the distal right coronary artery, near the posterior descending artery bifurcation site.
Acknowledgments
We thank Ms. Gyeong-Ja Jeong and Miss Youn-Jin Noh for their assistance in writing this manuscript.
References
1. Maehara A, Mintz GS, Bui AB, et al. Incidence, morphology, angiographic findings, and outcomes of intramural hematomas after percutaneous coronary interventions. Circulation. 2002. 105:2037–2042.
2. Hirose M, Kobayashi Y, Kreps EM, et al. Luminal narrowing due to intramural hematoma shift from left anterior descending coronary artery to left circumflex artery. Catheter Cardiovasc Interv. 2004. 62:461–465.
3. Murphy DA, Craver JM, King SB 3rd. Distal coronary artery dissection following percutaneous transluminal coronary angioplasty. Ann Thorac Surg. 1984. 37:473–478.
4. Zack PM, Ischinger T. Late progression of an asymptomatic intimal tear to occlusive coronary artery dissection following angioplasty. Cathet Cardiovasc Diagn. 1985. 11:41–48.
5. Block PC, Myer RK, Stertzer S, Fallon JT. Morphology after transluminal angioplasty in human beings. N Engl J Med. 1981. 305:382–385.
6. Stauffer JC, Sigwart U, Goy JJ, Kappenberger L. Milking dissection: an unusual complication of emergency coronary artery stenting for acute occlusion. Am Heart J. 1991. 121:1539–1542.
7. Shirodaria C, van Gaal WJ, Banning AP. A bleeding kiss: intramural haematoma secondary to balloon angioplasty. Cardiovasc Ultrasound. 2007. 5:21.
8. Song JK, Kang DH, Lee KM, et al. Two cases of aortic intramural hematoma diagnosed with transesophageal echocardiography. Korean Circ J. 1994. 24:904–909.
9. van der Lugt A, Gussenhoven EJ, von Birgelen C, Tai JA, Pieterman H. Failure of intravascular ultrasound to predict dissection after balloon angioplasty by using plaque characteristics. Am Heart J. 1997. 134:1075–1081.
10. Farb A, Virmani R, Atkinson JB, Kolodgie FD. Plaque morphology and pathologic changes in arteries from patients dying after coronary balloon angioplasty. J Am Coll Cardiol. 1990. 16:1421–1429.
11. Lee RT, Kamm RD. Vascular mechanics for the cardiologist. J Am Coll Cardiol. 1994. 23:1289–1295.
12. Waller BF. The eccentric coronary atherosclerotic plaque: morphologic observations and clinical relevance. Clin Cardiol. 1989. 12:14–20.
13. Sheris SJ, Canos MR, Weissman NJ. Natural history of intravascular ultrasound-detected edge dissections from coronary stent deployment. Am Heart J. 2000. 139:59–63.
14. Werner GS, Figulla HR, Grosse W, Kreuzer H. Extensive intramural hematoma as the cause of failed coronary angioplasty: diagnosis by intravascular ultrasound and treatment by stent implantation. Cathet Cardiovasc Diagn. 1995. 36:173–178.
15. Werner GS, Diedrich J, Kreuzer H. Sonographic and angiographic features of intramural hematoma as a cause of failed coronary angioplasty. J Invasive Cardiol. 1996. 8:208–214.
16. Mintz GS, Nissen SE, Anderson WD, et al. American College of Cardiology Clinical Expert Consensus Document on Standards for the Acquisition, Measurement, and Reporting of Intravascular Ultrasound Studies: a report of the American College of Cardiology Task Force on Clinical Expert Consensus Documents. J Am Coll Cardiol. 2001. 37:1478–1492.
17. Mahr P, Ge J, Haude M, Gorge G, Erbel R. Extramural vessel wall hematoma causing a reduced vessel diameter after coronary stenting: diagnosis by intravascular ultrasound and treatment by stent implantation. Cathet Cardiovasc Diagn. 1998. 43:438–443.
18. Fowlkes JB, Strieter RM, Downing LJ, et al. Ultrasound echogenicity in experimental venous thrombosis. Ultrasound Med Biol. 1998. 24:1175–1182.
19. Sawada T, Shite J, Shinke T, et al. Persistent malapposition after implantation of sirolimus-eluting stent into intramural coronary hematoma: optical coherence tomography observations. Circ J. 2006. 70:1515–1519.
20. Ahn SG, Tahk SJ, Choi JH, et al. Spontaneous coronary artery dissection manifested during ergonovine test and treated with intravascular ultrasound guided stenting: a case report. Korean Circ J. 2005. 35:264–268.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download