Abstract
Background and Objectives
Ventricular arrhythmia is an important cause of death in patients suffering with idiopathic dilated cardiomyopathy (IDCM). The decreased uptake or increased "washout" of 123I-metaiodobenzylguanidine (MIBG)-derived radioactivity is associated with a worse prognosis or severe disease in patients with cardiac diseases.
Subjects and Methods
Forty patients (28 men, mean age: 56.5±13.0 years) with angiographically proven IDCM were studied and divided into 2 groups: the patients with clinically documented ventricular tachycardia (VT) and IDCM (group A; 17 patients), and the patients without VT, but with IDCM (group B; 23 patients). The normal control group (7 men, mean age 62.4±7.9) was compared theses patients. 123I-MIBG was evaluated by the early (15 min) and delayed (3hours) uptake, the heart to mediastinum uptake (H/M) ratio and the global and regional washout ratio (WOR) that was defined as the [early H uptake-delayed H uptake]/early H uptake. The clinical variables, echocardiographic parameters and 123I-MIBG-derived parameters were compared among three groups.
Results
There were no significant differences in age and the echocardiographic parameters between the groups A and B. A significant negative correlation was present between the early inferior wall H/M ratio and the LVEDD (Spearman's correlation, r=-0.412, p=0.08), and the early inferior H/M ratio and the LVESD (Spearman's correlation, r=-0.463, p=0.03). Only the delayed lateral wall uptake was significantly lower in group A than that in group B (23.3±5.3 versus 27.8±8.4, respectively p=0.042). The regional WOR was not statically different among the three groups.
Figures and Tables
Fig. 1
Correlation between left ventricular (LV) function and early inferior wall H/M ratio. There was positive correlation between early inferior wall H/M ratio and LV ejection fraction (EF), and negative correlation is shown between LV end-diastolic dimension (LVEDD), LV end-systolic dimension (LVESD) and early inferior wall H/M ratio. VT: ventricular tachycardia, H/M: heart to mediastinum uptake.

Fig. 2
Difference of 123I- Metaiodobenzylguanidine (123I-MIBG) finding in a control patient and a patient with idiopathic dilated cardiomyopathy (IDCM). Compared with early and delayed 123I-MIBG uptake in control (2.04 and 2.34 respectively), idiopathic dilated cardiomyopathy (IDCM) patient showed diminished early and delayed 123I-MIBG uptake (1.50 and 1.31 respectively), and an increase of washout rate (arrowhead) (0.13 vs 0.41 respectively).
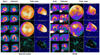
Fig. 3
Example of mismatch between 123I-metaiodobenzylguanidine (123I-MIBG) and 99mTc-methoxy isobutyl isonitrile (99mTc-MIBI) single photon emission computed tomography (SPECT). Compared with the 99mTc-MIBI, 123I-MIBG SPECT shows more extensive uptake defect in inferior and lateral wall especially in delayed image (arrowhead). That means sympathetic denervation is more severe or extensive than the degree of myocardial dysfunction.

Table 1
Clinical characteristics of the study patients
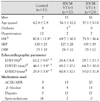
Values are mean±SD. *p<0.05, between control and IDCM VT (+), †p<0.05, between control and IDCM VT (-), ‡p<0.05, between IDCM VT (+) and IDCM VT (-), §p<0.05, among three groups. IDCM: idiopathic dilated cardiomyopathy, LVEF: left ventricular ejection fraction, EF: ejection fraction, LVEDD: left ventricular enddiastolic dimension, LVESD: LV end-systolic dimension, ACEI: angiotensin converting enzyme inhibitor, ARB: angiotensin converting enzyme receptor blocker, VT: ventricular tachycardia, HR: heart rate, SBP: systolic blood pressure, DBP: diastolic blood pressure, SD: standard deviations
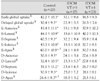
Table 2
Global and regional 123I-MIBG uptake at early and delayed phase according to the presence of VT
References
1. Higuchi T, Schwaiger M. Imaging cardiac neuronal function and dysfunction. Curr Cardiol Rep. 2006. 8:131–138.
2. Knuuti J, Sipola P. Is it time for cardiac innervation imaging? Q J Nucl Med Mol Imaging. 2005. 49:97–105.
3. Kang DY. Clinical application of I-123 MIBG cardiac imaging. Korean J Nucl Med. 2004. 38:331–337.
4. Ha JW, Lee JD, Chung N, et al. Iodine-123-metaiodobenzylguanidine myocardial scintigraphy in patient with dilated cardiomyopathy: correlation between myocardial MIBG uptake and echocardiographic parameter. Korean Circ J. 1996. 26:651–666.
5. Ha JW, Lee JD, Jang Y, et al. Assessment of cardiac sympathetic neuronal integrity using Iodine-123 metaiodobenzylguanidine myocardial scintigraphy and its clinical efficacy as a new noninvasive screening test for the diagnosis of coronary artery spasm. Korean Circ J. 1998. 28:183–193.
6. Wakabayashi T, Nakata T, Hashimoto A, et al. Assessment of underlying etiology and cardiac sympathetic innervation to identify patients at high risk of cardiac death. J Nucl Med. 2001. 42:1757–1767.
7. Imamura Y, Fukuyama T. Prognostic value of myocardial MIBG scintigraphy findings in patients with cardiomyopathy: importance of background correction for quantification of MIBG activity. Ann Nucl Med. 2002. 16:387–393.
8. Gill JS, Hunter GJ, Gane J, Ward DE, Camm AJ. Asymmetry of cardiac 123I- meta-iodobenzyl-guanidine scans in patients with ventricular tachycardia and a "clinically normal" heart. Br Heart J. 1993. 69:6–13.
9. Mitrani RD, Klein LS, Miles WM, et al. Regional cardiac sympathetic denervation in patients with ventricular tachycardia in the absence of coronary artery disease. J Am Coll Cardiol. 1993. 22:1344–1353.
10. Yoshioka K, Gao DW, Chin M, et al. Heterogeneous sympathetic innervation influences local myocardial repolarization in normally perfused rabbit hearts. Circulation. 2000. 101:1060–1066.
11. Ferrans VJ, Hibbs RG, Walsh JJ, Burch GE. Histochemical and electron microscopical studies on the cardiac necroses produced by sympathomimetic agents. Ann N Y Acad Sci. 1969. 156:309–332.
12. Kim SJ, Lee DY, Lee JD, et al. Evaluation of sympathetic innervation in cardiomyopathy with 123I-MIBG. Korean J Nucl Med. 1993. 27:195–202.
13. Gao DW, Talke PO, Dae MW. Effects of reduced sympathetic activity on myocardial metaiodobenzylguanidine (MIBG) washout. J Auton Pharmacol. 2001. 21:159–164.
14. Dae MW, O'Connell JW, Botvinick EH, et al. Scintigraphic assessment of regional cardiac adrenergic innervation. Circulation. 1989. 79:634–644.
15. Turpeinen A, Vanninen E, Magga J, Tuomainen P, Kuusisto J, Sipola P. Cardiac sympathetic activity is associated with elevated levels of proinflammatory cytokines in patients with idiopathic dilated cardiomyopathy. Eur J Nucl Med. 2004. 31:S292.
16. Kyuma M, Nakata T, Hashimoto A, et al. Incremental prognostic implications of brain natriuretic peptide, cardiac sympathetic nerve innervation, and noncardiac disorders in patients with heart failure. J Nucl Med. 2004. 45:155–163.
17. Maeno M, Ishida Y, Shimonagata T, et al. The significance of 201Tl/123I MIBG (metaiodobenzylguanidine) mismatched myocardial regions for predicting ventricular tachycardia in patients with idiopathic dilated cardiomyopathy. Kaku Igaku. 1993. 30:1221–1229.
18. Henderson EB, Kahn JK, Corbett JR, et al. Abnormal I-123 metaiodobenzylguanidine myocardial washout and distribution may reflect myocardial adrenergic derangement in patients with congestive cardiomyopathy. Circulation. 1988. 78:1192–1199.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download