Abstract
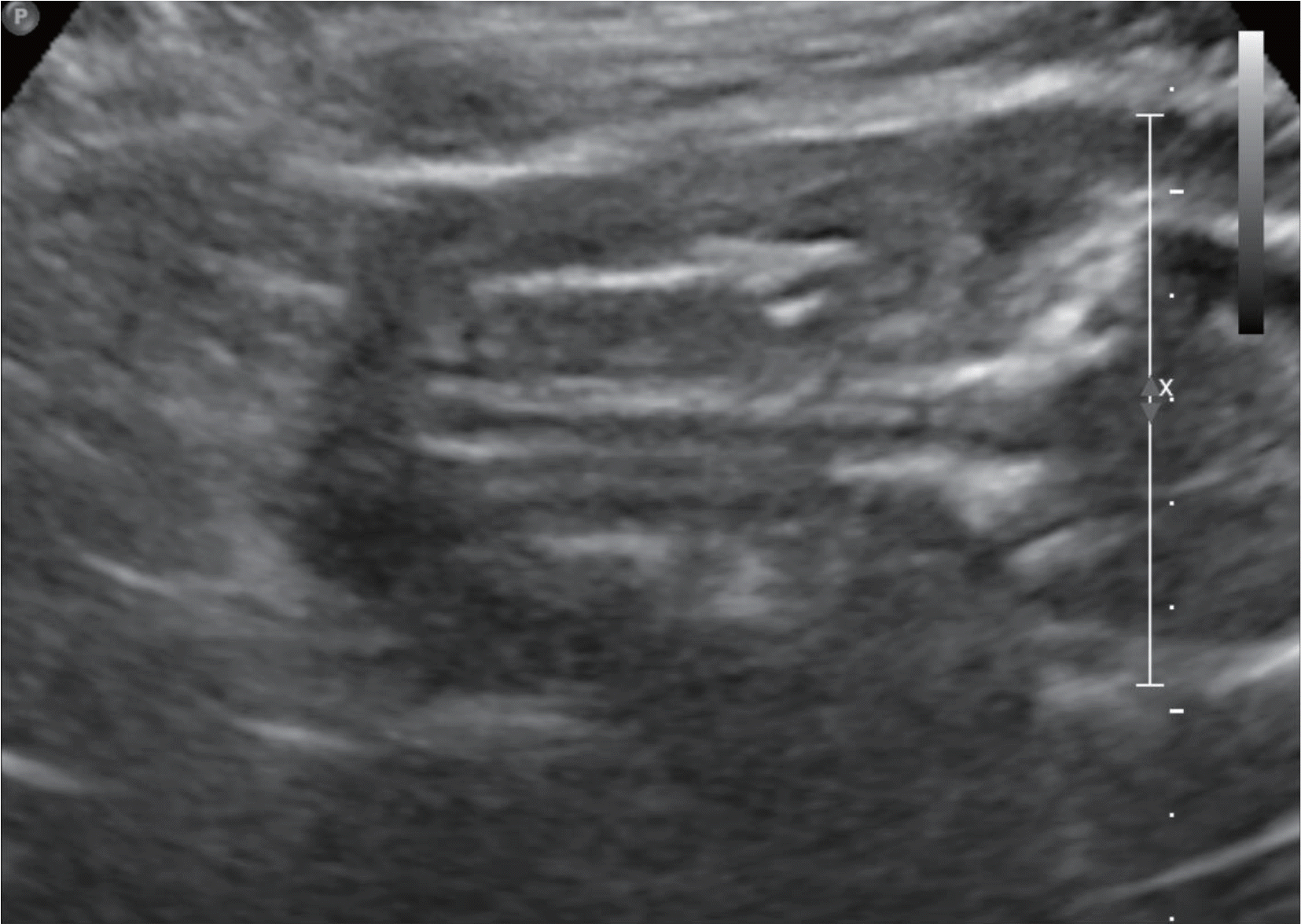
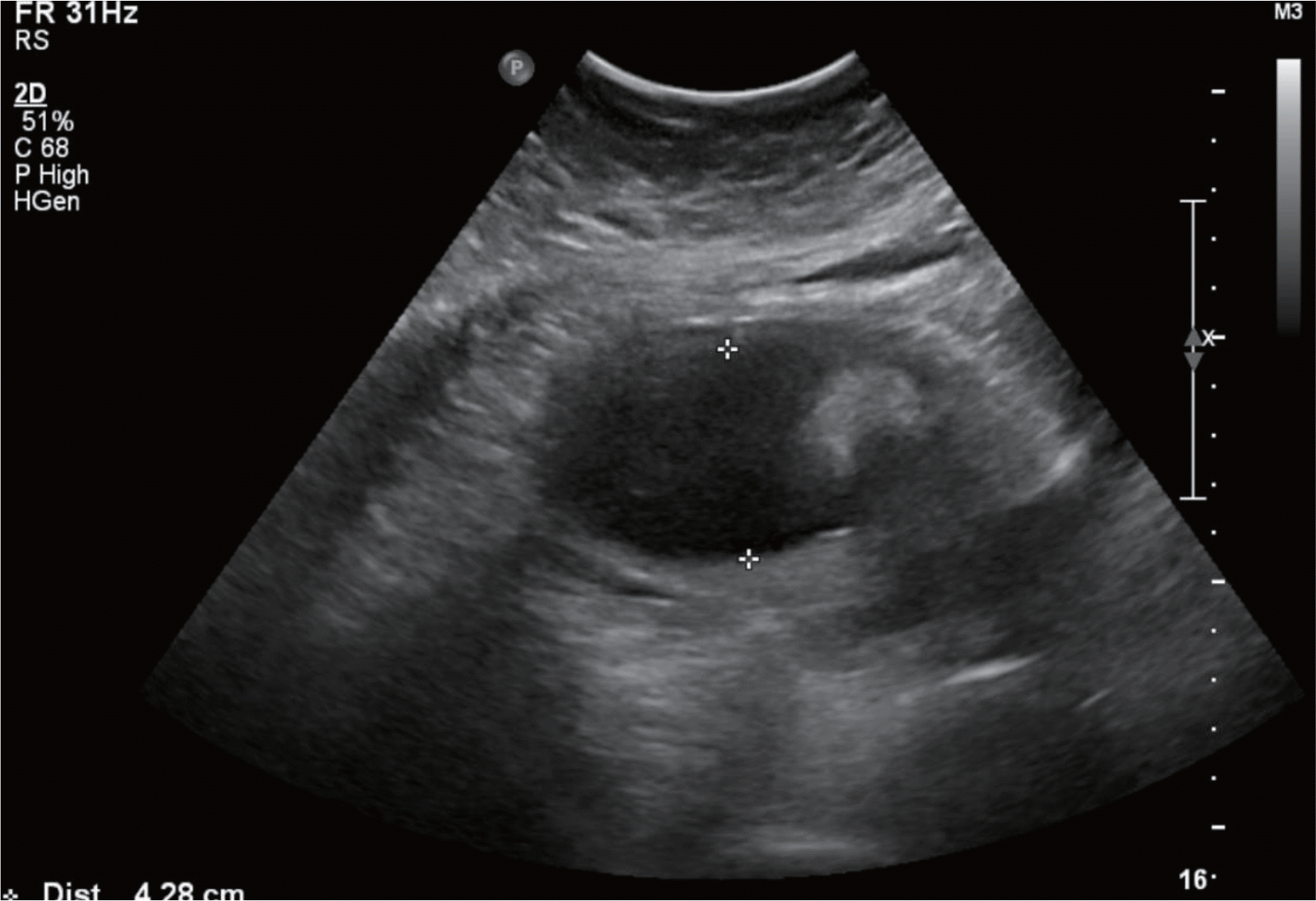
Various fetal and maternal conditions are known to be associated with oligohydramnios. In general, oligohydramnios developed early in pregnancy is less common but frequently has a poor prognosis. The use of angiotensin-converting enzyme inhibitors or angiotensin receptor antagonists during pregnancy has been associated with oligohydramnios as well as growth restriction, pulmonary hypoplasia with respiratory distress, acute renal failure, cranial malformation and fetal death. Although many researchers report a use of oral hypoglycemic agents such as glyburide or metformin in gestational diabetes mellitus, but potential adverse effects of glimepiride, which is relatively recently developed, is not well known owing to the lack of clinical data, especially early in pregnancy. A 41-year-old woman with chronic hypertension and type 2 diabetes mellitus was treated with drugs including metformin, glimepiride and angiotensin receptor antagonist until approximately 20 weeks' gestations, when severe oligohydramnios was noted. After the hospitalization for bed rest, fetal surveillance, and discontinuation of the agents, amniotic fluid reaccumulated, and the infant was delivered at term. We report this case with a brief review of literatures.
REFERENCES
1). Pryde PG., Sedman AB., Nugent CE., Barr M Jr. Angiotensin-converting enzyme inhibitor fetopathy. J Am Soc Nephrol. 1993. 3:1575–82.

2). Bullo M., Tschumi S., Bucher BS., Bianchetti MG., Simonetti GD. Pregnancy outcome following exposure to angiotensin-converting enzyme inhibitors or angiotensin receptor antagonists: a systematic review. Hypertension. 2012. 60:444–50.
3). Langer O., Conway DL., Berkus MD., Xenakis EM., Gonzales O. A comparison of glyburide and insulin in women with gestational diabetes mellitus. N Engl J Med. 2000. 343:1134–8.

4). Nicholson W., Baptiste-Roberts K. Oral hypoglycaemic agents during pregnancy: The evidence for effectiveness and safety. Best Practice & Research Clinical Obstet Gynaecol. 2011. 25:51–63.

5). Vanky E., Stridsklev S., Heimstad R., Romundstad P., Skogøy K., Kleggetveit O, et al. Metformin versus placebo from first trimester to delivery in polycystic ovary syndrome: a randomized, controlled multicenter study. J Clin Endocrinol Metab. 2010. 95:448–55.

6). Piacquadio K., Hollingsworth DR., Murphy H. Effects of in-utero exposure to oral hypoglycaemic drugs. Lancet. 1991. 338:866–9.

7). American College of Obstetricians and Gynecologists: Gestational diabetes. Obstet Gynecol. 2001. 98:525–38.
8). Kraus GW., Marchese JR., Yen SS. Prophylactic use of hydrochlorothiazide in pregnancy. JAMA. 1966. 198:1150–4.

9). Ahn HK., Nava-Ocampo AA., Han JY., Choi JS., Chung JH., Yang JH, et al. Exposure to amlodipine in the first trimester of pregnancy and during breastfeeding. Hypertens Pregnancy. 2007. 26:179–87.

10). Cooper WO., Hernandez-Diaz S., Arbogast PG., Dudley JA., Dyer S., Gideon PS, et al. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med. 2006. 354:2443–51.

11). Li DK., Yang C., Andrade S., Tavares V., Ferber JR. Maternal exposure to angiotensin converting enzyme inhibitors in the first trimester and risk of malformations in offspring: a retrospective cohort study. BMJ. 2011. 343:d5931.

12). Chisholm CA., Chescheir NC., Kennedy M. Reversible oligohydramnios in a pregnancy with angiotensin-converting enzyme inhibitor exposure. Am J Perinatol. 1997. 14:511–3.

13). Muller PR., James A. Pregnancy with prolonged fetal exposure to an angiotensin-converting enzyme inhibitor. J Perinatol. 2002. 22:582–4.

14). Celentano C., Prefumo F., di Vera E., Iannicco A., Gallo DP., Liberati M. Reversible acute fetal renal failure due to maternal exposure to angiotensin receptor blocker. Pediatr Nephol. 2008. 23:333–4.

15). Hanssens M., Keirse MJ., Vankelecom F., Van Assche FA. Fetal and neonatal effects of treatment with angiotensin-converting enzyme inhibitors in pregnancy. Obstet Gynecol. 1991. 78:128–35.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download