Abstract
Purpose
Despite the fact that intraarticular corticosteroids are a longstanding adjuvant treatment for inflammatory arthritis, their mechanisms of action are not completely understood. This study evaluated the osteoprotegerin (OPG) and receptor activator of nuclear factor-κB ligand (RANKL) levels in the serum and synovial fluid before and 2 weeks after injecting intraarticular corticosteroid.
Materials and Methods
Sixty seven patients were identified with joint effusions of the knee and suffering from rheumatoid arthritis. All the patients received an intraarticular injection of 40 mg of triamcinolone acetonide. The sera and synovial fluid were obtained before and 2 weeks after the injections. the erythrocyte sedimentation rate, C-reactive protein, rheumatoid factor, serum OPG and RANKL levels and synovial OPG and RANKL levels were measured before and 2 weeks after the injections.
Results
The intraarticular corticosteroids induced a significant decrease in the levels of synovial RANKL. The decrease in the level of synovial RANKL caused a significant increase in the synovial OPG/RANKL ratio. There were no changes in the hematological factors, serum OPG, serum RANKL or synovial OPG observed after the corticosteroid injection.
Rheumatoid arthritis (RA) might result in local bone destruction through the recruitment of osteoclasts to the inflammation site3). This process is regulated through the receptor activator of nuclear factor-κB ligand (RANKL), receptor activator of nuclear factor-κB (RANK) and osteoprotegerin (OPG) system22).
RANKL is a member of the tumor necrosis factor (TNF) ligand super-family. It is secreted mainly by osteoblasts13) and activated T cells25) after stimulation with proinflammatory cytokines, such as TNF and interleukin-19). RANKL interacts with its receptor RANK on the surface of premature osteoclasts, which induces osteoclast formation, activation, and survival12). In addition to its role in osteoclatogenesis, RANKL acts as an immune mediator that promotes the activation of monocytes19) as well as the interactions between dendritic cells and T cells23). OPG is a member of the TNF receptor superfamily, and is a soluble decoy receptor with the ability to block the interaction between RANKL and RANK12). A high RANKL/OPG ratio is associated with increased bone resorption8) and active synovitis6).
Intraarticular corticosteroids are potent anti-inflammatory compounds that have been used successfully by clinicians as an adjuvant therapy for inflammatory arthritis. However, the mechanism underlying the effects of intraarticular corticosteroids is not completely understood. Although recent data suggest that a corticosteroid treatment has a positive effect in preventing bone erosion in RA13), there is still some concern regarding the balance between their two potentially divergent effects (bone protection versus osteopenia). Therefore, this study examined the effects of intraarticular corticosteroids on the sera and synovial fluid levels of OPG and RANKL.
Sixty seven patients with joint effusions and were suffering from RA visited the authors' institution for their rheumatic diseases. Individuals with RA were defined as those who fulfilled the criteria of the ACR1). All patients received an intraarticular injection of 40 mg of triamcinolone acetonide. The sera and synovial fluids were obtained before and 2 weeks after the injections. The erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), rheumatoid factor (RF), serum OPG and RANKL levels and synovial OPG and RANKL levels were measured before and 2 weeks after the injections. All the other associated treatments including disease-modifying drugs, nonsteroidal anti-inflammatory drugs and oral corticosteroids were maintained at constant levels for at least 4 weeks before and throughout the study period. All the subjects provided informed consent before undergoing the examinations and measurements. The study was approved by the Clinical Research Ethics Committee of the University and hospital.
The serum was obtained from routinely-drawn blood samples, and centrifuged immediately. The synovial fluid was obtained on the same day and defibrinated mechanically. The samples were stored at -80℃ before measuring the RANKL and OPG levels. The serum and synovial OPG levels were measured using sandwich ELISA (Immundiagnostik, Bensheim, Germany) method. The assay included two highly specific antibodies against OPG. The antibody is capable of neutralizing the biological activity of the recombinant human OPG. The detection antibody was a biotin-labelled polyclonal antihuman OPG antibody derived from a goat and immunized with the human recombinant OPG. The serum and synovial OPG concentrations were calculated based on the protein concentration according to the manufacturer. The assay was designed to detect the monomeric, dimeric, and ligand-bound forms of circulating OPG. The intra-assay coefficient of variation (CV) for the OPG measurements was 8-10% and the interassay CV was <10%.
The RANKL levels in the serum were measured by sandwich ELISA. As the first step, the blood sample and the biotinylated anti-RANKL detection antibody were pipetted into the wells. Human RANKL, if present in the sample, binds to the precoated recombinant OPG and forms a sandwich with the detection antibody. After a washing step, which removed all the nonspecific bound material, the streptavidin-HRP conjugate was added to the wells. After removing the unbound conjugate by washing, tetramethylbenzidine was added to the wells as a substrate. RANKL was quantified by the enzyme catalyzed color changes on a standard ELISA reader. The extent of color development is directly proportional to the amount of RANKL present the sample. The intra-assay CV for the RANKL measurement was 3-5% and the interassay CV was 6-9%. The detection limits for the OPG and RANKL assays were 0.14 pmol/l and 0.08 pmol/l, respectively.
Statistical analysis was performed using SPSS 11.5 software for Windows (SPSS, Chicago, IL, USA). The data is expressed as the mean (ranges). The differences in the data before and 2 weeks after the injections were analyzed using a paired t-test, and the correlations are presented using the Pearson's correlation. A p-value<0.05 was considered significant.
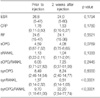
Table 1 shows the results of the patients with RA before and 2 weeks after the corticosteroid injections. The intraarticular corticosteroids induced a significant decrease in the synovial RANKL level. This decrease resulted in a significant increase in the synovial OPG/RANKL ratio, from a mean of 9.70 to a mean of 22.20. There were no changes in the hematology factors, serum OPG, serum RANKL or synovial OPG after the corticosteroid injections.
As expected, there was an inverse correlation between the OPG and RANKL levels in the sera and synovial fluids before and 2 weeks after the corticosteroid injections (Table 2, 3). There were positive correlations between the serum OPG and synovial OPG before and 2 weeks after the corticosteroid injections. In addition, there was a positive correlation between the serum RANKL and synovial RANKL before and 2 weeks after the injection. However, there was no correlation between the hematology factors including the erythrocyte sedimentation rate, C-reactive protein and rheumatoid factor and OPG or RANKL.
RA is characterized by chronic inflammation of the synovium, the predominance of pro-erosive mediators, and the subsequent destruction of bone and cartilage. Recently, a mechanism of bone destruction that could be dissociated from inflammation was proposed4,18). Cells with the specific surface markers of osteoclasts were found in areas of a pannus invasion into the bone at the sites of bone erosion in patients with RA5). RANKL and its naturally occurring decoy receptor, OPG, play important roles in normal bone remodeling10) as well as in diseases associated involving bone destruction, as demonstrated in animal models of arthritis10) and in human diseases such as RA20), erosive psoriatic arthritis17) and multiple myeloma21). RANKL induces osteoclastic bone destruction, and OPG protects against bone destruction by preventing the binding of RANKL with its receptor RANK4,18). Bone resorption is regulated locally by the relative levels of RANKL and OPG expression11).
Despite the fact that intraarticular corticosteroids have been a longstanding adjuvant treatment for inflammatory arthritis, their mechanisms of action are not completely understood. Therefore, this study evaluated the OPG and RANKL levels in the sera and synovial fluids before and 2 weeks after the intraarticular corticosteroid injections.
Since their first use as a treatment for RA more than 50 years ago, corticosteroids have been the subject of clinical debate based on their potential deleterious effects on the bone density and bone function, as well as their potential positive effects. Secondary osteoporosis is an important limitation of the long term systemic treatment with high doses of corticosteroids16). This observation has raised concerns regarding their use in RA. Although there is little data available regarding intraarticular corticosteroids2), it has been suggested that local corticosteroids might contribute to local disease control15). In our group of patients, treatment with corticosteroids reduced the signs and symptoms of local inflammation.
Since the anti-inflammatory effect of corticosteroids might help slow the progression of damage to the injected joints, this study evaluated the effect of corticosteroids on the OPG/RANKL system, which is considered to be a major determinant of the bone biology in inflammatory arthritis24). To date, there are few reports regarding the in vivo influence of effective antirheumatic drugs on the OPG/RANKL axis. Makrygiannakis et al.14) reported that intraarticular corticosteroids decreased the level of synovial RANKL expression in inflammatory arthritis by an immunohistochemical and microscopic analysis of the synovim. Conaghan et al.2) reported that intraarticular corticosteroids decrease the level of synovitis and new erosions of joint. In addition, Hirayama et al.7) reported a biphasic effect of glucocorticoids on osteoclasts in vitro. The first involved the stimulation of osteoclast formation through the promotion of proliferation and differentiation of osteoclast precursors. The second involved the inhibition of the bone-resorbing activity of mature osteoclasts. However, serological methods of OPG and RANKL in peripheral blood and synovial fluid were used in this study. It was found that corticosteroids modulated the OPG/RANKL system in the synovial fluid by down-regulating RANKL. However, intraarticular corticosteroids did not modulate the OPG/RANKL system in the serum. It is possible that the local delivery of corticosteroids at the site of inflammation in acute, severe flares of the disease is a valuable therapeutic choice for combating inflammation and bone destruction without the risk of osteoporosis observed with the long term systemic administration of high doses of corticosteroids.
This study demonstrated that treatment with intraarticular corticosteroids modulated the OPG/RANKL system in the synovial fluid toward a bone-protective effect in inflammatory arthritis. This mechanism might be one explanation for the mode of action of intraarticular corticosteroids.
Figures and Tables
References
1. Arnett FC, Edworthy SM, Bloch DA, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988. 31:315–324.

2. Conaghan PG, O'Connor P, McGonagle D, et al. Elucidation of the relationship between synovitis and bone damage: a randomized magnetic resonance imaging study of individual joints in patients with early rheumatoid arthritis. Arthritis Rheum. 2003. 48:64–71.

3. Goldring SR, Gravallese EM. Pathogenesis of bone lesions in rheumatoid arthritis. Curr Rheumatol Rep. 2002. 4:226–231.

4. Gravallese EM, Goldring SR. Cellular mechanisms and the role of cytokines in bone erosions in rheumatoid arthritis. Arthritis Rheum. 2000. 43:2143–2151.

5. Gravallese EM, Manning C, Tsay A, et al. Synovial tissue in rheumatoid arthritis is a source of osteoclast differentiation factor. Arthritis Rheum. 2000. 43:250–258.

6. Haynes DR, Barg E, Crotti TN, et al. Osteoprotegerin expression in synovial tissue from patients with rheumatoid arthritis, spondyloarthropathies and osteoarthritis and normal controls. Rheumatology (Oxford). 2003. 42:123–134.

7. Hirayama T, Sabokbar A, Athanasou NA. Effect of corticosteroids on human osteoclast formation and activity. J Endocrinol. 2002. 175:155–163.

8. Hofbauer LC, Heufelder AE. Role of receptor activator of nuclear factor-KappaB ligand and osteoprotegerin in bone cell biology. J Mol Med. 2001. 79:243–253.
9. Hofbauer LC, Lacey DL, Dunstan CR, Spelsberg TC, Riggs BL, Khosla S. Interleukin-1β and tumor necrosis factor-α, but not interleukin-6, stimulate osteoprotegerin ligand gene expression in human osteoblastic cells. Bone. 1999. 25:255–259.

10. Hofbauer LC, Schoppet M. Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases. JAMA. 2004. 292:490–495.

11. Horwood NJ, Elliott J, Martin TJ, Gillespie MT. Osteotropic agents regulate the expression of osteoclast differentiation factor and osteoprotegerin in osteoblastic stromal cells. Endocrinology. 1998. 139:4743–4746.

12. Kong YY, Boyle WJ, Penninger JM. Osteoprotegerin ligand: a regulator of immune responses and bone physiology. Immunol Today. 2000. 21:495–502.

13. Lacey DL, Timms E, Tan HL, et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998. 93:165–176.

14. Makrygiannakis D, af Klint E, Catrina SB, et al. Intraarticular corticosteroids decrease synovial RANKL expression in inflammatory arthritis. Arthritis Rheum. 2006. 54:1463–1472.

15. Möttönen TT, Hannonen PJ, Boers M. Combination DMARD therapy including corticosteroids in early rheumatoid arthritis. Clin Exp Rheumatol. 1999. 17:6 Suppl 18. S59–S65.
16. Reid IR. Glucocorticoid osteoporosis--mechanisms and management. Eur J Endocrinol. 1997. 137:209–217.

17. Ritchlin CT, Haas-Smith SA, Li P, Hicks DG, Schwarz EM. Mechanisms of TNF-α- and RANKL-mediated osteoclastogenesis and bone resorption in psoriatic arthritis. J Clin Invest. 2003. 111:821–831.

18. Romas E, Gillespie MT, Martin TJ. Involvement of receptor activator of NFκB ligand and tumor necrosis factor-α in bone destruction in rheumatoid arthritis. Bone. 2002. 30:340–346.

19. Seshasayee D, Wang H, Lee WP, et al. A novel in vivo role for osteoprotegerin lignad in activation of monocyte effector function and inflammatory response. J Biol Chem. 2004. 279:30202–30209.
20. Takayanagi H, Iizuka H, Juji T, et al. Involvement of receptor activator of nuclear factor κB ligand/osteoclast differentiation factor in osteoclastogenesis from synoviocytes in rheumatoid arthritis. Arthritis Rheum. 2000. 43:259–269.

21. Terpos E, Szydlo R, Apperley JF, et al. Soluble receptor activator of nuclear factor κB ligand-osteoprotegerin ratio predicts survival in multiple myeloma: proposal for a novel prognostic index. Blood. 2003. 102:1064–1069.

22. Theoleyre S, Wittrant Y, Tat SK, Fortun Y, Redini F, Heymann D. The molecular triad OPG/RANK/RANKL: involvement in the orchestration of pathophysiological bone remodeling. Cytokine Growth Factor Rev. 2004. 15:457–475.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download