Abstract
Predominant involvement of a particular group of fingers due to a central nervous system lesion has been described as pseudoperipheral palsy. Two patients visited our hospital with isolated weakness of a particular group of fingers due to small cortical infarctions. A 51-year-old woman suddenly developed weakness in her left index and middle fingers. The brain MRI showed a small infarct in the right frontal cortex. A 67-year-old man was sudden difficulty using his chopsticks and had weakness in his right thumb and index finger. The brain MRI showed a small infarct in the left precentral cortex.
In 1909, Lhermitte described the predominant involvement of a particular group of fingers due to a central nervous system (CNS) lesion as "pseudoperipheral palsy" (1). Subsequently, many researchers have struggled to identify the primary motor cortex area associated with individual fingers via clinical, imaging (such as MRI, functional MRI or PET) and electrophysiologic methods (2). However, the brain motor topography of human fingers remains controversial. Here, we described two cases of isolated weakness of a particular group of fingers due to a small cortical infarction.
A 51-year-old woman with a history of medicated hypertension suddenly developed weakness in her left index and middle fingers.
On examination, she was alert and a motor examination revealed Grade IV flexion, extension, abduction, and adduction of the left index and middle fingers according to the Medical Research Council (MRC) scoring system. The strengths of the other fingers, wrist, elbow, shoulder, and leg were normal. There was no evidence of apraxia, and she had normal sensory perception in all modalities, including the affected fingers. No pain or paresthesia was felt in her fingers, all deep tendon reflexes were normal, and no pathologic reflexes were observed.
There were no significant laboratory abnormalities, and electrocardiography and echocardiography were normal. A carotid duplex scan revealed a small plaque echo in the left common carotid artery.
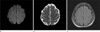
The brain MRI showed a small infarct in the right frontal cortex (Fig. 1a, b and c), the patient had not experienced any previous strokes that may have affected her strengths.
The patient was treated with antiplatelet therapy. The left finger weakness gradually improved, but she still complained of clumsiness.
A 67-year-old man presented with sudden difficulty using his chopsticks. He complained of weakness in his right thumb and index finger and was admitted to our hospital on the day of onset. He had no significant medical history, but was newly diagnosed with diabetes mellitus during his stay in the hospital. On examination, he was alert and blind from birth. Motor examination revealed mild weakness of his right thumb and index finger: extension, abduction, and adduction of the right thumb was Grade II according to MRC scoring, whereas flexion, extension, abduction, and adduction of the right index were finger Grade IV. The strengths of the other fingers, wrist, elbow, shoulder, and leg were normal. There was no evidence of apraxia. Sensations were normally perceived in all modalities, including the affected fingers. He experienced no pain or paresthesia in his fingers. His deep tendon reflexes were normal, and no pathologic reflexes were observed.
There were no significant laboratory abnormalities, and electrocardiography and echocardiography were normal. A carotid duplex scan revealed a small plaque echo in the left common carotid artery.
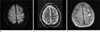
The brain MRI showed a small infarct in the left precentral cortex. (Fig. 2a, b and c)
Case 1 presented with predominant weakness of the index and middle fingers, and case 2 involved the thumb and index finger. The weakness in both cases was due to a small cortical infarction in the precentral gyrus.
Traditional views have suggested a motor homunculus in which separate regions of the primary motor cortex control each digit. This indicates that the hand-controlling area in the cerebral motor cortex is located in the middle to lower portion of the anterior wall of the central sulcus, that is, in Brodmann area 4, which is adjacent to Brodmann areas 3a and 3b-the primary sensory cortex of the hand (3).
In the previous clinical study, the involvement of a particular group of fingers due to a CNS lesion was divided into predominant involvement of the radial-side fingers and ulnar-side finger. According to the location of the lesion in MRI, a medially located lesion was related to the ulnar-side finger, whereas a laterally located lesion was related to the radial-side finger (4). Both of our cases showed predominant involvement of the radial-side fingers and involved lesions located in the lateral aspect of the precentral gyrus. These findings correspond with those of a previous functional MRI study that showed activation of the primary motor cortex area responsible for hand movement (5).
The clinical observations in the two present cases provided little evidence of separate primary motor cortex regions control of each radial-side digit. As the radial-side is located medially and the ulnar-side is located laterally, the thumb, index, and middle finger are also sequentially located lateral to medial. In the previous study using PET, movement of the index finger showed more medial activation of the cerebral cortex compared to that resulting from movement of the thumb (6).
However, this should not be viewed as a simple issue. Outputs of the hand-controlling area of the primary motor cortex tend to both converge and diverge. Outputs converge from a large territory in the hand-controlling area to exert control of single finger muscles (7), whereas outputs diverge such that single neurons can an effect multiple muscles (8). In addition, the topographical brain territories of each finger are not equal. For example, the thumb, the most functionally important digit is controlled by a large amount of the primary motor cortex and many interconnections. Also, the somatotropic gradient of the hand-controlling area is superimposed on a base of distributed activation. Thus, by function, the brain region which controls a particular finger will share or topographically overlap with the brain region for another finger (8). Therefore, it is very difficult to precisely localize a single finger-controlling region within the brain, and we can only suggest a trend of finger control.
With the development of neuroimaging and electro-physiologic technique, the topography of the brain has been studied via various methods. Further research developments will help to elucidate the topography of the brain not only at the level of isolated fingers, but for each movement of the fingers.
Figures and Tables
References
1. Kim JS. Predominant involvement of a particular group of fingers due to small, cortical infarction. Neurology. 2001; 56:1677–1682.
2. Uematsu S, Lesser R, Fisher RS, et al. Motor and sensory cortex in humans: topography studied with chronic subdural stimulation. Neurosurgery. 1992; 31:59–71. discussion 71-2.
3. White LE, Andrews TJ, Hulette C, et al. Structure of the human sensorimotor system. I: Morphology and cytoarchitecture of the central sulcus. Cereb Cortex. 1997; 7:18–30.
4. Kim JS, Chung JP, Ha SW. Isolated weakness of index finger due to small cortical infarction. Neurology. 2002; 58:985.
5. Kleinschmidt A, Nitschke MF, Frahm J. Somatotopy in the human motor cortex hand area. A high-resolution functional MRI study. Eur J Neurosci. 1997; 9:2178–2186.
6. Grafton ST, Woods RP, Mazziotta JC. Within-arm somatotopy in human motor areas determined by positron emission tomography imaging of cerebral blood flow. Exp Brain Res. 1993; 95:172–176.
7. Buys EJ, Lemon RN, Mantel GW, Muir RB. Selective facilitation of different hand muscles by single corticospinal neurones in the conscious monkey. J Physiol. 1986; 381:529–549.
8. Schieber MH. Somatotopic gradients in the distributed organization of the human primary motor cortex hand area: evidence from small infarcts. Exp Brain Res. 1999; 128:139–148.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download