Abstract
We report the case of a 16-year-old boy with a solid pancreatic mass which proved to be a nonfunctioning, malignant pancreatic neuroendocrine tumor (PNET). In pediatric patients, malignant pancreatic tumors are rare, especially malignant PNET. When dynamic contrast enhanced MRI showed a well enhancing solid pancreatic tumor on arterial and delayed phases and combined with malignant features, such as vascular invasion, invasion of adjascent organs, and lymphadenopathy, we should include malignant pancreatic neuroendocrine tumor in the differential diagnosis of childhood pancreatic tumors.
Although various types of pancreatic neoplasms have often been studied, malignant pancreatic endocrine tumors in children have rarely been reported in the medical literature. We herein present the CT and MR imaging findings, including MR cholangiopancreaticography (MRCP), of a nonfunctioning, malignant pancreatic neuroendocrine tumor (PNET) in a 16-year-old boy. To the best of our knowledge, this is the first such report in the radiology literature regarding the imaging findings of a malignant endocrine tumor of detected in a child.
A 16-year-old boy suffered from epigastric pain, radiating to the right upper quadrant of the abdomen for one year. He visited a community clinic where ultrasound examination showed a mass-like lesion in the head of the pancreas. He was referred to our hospital for further study of the pancreas. On physical examination, a palpable abdominal mass was detected in the right upper quadrant of the abdomen. The laboratory studies including those regarding pancreatic enzymes and tumor markers, were within normal limits.
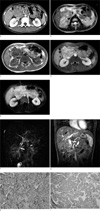
Contrast-enhanced pancreas CT scan (Fig. 1a) yielded a 4×5 cm-size solid mass with a multilobulated contour and with inhomogeneous enhancement in the head of the pancreas. Multiple enlarged lymph nodes were noted on portocaval and aortocaval spaces, and along the root of the mesentery. The pancreatic parenchyma was atrophied with upstream dilatation of the main pancreatic duct.
For further evaluation of the pancreatic mass, vascular invasion and bile duct obstruction, we performed pancreas MR imaging, including MR angiography and MRCP. The mass showed heterogeneous high signal intensity on T2-weighted image (Fig. 1b) and intermediate to low signal intensity on T1-weighted image (Fig. 1c). On dynamic T1-weighted MR image obtained after intravenous administration of gadolinium, the mass showed inhomogeneous enhancement with a reticular pattern on the portal phase (Fig. 1d) and persistent intense enhancement on the delayed phase (Fig. 1e). On MRCP, despite the large size of the mass, the common bile duct was not dilated but was displaced by the mass (Fig. 1f). Coronal T1-weighted MR image of MR angiography showed encasement of the main portal vein by the mass (Fig. 1g), which suggested its malignant features in addition to the presence of metastatic lymphadenopathy. These CT and MRI findings were indicative of a primary malignant pancreatic neoplasm.
Exploratory surgery revealed a large, hard mass in the pancreatic head and with conglomerated lymphadenopathy along the mesenteric root. As complete resection of the mass was not possible, metastatic lymph nodes were extracted.
Microscopy of the specimens revealed that the tumor cells had a trabacular pattern over fibrotic and hyaline stroma (Fig. 1h). Immunohistochemistry staining revealed positive reaction for synaptophysin, neuron-specific enolase, Chromogranin A, and Pan CK, and was weakly positive for alpha-1-antipchymotrypsin (Fig. 1i). However, there were no detected endocrine substances including insulin, glucagon, and somatostatin. According to these results, the histopathologic diagnosis of the mass was well differentiated endocrine carcinoma of the pancreas, and the confirmative diagnosis of non-functioning, malignant pancreatic neuroendocrine tumor (PNET) was thus made.
Pancreatic neoplasms are rarely seen in children. They can be divided into epithelial and nonepithelial types. Epithelial tumors may be further classified as exocrine or endocrine tumors. Exocrine tumors include acinar cell orign tumors i.e. pancreatoblastoma, acinar cell carcinoma; ductal cell orgin tumors, i.e. ductal cell adenocarcinoma; and undetermined cell origin tumors, i.e. solid-pseudopapillary tumor. Endocrine cell tumors are uncommonly encountered in older children, they can be functioning or nonfunctioning. Nonepithelial neoplasms, such as lymphoma or sarcoma, arising primarily in the pancreas are quite rare in children. Among these neoplasms, pancreatoblastoma and solid-pseudopapillary tumors often occur in children and adolescents (1).
When the imaging findings suggest malignant pancreatic tumor, pancreatoblastoma should not be ruled out in the differential diagnosis in infants and young children, as it is the most common childhood neoplasm. Pancreatoblastomas tend to be large, solitary tumors that most frequently arise from the pancreas body and/or tail or that involve the entire pancreas rather than usually being located in the pancreatic head. There may be direct extension to other abdominal organs, including the spleen, left kidney, and omentum. Hepatic metastasis, vascular encasement, and calcification are not uncommon (2). The other tumor that should be included in differential diagnosis of pancreatic tumor of children is solid pseudopapillary tumor. This tumor usually occurs in adolescent or young adult female. It is heterogeneous in internal architecture, with a complex mass of solid and cystic hemorrhagic and necrotic portions. The findings of fibrous capsule and internal hemorrhage are the features that can distinguish solid pseudopapillary tumor from other pancreatic tumors (3).
Although malignant pancreatic neuroendocrine tumors are rarely seen in children, they should be included in the differential diagnosis when a malignant pancreatic mass is suspected. Among the endocrine tumors of the pancreas occurring in children, most cases of malignant endocrine tumors are found to be functioning endocrine tumors, such as malignant insulinoma or gastrinoma (4). Endocrine tumor of the pancreas can be associated with inherited disease processes such as multiple endocrine neoplasia (MEN type 1) and von Hippel-Lindau disease. As suggested by the recent studies that have reported the detection of only two cases of tuberous sclerosis complex in children (5, 6), there are very few reported studies of this tumor occurring in children.
Nonfunctioning PNET is pathologically indistinguishable from functioning PNET, both of which are distinguished by the clinical or biochemical evidence of hormone hypersecretion. Widely accepted CT findings of nonfunctioning PNET include the following: a well-defined pancreatic mass of an unusually large size; moderate to strong enhancement seen on the arterial phase for either primary or hepatic metastases; well-enhanced lymph node enlargement; and frequent vascular encasement (7, 8).
Differentiating benign from malignant PNET is not easy. Distinctions between benign and malignant tumors can be made based on the tumor size, lymph node involvement, and the presence of distant metastasis. Tumors are also considered to be malignant if there is any histologic evidence of vascular, lymphatic or perineural invasion.
On CT and MRI scans of our patient, the initial differential diagnosis of this tumor included pancreatoblastoma and malignant PNET due to its solid nature, the absence of cystic or necrotic portions, its heterogeneous enhancement pattern, lymph node metastasis, and the portal vein encasement. It has been reported that some pancreatic endocrine tumors show delayed contrast enhancement on dynamic CT, caused by the presence of tumor thrombi in the veins around the mass (9). Some researchers have reported that delayed phase T1-weighted MR imaging is useful for the detection of PNET, especially scirrhous type (10). In our case, the delayed phase of dynamic MRI showed persistent inhomogeneous enhancement of both the pancreatic mass and the metastatic lymph nodes. Considering this dynamic MR imaging findings, the diagnosis was closer to that of malignant PNET.
The treatment of malignant PNET is primarily by surgical resection, especially for unmetastasized tumors; unfortunately, as surgical resection was not feasible in our case due to extensive peritoneal and retroperitoneal adhesion of tumor and metastatic lymphadenopathy, only biopsies of the mass and lymph node were performed.
In conclusion, the imaging findings of malignant PNET in pediatric patients do not different from those of adult onset ones, as a solid pancreatic mass with malignant features such as vascular invasion, adhesion to adjascent organs or lymph node metastasis. The dynamic MR imaging might be useful in the differential diagnosis of malignant pancreatic tumors occurring in children. A larger number of reported cases, as well as analysis of malignant PNET will be needed in order to further advance our understanding of the imaging features of this tumor.
Figures and Tables
Fig. 1
A 16-year-old boy with epigastric pain for one year.
(a) Ccontrast-enhanced CT of the abdomen showed a multilobulated contoured solid mass with inhomogeneous enhancement in the region of the pancreatic head (arrows).
(b) The axial T2-weighted MR image obtained at the same level as in A, depicts a multilobulated mass with heterogeneous high signal intensity.
(c) The mass showed intermediate to low signal intensity on the axial T1-weighted MR image.
(d) The mass revealed a reticular pattern of inhomogeneous enhancement on the late portal phase of dynamic gadolinium-enhanced T1-weighted MR image.
(e) On the delayed phase of dynamic gadolinium-enhanced T1-weighted MR image, persistent and intense enhancement of the mass and multiple conglomerated metastatic lymphadenopathy were seen (arrow).
(f) MRCP showed that despite the large size of the mass, the common bile duct (arrow) was not dilated but had been displaced by the mass.
(g) The coronal T1-weighted MR image showed encasement of the main portal vein (arrow) by the mass, which suggested the malignant features of the mass.
(h) Photomicrograph (H & E,×400 ) of the specimen showed that the tumor cells had a trabacular pattern over the fibrotic and hyaline stroma.
(i) The immunohistochemistry staining for synaptophysin (×100) was positive which confirmed the diagnosis of malignant endocrine neoplasm of the pancreas.
References
1. Shorter NA, Glick RD, Klimstra DS, Brennan MF, LaQuaglia MP. Malignant pancreatic tumors in childhood and adolescence: the Memorial Sloan-Kettering experience, 1967 to present. J Pediatr Surg. 2002. 37:887–892.
2. Roebuck DJ, Yuen MK, Wong YC, Shing MK, Lee CW, Li CK. Imaging features of pancreatoblastoma. Pediatr Radiol. 2001. 31:501–506.
3. Chung EM, Travis MD, Conran RM. Pancreatic tumors in children: radiologic-pathologic correlation. Radiographics. 2006. 26:1211–1238.
4. Grosfeld JL, Vane DW, Rescorla FJ, McGuire W, West KW. Pancreatic tumors in childhood: analysis of 13 cases. J Pediatr Surg. 1990. 25:1057–1062.
5. Verhoef S, van Diemen-Steenvoorde R, Akkersdijk WL, Bax NM, Ariyurek Y, Hermans CJ, et al. Malignant pancreatic tumour within the spectrum of tuberous sclerosis complex in childhood. Eur J Pediatr. 1999. 158:284–287.
6. Francalanci P, Diomedi-Camassei F, Purificato C, Santorelli FM, Giannotti A, Dominici C, et al. Malignant pancreatic endocrine tumor in a child with tuberous sclerosis. Am J Surg Pathol. 2003. 27:1386–1389.
7. Procacci C, Carbognin G, Accordini S, Biasiutti C, Bicego E, Romano L, et al. Nonfunctioning endocrine tumors of the pancreas: possibilities of spiral CT characterization. Eur Radiol. 2001. 11:1175–1183.
8. Stafford-Johnson DB, Francis IR, Eckhauser FE, Knol JA, Chang AE. Dual-phase helical CT of nonfunctioning islet cell tumors. J Comput Assist Tomogr. 1998. 22:335–339.
9. Koito K, Namieno T, Nagakawa T, Morita K. Delayed enhancement of islet cell carcinoma on dynamic computed tomography: a sign of its malignancy. Abdom Imaging. 1997. 22:304–306.
10. Ichikawa T, Peterson MS, Federle MP, Baron RL, Haradome H, Kawamori Y, et al. Islet cell tumor of the pancreas: biphasic CT versus MR imaging in tumor detection. Radiology. 2000. 216:163–171.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download