Abstract
Plant root hairs are commonly found artifacts in parasitology specimens and may be confused with helminthes by an untrained eye. We report a case of brain tuberculoma where the tissue sample was contaminated with root hair derived from tap water; the presence of this root hair, which mimicked a larva, led to diagnostic confusion. Therefore, tap water should be considered a source of root hair and vegetable matter.
Root hair from plants and vegetable matter are commonly seen in inherently contaminated clinical samples such as stool [1] and sputum [2]. Sterile samples such as aseptically collected pus or tissues are free of such artifacts. However, the presence of artifacts in samples may be due to the rare instances of basal laboratory media contamination during tissue processing. We present a cautionary report stating that the contamination of brain tissue smears with unfiltered tap water may lead to unnecessary further processing and investigation.
A 57-yr-old man under treatment for tuberculous meningitis and hydrocephalus was admitted to a tertiary care hospital for the investigation of drowsiness. He had been receiving first-line anti-tubercular treatment (ATT) and 10 mg/day of prednisone for the past 5 months. The initial workup included a head computed tomography scan, which showed multiple small space-occupying lesions in both the cerebellar hemispheres. The baseline laboratory workup included complete blood count, urinalysis, and stool examination, the results of which were normal. A craniotomy and biopsy were also performed, and the perioperative findings were unremarkable. The tissue samples were sent for histopathological examination and bacterial and mycological culturing. Microscopic examination of a potassium hydroxide (KOH)-treated tissue sample showed a single 2-mm structure resembling a "worm," with tapering ends. The same tissue sample was re-examined for this structure by teasing the tissue with a sterile scalpel and suspending it in sterile saline; similar segments were seen (Fig. 1), and a diagnostic dilemma arose as to the identification of these structures. The initial histopathological findings had not revealed similar structures. Therefore, we re-examined various areas in the tissue by repeated sectioning. Meanwhile, the attending physician was alerted to the possibility of Central Nervous System (CNS) helminthosis, and an empiric treatment with 200 mg/day of albendazole was initiated, while additional clues to a disseminated helminthic infection were sought. However, the peripheral smear examination did not reveal eosinophilia, and the 3 stool examinations were negative for larvae. No such helminth-like structure was seen on examination of the different sections of the tissue, although minimal inflammatory cells and reactive glial cells were noted. Finally, parasitic infestation was ruled out, the albendazole treatment was discontinued, and the patient was tentatively diagnosed with a tuberculoma.
As a follow-up of the initial diagnostic confusion, saline and KOH blanks were examined for similar structures. Structures with an internal canal and tapering ends of lengths varying between 2 mm and 5 mm were found in the media and in tap water from the microbiology laboratory. These structures were distinct root hair artifacts (Fig. 2). Gram-stained slides that had been washed under laboratory tap water showed similar structures, which had been disregarded as artifacts by microscopists. On further investigation, it was found that the laboratory filtration unit had malfunctioned. However, the hospital tap water and water in other laboratories in the hospital were free of such root hair. We thought that construction work that involved the laying of water pipes near the microbiology laboratory might have led to the appearance of such the root hair in tap water. Surprisingly enough, the plant material persisted even after autoclaving. When the unit was repaired, the water was free of these structures.
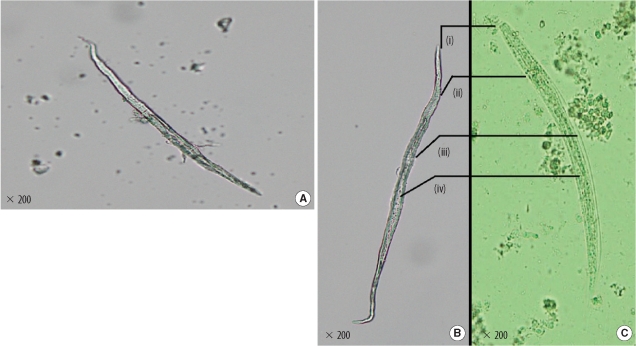
Root hairs are commonly found artifacts in parasitology laboratories [3]. The presence of root hair in clinical material is known to create diagnostic confusion, because the hair strands mimic nematode larvae [4]. However, medical and botanical literature is devoid of reports citing tap water as a source of plant root hair. In this paper, we report tap water as the source of plant root hair. Although parasites themselves also contaminate tap water, the morphological appearance of the structures in our case is representative of root hair and not nematode larvae (Fig. 2B). Worms have both internal and external structures, such as an intestine and mouthparts, which differentiate them from root hair [2]. When the patient's tissue was carefully observed for the doubtful structures, an elongated central refractile core suggestive of a root hair was seen (Fig. 1B).
In general, all basal media in microbiology laboratories are autoclaved prior to use. Although we ensured sterile processing, 2 factors led to the contamination of the sample. First, the malfunctioning of the filtration unit, and second, the homogenization of the sample with saline during its processing. Although saline suspension of tissue samples is not recommended, we carried it out since the sample received by the laboratory was dry and its quantity was insufficient for all the tests requested. To prevent such occurrences, clinicians sending tissues to a laboratory for testing must ensure adequate tissue quantity and suspend the tissue in an appropriate medium while the laboratory staff must strive to minimize contamination of basal media and samples to prevent errors. Fortunately, since the problem was pursued and remedied in time in our laboratory, we did not experience similar problems with the other samples.
In our case, a provisional diagnosis of CNS helminthosis was considered only because of the patient's unusual history and physical examination findings, including a slow response to ATT, cerebrospinal fluid shunting, and steroid therapy, and the absence of focal neurological findings. The tissue structures seen in Fig. 1 were not diagnostic; however, the possibility of other infections such as disseminated strongyloidiasis and angiostrongylus remained.
Additional investigations and histopathological sections stained and examined at a different laboratory in the hospital, which had a separate water supply, aided us in excluding the diagnosis of CNS helminthosis.
In conclusion, root hair from plant and vegetable matter are artifacts frequently encountered in laboratory practice. Other than clinical material, unfiltered tap water should also be considered an important source of root hair. Further, laboratories are advised to use filtered distilled water in all wet-lab experiments.
References
1. Paul Y. Undigested food presenting as bizarre objects in stool. Indian Pediatr. 1994; 31:1130–1131. PMID: 7883378.
2. Martínez-Girón R, González-López JR, Esteban JG, García-Miralles MT, Alvarez-de-los-Heros C, Ribas-Barceló A. Worm-like artifacts in exfoliative cytology. Diagn Cytopathol. 2006; 34:636–639. PMID: 16900479.

3. Isenberg HD, editor. Clinical microbiology procedures handbook. 2004. 2nd ed. Washington DC: ASM Press.
4. Garcia LS, Bruckner DA, editors. Diagnostic medical parasitology. 1997. 3rd ed. Washington DC: ASM Press;p. 749.
Fig. 1
Wet mount preparation with saline of 2 separate sections of the brain tissue. The structures shown created a diagnostic dilemma as such artifacts are not common: (A) shows 2 distorted structures that have a definite outer covering but no pseudocoelom, (B) shows a tapering structure with a central refractile core, more suggestive of a root hair.

Fig. 2
(A) Microscopic examination of the root hair found in the laboratory tap water. Differences between root hair (B) and Strongyloides rhabditiform larvae (C): (i) mouth parts with distinct papilla in the nematode larva but indistinct in the root hair; (ii) the nerve ring apparent in the larva but absent in the root hair; (iii) the distinct cuticle and pseudocoelom found in the nematode larva is absent in the root hair, thereby resulting in a distorted morphology; (iv) the empty, rough conducting channel seen in the root hair must not be confused with nematode intestines.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download