Abstract
Background
Gonadotropin-releasing hormone (GnRH) stimulation test is the gold standard to identify central precocious puberty (CPP). This test requires multiple blood samples at different time points to measure gonadotropin levels, and is therefore expensive, time-consuming, and uncomfortable for patients. We aimed to simplify the GnRH stimulation test to require fewer blood samples.
Methods
A study of 166 girls with precocious puberty was undertaken. Blood samples were obtained at 0, 15, 30, 45, 60, 90, and 120 min after GnRH administration, and the levels of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) were measured. For each parameter, the sensitivities and specificities were estimated and ROC curves were constructed.
Results
One hundred and twenty-eight patients (77.1%) were diagnosed for CPP. Peak LH levels were achieved 30 min after GnRH stimulation in patients with CPP. Further, 98.4% of the 45-min samples were diagnostic for CPP, and the cumulative frequency of LH values of ≥5 IU/L was 100% at 45 min. Using this cut-off value for LH, the ROC curve for LH at 45 min showed the highest sensitivity (98.4%) and specificity (100%) in the diagnosis of CPP.
Precocious puberty is generally defined as the onset of secondary sexual characteristics before 8 yr of age in girls and 9 yr in boys [1, 2]. Precocious puberty caused by central activation of the hypothalamic-pituitary-gonadal (HPG) axis is classified as gonadotropin-dependent, also called true or central precocious puberty (CPP). Isolated forms of abnormal pubertal development, such as premature thelarche (PT), are not associated with central activation of the HPG axis, and are classified as peripheral precocious puberty [3]. CPP occurs at least 10-fold more frequently in girls than in boys; in more than 90% of girls, sexual precocity is idiopathic. It is estimated that precocious puberty affects between 1 in 5,000 and 1 in 10,000 children, and a recent study found a much higher incidence of CPP in girls compared with 40 yr ago [4, 5]. Gonadotropin releasing hormone (GnRH) stimulation test is the gold standard to reveal premature activation of the HPG axis in patients with precocious puberty [6, 7].
However, this test needs a clinical investigation unit and repeated blood sampling at 5-8 time points to measure the levels of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) [8-10]. The duration of the test is 90 to 120 min and 15 to 25 mL of blood is required. As such, this test is comparatively expensive, time-consuming, painful, and uncomfortable for patients [9-13]. To avoid these problems, several alternatives such as measurement of basal LH and FSH values, and the subcutaneous leuprolide acetate test with a single sample, have been attempted [14-17]. None of these alternative methods have yet been standardized sufficiently or proven equal or superior to GnRH test.
In this study, we investigated whether the GnRH test to diagnose CPP could be simplified without altering its validity, by estimating the values of LH and FSH at various time points after GnRH stimulation.
This retrospective study was done by reviewing the medical records of 166 girls, who presented at our hospital with early pubertal signs between July 2009 and December 2010. Evaluation included documentation of the history of onset and the progression of pubertal changes, including Tanner breast and pubic hair stages, height and weight, growth velocity, and GnRH stimulation testing. The indication for the GnRH stimulation test was the onset of pubertal signs before the age of 8 yr in girls. Bone age was evaluated by the method of Greulich and Pyle [18]. This study was approved by the Institutional Review Board of our hospital.
A standard dose of 100 µg GnRH (Relefact; Sanofi-Aventis, Frankfurt am Main, Germany) was administered as an intravenous (IV) bolus. An IV cannula was inserted and blood samples were obtained immediately before the injection and at 15, 30, 45, 60, 90, and 120 min after the injection.
Serum LH, FSH, and estradiol were measured by direct chemiluminescence, using the ADVIA Centaur® Immunoassay System (Siemens Healthcare Diagnostics Inc., Tarrytown, NY, USA). The detection limits for LH, FSH, and estradiol were 0.07 IU/L, 0.6 IU/L, and 10 pmol/L, respectively. A stimulated LH value of ≥5 IU/L was considered diagnostic for CPP in patients with pubertal signs [19]. Patients who had a stimulated LH value of <5 IU/L were classified as having PT.
Statistical analyses were performed using the Statistical Package for Social Sciences software package for Windows (version 19.0; SPSS Inc., Chicago, IL, USA). Values are reported as median, range, and frequency. The Mann-Whitney U test was used to compare medians. A P value of less than 0.05 was considered statistically significant. The diagnostic values of LH and FSH, as well as the LH/FSH ratio at different time points during the GnRH test, were evaluated using ROC curve analysis.
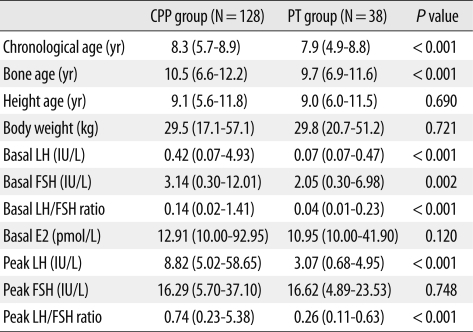
A hundred and sixty-six girls showing signs of early puberty were enrolled in this study. In 128 out of 166 (77.1%) tests, the peak LH was above the cut-off value, and the test was diagnostic for CPP. The remaining girls (38 out of 166, 22.9%) had an evoked LH value of <5 IU/L, and were classified as undergoing PT. The clinical characteristics and hormone levels of both groups are presented in Table 1. Significant differences in chronological age and bone age were seen between the 2 groups. Basal LH and FSH levels in the group with CPP were higher than in the PT group (P<0.001). Evaluation of the LH/FSH ratio demonstrated statistically significant differences between the CPP and PT groups in both the basal and peak states (P<0.001).
Basal and peak LH levels in the CPP group were significantly greater than in the PT group (both, P<0.001) (Fig. 1). Both LH and FSH levels showed a rapid increase after GnRH stimulation in both groups. However, the increase in LH levels was more robust in the CPP group than in the PT group, while FSH responses were almost similar in the 2 groups (Fig. 2). The peak LH level was achieved 30 min after GnRH stimulation in the CPP group, and 45 min after stimulation in the PT group, while FSH levels continued to rise throughout the test in both groups (Fig. 2). The peak LH/FSH ratio was achieved in 15 min after GnRH stimulation in both groups (Fig. 2).
Among CPP patients, 98.4% (126 of 128 tests) of the 45-min samples were diagnostic for CPP, with LH levels of ≥5 IU/L (Table 2). The 2 patients who had stimulated LH levels below this cut-off value at 45 min, showed values above the cut-off at 30 min. Therefore, the cumulative frequency of LH values of ≥5 IU/L at 45 min was 100% (Table 2).
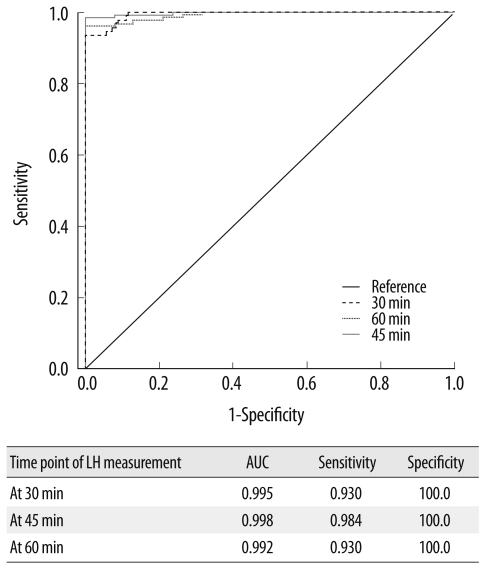
Fig. 3 shows the ROC curves of LH at different time points of the test in all patients. The ROC curve for LH at 45 min had the greatest area under the curve (AUC) and was hence the most effective for diagnosing CPP. 5 IU/L was considered as the cut-off value for LH in the diagnosis of CPP, and the ROC curves indicate that an LH cut-off value of 4.99 IU/L at 45 min resulted in high sensitivity (98.4%) and specificity (100%) in the diagnosis of CPP.
Pubertal development of breast was compatible with Tanner stages 2 and 3 in 89.8% and 10.2% of girls, respectively. Pubic hair development was compatible with Tanner stages 1 and 2 in 95.2% and 4.8% of girls, respectively.
CPP may have profound physical and psychological effects on affected children and their families. It is accompanied by growth acceleration, advancement of bone age, and elevated sex steroid hormone levels for age. Therefore, early menarche and significant impairment of final height can result in untreated CPP patients. Mogensen et al. [20] reported a significant increase in the number of CPP patients over the recent 16-yr period. This trend also occurred in Korean girls with a 4- to 5-fold increment in CPP patients over the recent 5-yr period.
The gold standard for laboratory confirmation of CPP is the GnRH stimulation test. However, this test results in significant cost, time, and discomfort to the patient, as well as a potential delay in diagnosis. Different cut-off levels of peak LH are used in the diagnosis of CPP in patients with early pubertal signs. Further, cut-off levels differ depending on the assays used to measure LH [19, 21, 22]. Previous studies have suggested that GnRH-stimulated LH levels between 30 and 60 min are sufficient to diagnose CPP [21, 23-25]. In Korean patients with CPP, Choi et al. [26] tried to simplify the standard GnRH stimulation test, suggesting that a single LH determination at 30 min was diagnostic. However, their study included only 33 patients. We analyzed the LH and FSH response to the GnRH test performed to diagnose precocious puberty in 166 patients. To our best knowledge, the current study comprises the largest homogeneous Korean population in this regard.
We measured LH levels by a direct chemiluminescence immunoassay, and a cut-off stimulated LH value of ≥5 IU/L was considered diagnostic for CPP. In 126 out of 128 (98.4%) CPP patients tested, 45-min samples were diagnostic for CPP using this cut-off value. The remaining 2 patients, who had stimulated LH levels of <5 IU/L at 45 min, had levels above the cut-off value at 30 min. Therefore, the cumulative frequency of LH values of ≥5 IU/L at 45 min was 100%. These findings indicate that 2 samples, obtained at 30 and 45 min after stimulation, can be used in a simplified GnRH test for discriminating patients with CPP from patients with PT.
Neely et al. [14] reported that a basal LH value of >0.1 IU/L was diagnostic for CPP with 94% sensitivity and 88% specificity. They also reported that a cut-off value of >0.3 IU/L increased the specificity to 100%, although the sensitivity decreased. In our study, the basal LH level in the group with CPP was higher than that of the PT group (0.42 and 0.07 IU/L, respectively, P<0.001). However, analysis of the diagnostic value of basal LH using ROC curves revealed low sensitivity and specificity of basal LH levels for the diagnosis of CPP (data not shown). In addition, we observed that 29 of the 128 patients (22.7%) diagnosed with CPP by the GnRH stimulation test showed undetectable basal LH levels (LH <0.07 IU/L). Therefore, an elevated basal LH was highly predictive of a positive GnRH test result, whereas a low basal LH did not eliminate central pubertal activation. We, therefore, do not suggest using basal LH as a single diagnostic marker for CPP.
The LH/FSH ratio has been considered of value by some investigators who reported that CPP in girls is usually accompanied by an LH/FSH ratio of >1.0 [8]. However, nocturnal LH secretion and the LH response to GnRH may be low at early puberty, and the LH/FSH ratio may remain low until mid-puberty [22]. Changes in this ratio at puberty are predominantly caused by increasing LH levels. In the current study, LH/FSH ratio showed significant differences between CPP and PT groups in both basal and peak states (P<0.001), with the basal and peak LH/FSH ratios being 0.14 and 0.74, respectively, in the CPP group. A basal LH/FSH ratio of >0.2 as a diagnostic tool for CPP had a sensitivity of 36.7% and specificity of 97.4%. With an LH/FSH ratio cut-off value of >0.5, the diagnostic sensitivities at 15, 30, 45, 60, 90, and 120 min were 77.3%, 76.6%, 69.5%, 57.8%, 40.1%, and 35.2%, respectively, while the specificities were 94.7%, 97.4%, 98.9%, 100%, 100%, and 100%, respectively. The limited utility of the LH/FSH ratio seems to be a consequence of the considerable variation of FSH levels seen in patients.
Although FSH concentrations were also observed to increase in response to the GnRH stimulation test in this study, FSH levels were indistinguishable between the CPP and PT groups, and the peak FSH levels of the CPP and PT groups had poor diagnostic value (P=0.748). Data from other studies also indicate that FSH has poor diagnostic value for CPP [10, 14, 21]. Therefore, FSH concentrations cannot be used in diagnosing CPP, and the accurate measurement of LH suffices for diagnostic confirmation of CPP.
In conclusion, a single LH value measured 45 min after GnRH stimulation is highly sensitive and specific for the diagnosis of CPP, using a cut-off value of ≥5 IU/L. Two samples, obtained at 30 and 45 min after stimulation, were able to accurately diagnose all CPP patients in this study. Although further study is required, we suggest that repeated blood sampling is unnecessary, and that 2 samples obtained at 30 and 45 min can be used in a simplified GnRH test to evaluate precocious puberty. The proposed simplified test will substantially diminish the cost and discomfort to the patient.
References
1. Parent AS, Teilmann G, Juul A, Skakkebaek NE, Toppari J, Bourguignon JP. The timing of normal puberty and the age limits of sexual precocity: variations around the world, secular trends, and changes after migration. Endocr Rev. 2003; 24:668–693. PMID: 14570750.

2. Carel JC, Eugster EA, Rogol A, Ghizzoni L, Palmert MR, Antoniazzi F, et al. Consensus statement on the use of gonadotropin-releasing hormone analogs in children. Pediatrics. 2009; 123:e752–e762. PMID: 19332438.

3. Lee PA. Central precocious puberty. An overview of diagnosis, treatment, and outcome. Endocrinol Metab Clin North Am. 1999; 28:901–918. PMID: 10609126.
4. Partsch CJ, Heger S, Sippell WG. Management and outcome of central precocious puberty. Clin Endocrinol (Oxf). 2002; 56:129–148. PMID: 11874402.

5. Brito VN, Batista MC, Borges MF, Latronico AC, Kohek MB, Thirone AC, et al. Diagnostic value of fluorometric assays in the evaluation of precocious puberty. J Clin Endocrinol Metab. 1999; 84:3539–3544. PMID: 10522992.

6. Lee PA. Laboratory monitoring of children with precocious puberty. Arch Pediatr Adolesc Med. 1994; 148:369–376. PMID: 8148936.

7. Poomthavorn P, Khlairit P, Mahachoklertwattana P. Subcutaneous gonadotropin-releasing hormone agonist (triptorelin) test for diagnosing precocious puberty. Horm Res. 2009; 72:114–119. PMID: 19690429.

8. Pescovitz OH, Hench KD, Barnes KM, Loriaux DL, Cutler GB Jr. Premature thelarche and central precocious puberty: the relationship between clinical presentation and the gonadotropin response to luteinizing hormone-releasing hormone. J Clin Endocrinol Metab. 1988; 67:474–479. PMID: 3137242.

9. Parker KL, Baens-Bailon RG, Lee PA. Depot leuprolide acetate dosage for sexual precocity. J Clin Endocrinol Metab. 1991; 73:50–52. PMID: 1904452.

10. Cavallo A, Richards GE, Busey S, Michaels SE. A simplified gonadotrophin-releasing hormone test for precocious puberty. Clin Endocrinol (Oxf). 1995; 42:641–646. PMID: 7634506.

11. Toguchi H. Pharmaceutical manipulation of leuprorelin acetate to improve clinical performance. J Int Med Res. 1990; 18(Suppl 1):35–41. PMID: 2138986.
12. Manasco PK, Pescovitz OH, Hill SC, Jones JM, Barnes KM, Hench KD, et al. Six-year results of luteinizing hormone releasing hormone (LHRH) agonist treatment in children with LHRH-dependent precocious puberty. J Pediatr. 1989; 115:105–108. PMID: 2661787.

13. Witchel SF, Baens-Bailon RG, Lee PA. Treatment of central precocious puberty: comparison of urinary gonadotropin excretion and gonadotropin-releasing hormone (GnRH) stimulation tests in monitoring GnRH analog therapy. J Clin Endocrinol Metab. 1996; 81:1353–1356. PMID: 8636332.

14. Neely EK, Wilson DM, Lee PA, Stene M, Hintz RL. Spontaneous serum gonadotropin concentrations in the evaluation of precocious puberty. J Pediatr. 1995; 127:47–52. PMID: 7608810.

15. Houk CP, Kunselman AR, Lee PA. Adequacy of a single unstimulated luteinizing hormone level to diagnose central precocious puberty in girls. Pediatrics. 2009; 123:e1059–e1063. PMID: 19482738.

16. Houk CP, Kunselman AR, Lee PA. The diagnostic value of a brief GnRH analogue stimulation test in girls with central precocious puberty: a single 30-minute post-stimulation LH sample is adequate. J Pediatr Endocrinol Metab. 2008; 21:1113–1118. PMID: 19189683.

17. Ibáñez L, Potau N, Zampolli M, Virdis R, Gussinyé M, Carrascosa A, et al. Use of leuprolide acetate response patterns in the early diagnosis of pubertal disorders: comparison with the gonadotropin-releasing hormone test. J Clin Endocrinol Metab. 1994; 78:30–35. PMID: 7507123.

18. Greulich WW, Pyle SI, editors. Radiographic atlas of skeletal development of the hand and wrist. 1959. 2nd ed. Stanford: Stanford University Press.
19. Neely EK, Hintz RL, Wilson DM, Lee PA, Gautier T, Argente J, et al. Normal ranges for immunochemiluminometric gonadotropin assays. J Pediatr. 1995; 127:40–46. PMID: 7608809.

20. Mogensen SS, Aksglaede L, Mouritsen A, Sørensen K, Main KM, Gideon P, et al. Diagnostic work-up of 449 consecutive girls who were referred to be evaluated for precocious puberty. J Clin Endocrinol Metab. 2011; 96:1393–1401. PMID: 21346077.

21. Resende EA, Lara BH, Reis JD, Ferreira BP, Pereira GA, Borges MF. Assessment of basal and gonadotropin-releasing hormone-stimulated gonadotropins by immunochemiluminometric and immunofluorometric assays in normal children. J Clin Endocrinol Metab. 2007; 92:1424–1429. PMID: 17284632.

22. Oerter KE, Uriarte MM, Rose SR, Barnes KM, Cutler GB Jr. Gonadotropin secretory dynamics during puberty in normal girls and boys. J Clin Endocrinol Metab. 1990; 71:1251–1258. PMID: 2121771.

23. Eckert KL, Wilson DM, Bachrach LK, Anhalt H, Habiby RL, Olney RC, et al. A single-sample, subcutaneous gonadotropin-releasing hormone test for central precocious puberty. Pediatrics. 1996; 97:517–519. PMID: 8632938.

24. Cavallo A, Zhou XH. LHRH test in the assessment of puberty in normal children. Horm Res. 1994; 41:10–15. PMID: 8013936.

25. Brito VN, Latronico AC, Arnhold IJ, Mendonca BB. A single luteinizing hormone determination 2 hours after depot leuprolide is useful for therapy monitoring of gonadotropin-dependent precocious puberty in girls. J Clin Endocrinol Metab. 2004; 89:4338–4342. PMID: 15356030.

26. Choi JH, Shin YL, Yoo HW. The predictive factors for central nervous system lesion in central precocious puberty and the utility of single timed LH after GnRH administration. J Korean Soc Pediatr Endocrinol. 2002; 7:206–214.
Fig. 1
Comparison of basal (A) and peak (B) LH concentrations following GnRH stimulation, between CPP patients (N=128) and PT patients (N=38). Box-plots show the median, interquartile range, outliers, and extreme cases.
Abbreviations: LH, luteinizing hormone; GnRH, gonadotropin releasing hormone; CPP, central precocious puberty; PT, premature thelarche.
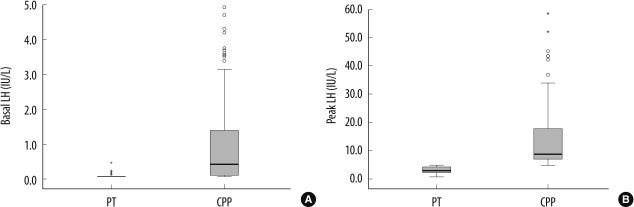
Fig. 2
Comparison of (A) LH and (B) FSH concentrations, and (C) LH/FSH ratio, versus time after GnRH stimulation, between CPP and PT patients. Data are presented as median and 95% CI.
Abbreviations: LH, luteinizing hormone; FSH, follicle-stimulating hormone; GnRH, gonadotropin releasing hormone; CPP, central precocious puberty; PT, premature thelarche; CI, confidence intervals.
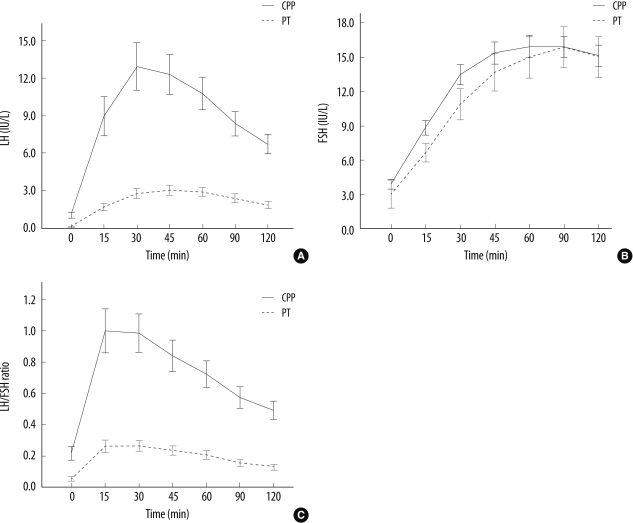
Fig. 3
ROC curves of LH at different time points (30, 45, and 60 min) after stimulation in all patients.
Abbreviations: LH, luteinizing hormone; AUC, area under curve; ROC, receiver operating characteristic.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download