Abstract
Background:
Maternal serum prenatal quadruple screening includes testing for alpha-fetoprotein (AFP), human chorionic gonadotrophin (hCG), unconjugated estriol (uE3), and dimeric inhibin A (DIA). We evaluated quadruple screening using an automated platform and looked for any ethnic differences in the median values of each marker.
Methods:
We measured the concentrations of each quadruple test analyte using the UniCel DxI 800 system (Beckman Coulter, USA) in 788 Korean mid-trimester maternal serum samples and calculated their median values using Benetech software (Benetech, Canada). We also compared the results with those obtained using the Immulite 2000 assay (Siemens Healthcare Diagnostics, USA) or ELISA (DSL, USA) in 442 samples.
Results:
We obtained mid-trimester median values for each marker. The following are the comparative results for each test using the Immulite 2000 assay or ELISA (x) and the UniCel DxI 800 immunoassay (y): AFP, y=1.10x+0.01, r=0.925; uE3, y=0.28x+0.24, r=0.885; hCG, y=1.22x-3047.8, r=0.944; and DIA, y=0.86x+15.31, r=0.833. Assay results for each of the four markers showed good correlations. However, significant biases necessitated new median calculations of prenatal risk estimates in all four tests.
Conclusions:
We established gestational age-specific second-trimester median values for four markers in Korean samples using the UniCel DxI 800 immunoassay system. Despite significant bias, there were good correlations between the results obtained using the UniCel DxI 800 immunoassay and those obtained using the Immulite 2000 assay.
Go to : 
REFERENCES
1.Wald NJ., Cuckle HS., Densem JW., Nanchahal K., Royston P., Chard T, et al. Maternal serum screening for Down's syndrome in early pregnancy. BMJ. 1988. 297:883–7.

2.Egan JF., Kaminsky LM., DeRoche ME., Barsoom MJ., Borgida AF., Benn PA. Antenatal Down syndrome screening in the United States in 2001: a survey of maternal-fetal medicine specialists. Am J Obstet Gynecol. 2002. 187:1230–4.

3.Kim JM., Sim AS., Lee EH. Amniotic chromosomal analysis in pregnant women identified by triple-marker testing as screen positive. Korean J Lab Med. 2006. 26:123–30.

4.Kim SK., Bai SW., Chung JE., Jung YN., Park KH., Cho DJ, et al. Triple marker screening for fetal chromosomal abnormalities in Korean women of advanced maternal age. Yonsei Med J. 2001. 42:199–203.

5.Han SH., An JW., Jeong GY., Yoon HR., Lee A., Yang YH, et al. Clinical and cytogenetic findings on 31,615 mid-trimester amniocenteses. Korean J Lab Med. 2008. 28:378–85.

6.Benn PA., Kaminsky LM., Ying J., Borgida AF., Egan JF. Combined second-trimester biochemical and ultrasound screening for Down syndrome. Obstet Gynecol. 2002. 100:1168–76.

7.Benn PA., Fang M., Egan JF., Horne D., Collins R. Incorporation of inhibin-A in second-trimester screening for Down syndrome. Obstet Gynecol. 2003. 101:451–4.

8.Wald NJ., Rodeck C., Hackshaw AK., Rudnicka A. SURUSS in perspective. Semin Perinatol. 2005. 29:225–35.

9.Lambert-Messerlian GM., Palomaki GE., Canick JA. Inhibin A measurement using an automated assay platform. Prenat Diagn. 2008. 28:399–403.

10.National Committee for Clinical Laboratory Standards. Evaluation of precision performance of quantitative measurement methods: approved guideline. NCCLS document EP5-A2. 2nd ed.Wayne, PA: NCCLS;2004.
11.Rawlins ML., La'ulu SL., Erickson JA., Roberts WL. Performance characteristics of the Access Inhibin A assay. Clin Chim Acta. 2008. 397:32–5.

12.Erickson JA., Ashwood ER., Gin CA. Evaluation of a dimeric inhibin-A assay for assessing fetal Down syndrome: establishment, comparison, and monitoring of median concentrations for normal pregnancies. Arch Pathol Lab Med. 2004. 128:415–20.

13.Vranken G., Reynolds T., Van Nueten J. Medians for second-trimester maternal serum markers: geographical differences and variation caused by median multiples-of-median equations. J Clin Pathol. 2006. 59:639–44.

14.Crandall BF., Lebherz TB., Schroth PC., Matsumoto M. Alpha-fetoprotein concentrations in maternal serum: relation to race and body weight. Clin Chem. 1983. 29:531–3.

15.Bogart MH., Jones OW., Felder RA., Best RG., Bradley L., Butts W, et al. Prospective evaluation of maternal serum human chorionic gonadotropin levels in 3428 pregnancies. Am J Obstet Gynecol. 1991. 165:663–7.

16.Simpson JL., Elias S., Morgan CD., Shulman L., Umstot E., Andersen RN. Second trimester maternal serum human chorionic gonadotropin and unconjugated oestriol levels in blacks and whites. Lancet. 1990. 335:1459–60.

17.Benn PA., Clive JM., Collins R. Medians for second-trimester maternal serum alpha-fetoprotein, human chorionic gonadotropin, and unconjugated estriol; differences between races or ethnic groups. Clin Chem. 1997. 43:333–7.
18.Benn PA., Collins R. Evaluation of effect of analytical imprecision in maternal serum screening for Down's syndrome. Ann Clin Biochem. 2001. 38:28–36.
19.Serdar MA., Tutuncu L., Olgun A., Hasimi A., Ozgurtas T., Erbil MK. The effects of analytical factors on second trimester risk estimations. Int J Gynaecol Obstet. 2006. 93:28–32.

Go to : 
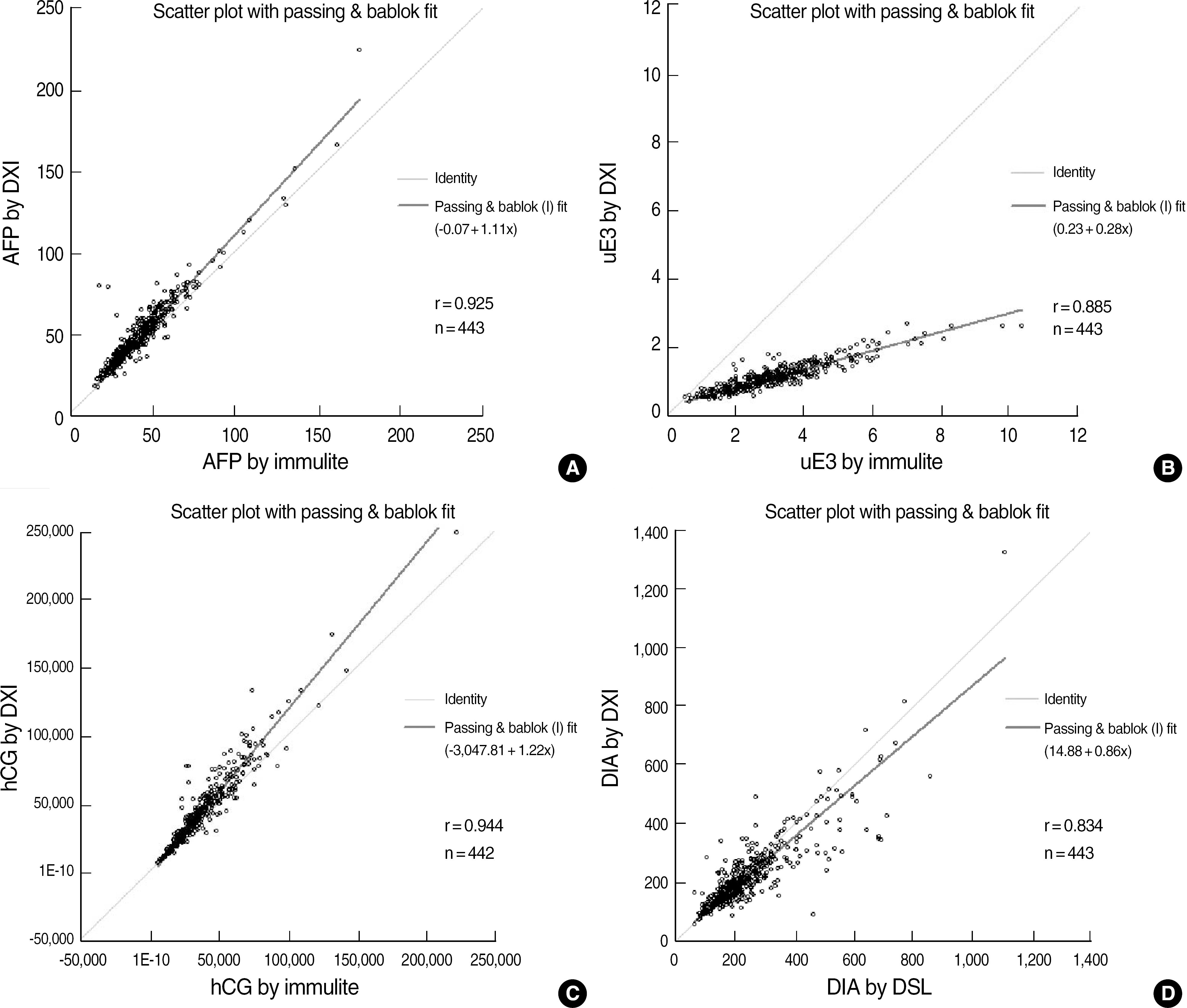 | Fig. 1.Comparison study of each of the four tests of the quadruple marker by DxI vs Immulite or DSL ELISA. (A) AFP, (B) uE3, (C) hCG, (D) DIA.
Abbreviations: DXI, UniCel DxI 800 (Beckman Coulter, Fullerton, CA, USA); Immulite, Immulite 2000 (Siemens Healthcare Diagnostics, Deerfield, IL, USA); DSL ELISA (DSL, Webster, TX, USA); AFP, alpha-fetoprotein; uE3, unconjugated estriol; hCG, human chorionic gonadotrophin; DIA, dimeric inhibin A (DIA).
|
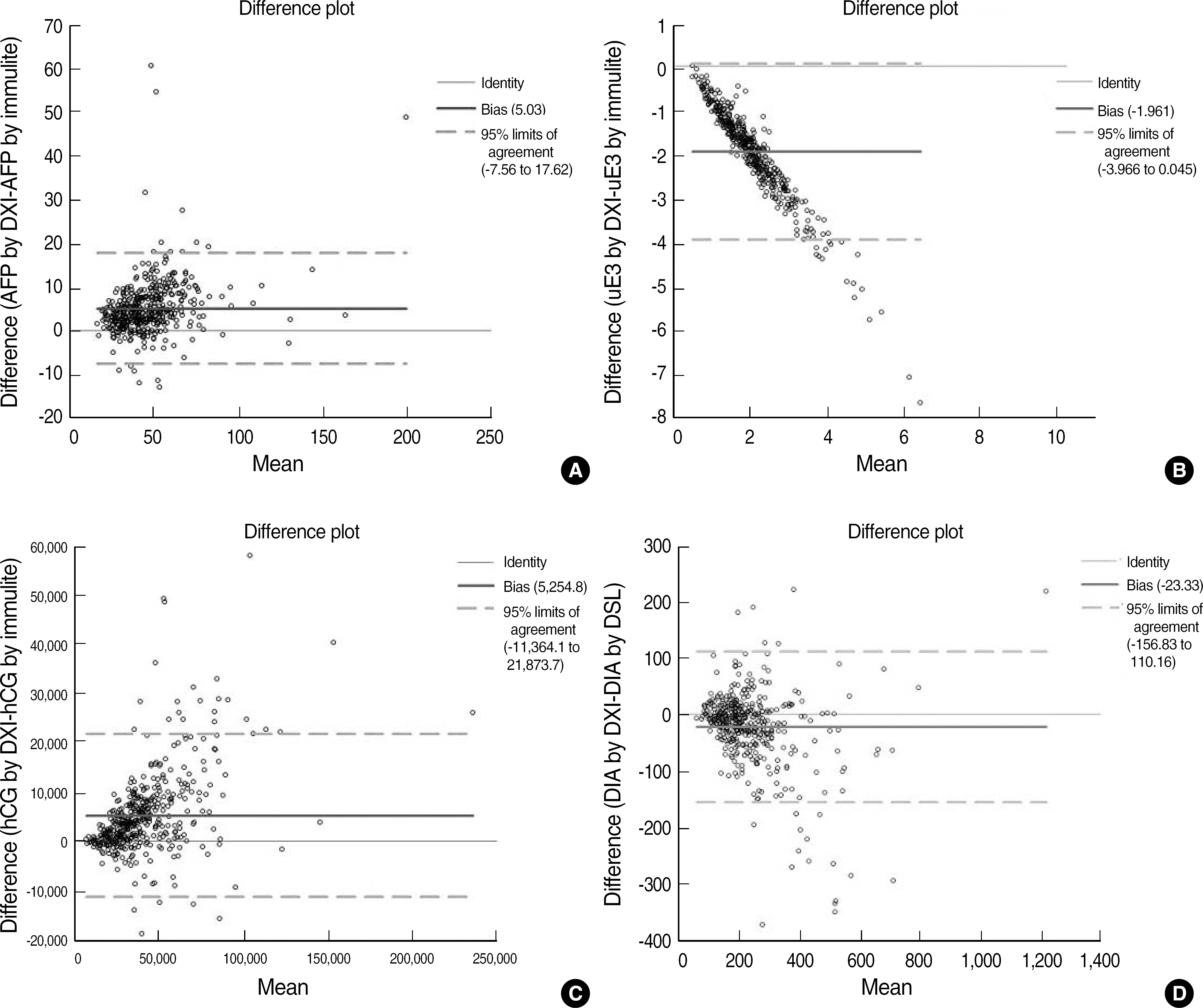 | Fig. 2.Bland-Altman plots for each of the four markers in the quadruple test using Unicel DxI 800 vs Immulite 2000 or DSL ELISA. (A) AFP, (B) uE3, (C) hCG, (D) DIA.
Abbreviations: See Fig. 1.
|
Table 1.
Precision of the Beckman Coulter DxI for the quadruple assay
Table 2.
Median values of each of the four quadruple markers determined using the Beckman Coulter DxI
| Gestational week∗ | N. of samples | AFP (ng/mL) | uE3 (ng/mL) | hCG (IU/mL) | DIA (pg/mL) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Obs | Reg† | Obs | Reg | Obs | Reg | Obs | Reg | ||
| 15th | 130 | 40.9 | 39.7 | 0.8 | 0.9 | 46.2 | 46.7 | 222.4 | 222.3 |
| 16th | 219 | 44.1 | 45.4 | 1.1 | 1.1 | 37.4 | 36.3 | 188.0 | 190.0 |
| 17th | 160 | 53.4 | 52.0 | 1.4 | 1.4 | 28.7 | 30.2 | 185.3 | 178.1 |
| 18th | 165 | 58.8 | 59.6 | 1.7 | 1.7 | 25.9 | 26.6 | 176.8 | 183.0 |
| 19th | 97 | 67.0 | 68.2 | 2.0 | 2.1 | 26.6 | 24.6 | 209.2 | 206.3 |
| 20th | 17 | 89.1 | 78.1 | 2.8 | 2.7 | 21.1 | 23.4 | 258.9 | 254.9 |
| Total | 788 | ||||||||
Table 3.
Comparison of the median values (pg/mL) of dimeric inhibin A
| Gestational week∗ | Lambert-Messerlian et al.† [9] | Rawlins et al.† [11] | Present study | ||||||
|---|---|---|---|---|---|---|---|---|---|
| N | Observed | Regressed | N | Observed | N | Observed | Regressed‡ | Maternal weight-corrected values§ | |
| 15th | 82 | 157.3 | 147.0 | 2,422 | 166 | 130 | 222.4 | 222.3 | 225.9 |
| 16th | 126 | 126.0 | 138.6 | 3,897 | 149 | 219 | 188.0 | 190.0 | 195.4 |
| 17th | 106 | 134.3 | 135.0 | 2,851 | 150 | 160 | 185.3 | 178.1 | 187.2 |
| 18th | 103 | 142.1 | 136.5 | 1,079 | 151 | 165 | 176.8 | 183.0 | 178.8 |
| 19th | 90 | 147.4 | 143.0 | 526 | 161 | 97 | 209.2 | 206.3 | 206.2 |
| 20th | 58 | 144.4 | 155.4 | 279 | 174 | 17 | 258.9 | 254.9 | 277.7 |
| Total | 565 | 11,054 | 788 | ||||||
Table 4.
Comparison of dimeric inhibin A (DIA) between automated assay and ELISA
| Study | N | Slope | Intercept | r | Mean % difference | Mean ratio of the two assays |
|---|---|---|---|---|---|---|
| Lambert-Messerlian et al.∗ [9] | 565 | 1.00 | -0.776 | 0.960 | NE | 0.768 |
| Rawlins et al.∗ [11] | 119 | 0.88 | -3.67 | 0.980 | -14.6% | NE |
| Present study | 447 | 0.86 | 14.88 | 0.834 | -8.0% | 0.949 |
Table 5.
Regressed median values of alpha-fetoprotein (AFP) compared with those of samples from other countries
| Gestational week∗ | AFP (IU/mL) | ||||||
|---|---|---|---|---|---|---|---|
| Vranken et al. [13] | Present study | ||||||
| Belgium | Canada | Germany | UK | USA | Korea† | Weight-corrected‡ | |
| 14th | 22.1 | - | 21.8 | 23.6 | - | - | - |
| 15th | 25.8 | 27.5 | 25.3 | 27.0 | 25.7 | 32.8 | 31.9 |
| 16th | 30.2 | 31.1 | 29.2 | 30.8 | 29.7 | 37.5 | 38.2 |
| 17th | 35.3 | 35.1 | 32.5 | 35.2 | 34.4 | 43.0 | 43.8 |
| 18th | 41.2 | 39.7 | 37.5 | 40.2 | 39.7 | 49.2 | 49.8 |
| 19th | 48.2 | 44.9 | 46.3 | 46.0 | 46.0 | 56.3 | 55.8 |
| 20th | - | 50.6 | - | 52.5 | 53.2 | 64.5 | 78.0 |
Table 6.
Regressed median values of uE3 compared with those of samples from other countries
| Gestational week∗ | uE3 (nmol/L) | |||||
|---|---|---|---|---|---|---|
| Vranken et al. [13] | Present study† | |||||
| Belgium | Canada | Germany | UK | Korea | Weight-corrected‡ | |
| 14th | 2.15 | - | 2.22 | 2.26 | - | - |
| 15th | 2.80 | 2.53 | 3.02 | 2.84 | 3.12 | 2.93 |
| 16th | 3.66 | 3.19 | 3.92 | 3.58 | 3.82 | 3.81 |
| 17th | 4.77 | 3.99 | 5.00 | 4.51 | 4.86 | 4.87 |
| 18th | 6.22 | 4.96 | 6.21 | 5.67 | 5.90 | 5.92 |
| 19th | 8.11 | 6.21 | 7.95 | 7.14 | 7.29 | 6.99 |
| 20th | - | 7.77 | - | 8.99 | 9.37 | 10.00 |
Table 7.
Regressed median values of human chorionic gonadotrophin (hCG) compared with those of samples from other countries
| Gestational week∗ | hCG (IU/mL) | |||||
|---|---|---|---|---|---|---|
| Vranken et al. [13] | Present study | |||||
| Belgium | Canada | Germany | UK | Korea | Weight-corrected† | |
| 14th | 37.3 | - | 34.9 | 40.0 | - | - |
| 15th | 30.2 | 42.4 | 26.5 | 33.2 | 46.7 | 43.9 |
| 16th | 24.5 | 34.1 | 21.4 | 27.6 | 36.3 | 35.6 |
| 17th | 19.9 | 28.3 | 18.9 | 22.9 | 30.2 | 26.6 |
| 18th | 16.1 | 24.1 | 17.3 | 19.1 | 26.6 | 23.4 |
| 19th | 13.0 | 21.2 | 14.8 | 15.9 | 24.6 | 23.2 |
| 20th | - | 19.1 | - | 13.2 | 23.4 | 19.6 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download