Abstract
Purpose
Breast augmentation with cohesive silicone gel implant has been popular but there remains the risk of implant rupture. We investigated the diagnosis and treatment of cohesive gel implant rupture.
Methods
Ten cases of cohesive gel implant rupture between August 2006 and August 2010 in ooo were reviewed in this study. The diagnostic role of Magnetic resonance imaging (MRI) and ultrasonography (US), and operative findings of cases were studied retrospectively.
Results
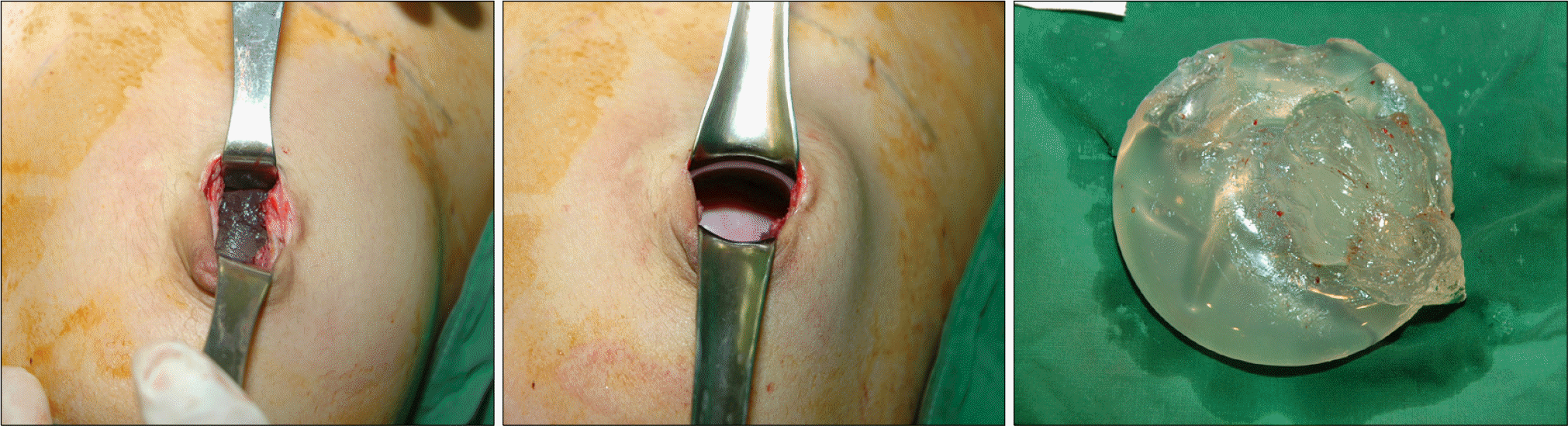
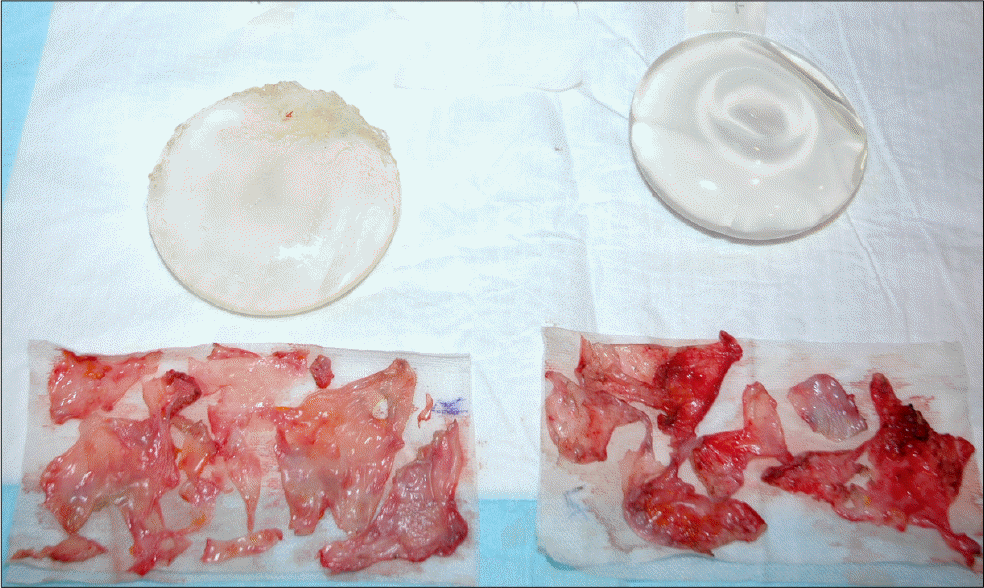
The mean interval from previous surgery was 14.7 months ranging from 3 to 44 months. Nine cases were visited due to abrupt changes in texture of implants and 1 case for revision of capsular contracture. Seven of 10 cases had capsular contracture, simultaneously. We diagnosed the first case by US and MRI but only US was used in the other 9 cases. US showed discontinuity of the implant membrane and multiple parallel echogenic lines within the implant interior (stepladder sign), and MRI showed the presence of multiple curvilinear low-signal-intensity lines seen within the high-signal-intensity silicone gel (linguine sign). All the ruptured gel remained in place within the capsule and did not migrate into the surrounding area. Surgeries were implant replacement in 3, replacement with capsulectomy in 6 with capsular contracture, and subpectoral conversion with capsulectomy and mastopexy in 1 case.
Go to : 
References
1. Cronin TD. Augmentation mammaplasty: a new "natural feel" prosthesis. Transactions of the Third international Congress of Plastic Surgery. 1963. 41–9.
2. Miyoshi K, Kobayashi Y. Hypergammaglobulinemia by prolonged adjuvanticity in men: disorders developed after augmentation mammoplasty. Jpn Med J. 1964; 2122:9–14.
3. National Toxicology Program. Bioassay of 2,4-diaminotoluene for possible carcinogenicity. Natl Cancer Inst Carcinog Tech Rep Ser. 1979; 162:1–139.
4. Endo LP, Edwards NL, Longley S, Corman LC, Panush RS. Silicone and rheumatic diseases. Semin Arthritis Rheum. 1987; 17:112–8.

5. Uchida J. Clinical application of crosslinked dimethylpolysi-loxane, restoration of breast, cheeks, atrophy of infantile paralysis, funnel-shape chest, etc. Jpn J Plast Reconstr Surg. 1961; 4:303.
6. van Nunen SA, Gatenby PA, Basten A. Post-mammoplasty connective tissue disease. Arthritis Rheum. 1982; 25:694–7.

7. Berkel H, Birdsell DC, Jenkins H. Breast augmentation: a risk factor for breast cancer? N Engl J Med. 1992; 326:1649–53.
8. Duffy MJ, Woods JE. Health risks of failed silicone gel breast implants: a 30-year clinical experience. Plast Reconstr Surg. 1994; 94:295–9.
9. Sanchez-Guerrero J, Schur PH, Sergent JS, Liang MH. Silicone breast implants and rheumatic disease. Clinical, immunologic, and epidemiologic studies. Arthritis Rheum. 1994; 37:158–68.
10. Tebbetts J. McGhan's biodimensional augmentation system cohesive gel mammary implants. Instructional video 55'. McGhan Medical Corporation. 1993.
11. Heden P, Bone B, Murphy DK, Slicton A, Walker PS. Style 410 cohesive silicone breast implants: safety and effectiveness at 5 to 9 years after implantation. Plast Reconstr Surg. 2006; 118:1281–7.
12. Janowsky EC, Kupper LL, Hulka BS. Meta-analyses of the relation between silicone breast implants and the risk of con-nective-tissue diseases. N Engl J Med. 2000; 342:781–90.

13. Lipworth L, Tarone RE, McLaughlin JK. Silicone breast implants and connective tissue disease: an updated review of the epidemiologic evidence. Ann Plast Surg. 2004; 52:598–601.
14. Bryant H, Brasher P. Breast implants and breast cancer–rea-nalysis of a linkage study. N Engl J Med. 1995; 332:1535–9.
15. Cher DJ, Conwell JA, Mandel JS. MRI for detecting silicone breast implant rupture: meta-analysis and implications. Ann Plast Surg. 2001; 47:367–80.

16. Ikeda DM, Borofsky HB, Herfkens RJ, Sawyer-Glover AM, Birdwell RL, Glover GH. Silicone breast implant rupture: pit-falls of magnetic resonance imaging and relative efficacies of magnetic resonance, mammography, and ultrasound. Plast Reconstr Surg. 1999; 104:2054–62.

17. Lalonde L, David J, Trop I. Magnetic resonance imaging of the breast: current indications. Can Assoc Radiol J. 2005; 56:301–8.
18. Gorczyca DP, Gorczyca SM, Gorczyca KL. The diagnosis of silicone breast implant rupture. Plast Reconstr Surg. 2007; 120:49S–61S.

19. Lee S. Complications and reasons for dissatisfaction in augmentation mammoplasty- analysis of 42 cases of re-operation. J Korean Breast Cancer Soc. 2004; 7:121–5.
20. Schnur PL, Weinzweig J, Harris JB, Moyer TP, Petty PM, Nixon D, et al. Silicon analysis of breast and periprosthetic capsular tissue from patients with saline or silicone gel breast implants. Plast Reconstr Surg. 1996; 98:798–803.

21. Holmich LR, Vejborg IM, Conrad C, Sletting S, Hoier- Madsen M, Fryzek JP, et al. Untreated silicone breast implant rupture. Plast Reconstr Surg. 2004; 114:204–14. discussion 15–6.
22. Deapen DM, Brody GS. Breast augmentation and the risk of subsequent breast cancer. N Engl J Med. 1993; 328:662–3.

23. Cunningham B, McCue J. Safety and effectiveness of Mentor's MemoryGel implants at 6 years. Aesthetic Plast Surg. 2009; 33:440–4.

24. Heden P, Jernbeck J, Hober M. Breast augmentation with anatomical cohesive gel implants: the world's largest current experience. Clin Plast Surg. 2001; 28:531–52.
25. Brown MH, Shenker R, Silver SA. Cohesive silicone gel breast implants in aesthetic and reconstructive breast surgery. Plast Reconstr Surg. 2005; 116:768–79. discussion 80–1.

26. O'Toole M, Caskey CI. Imaging spectrum of breast implant complications: mammography, ultrasound, and magnetic resonance imaging. Semin Ultrasound CT MR. 2000; 21:351–61.
27. DeBruhl ND, Gorczyca DP, Ahn CY, Shaw WW, Bassett LW. Silicone breast implants: US evaluation. Radiology. 1993; 189:95–8.

28. McCarthy CM, Pusic AL, Kerrigan CL. Silicone breast implants and magnetic resonance imaging screening for rupture: do U.S. Food and Drug Administration recommendations reflect an evidence-based practice approach to patient care? Plast Reconstr Surg. 2008; 121:1127–34.
Go to : 
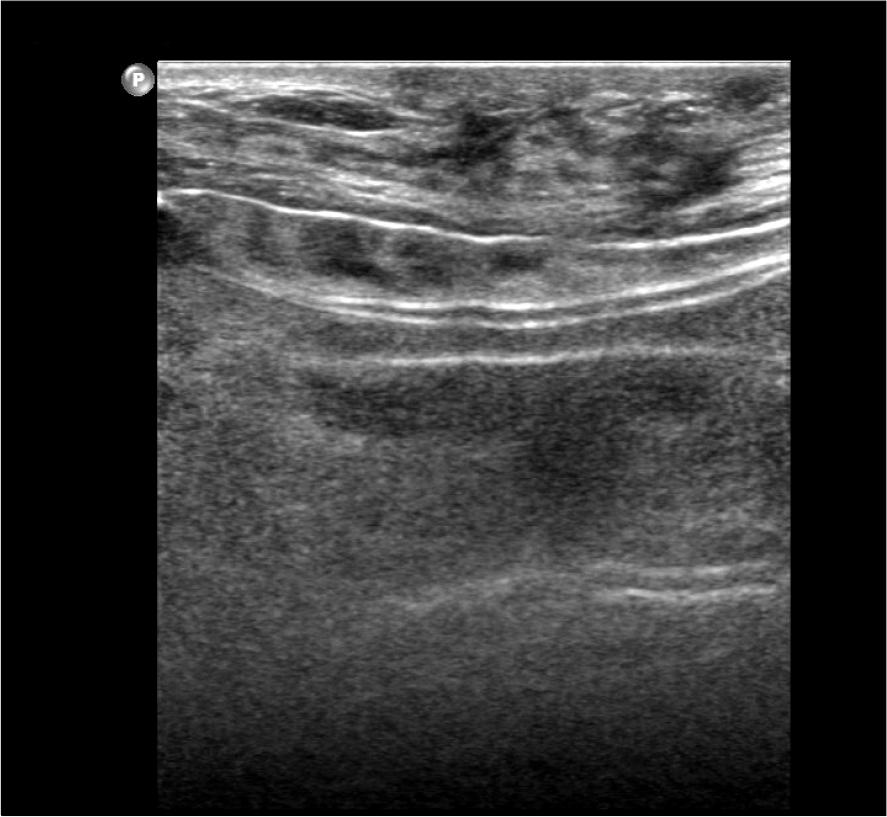 | Fig. 1.Ultrasonography showed discontinuity of the implant membrane and multiple parallel echogenic lines within the implant interior (stepladder sign). |
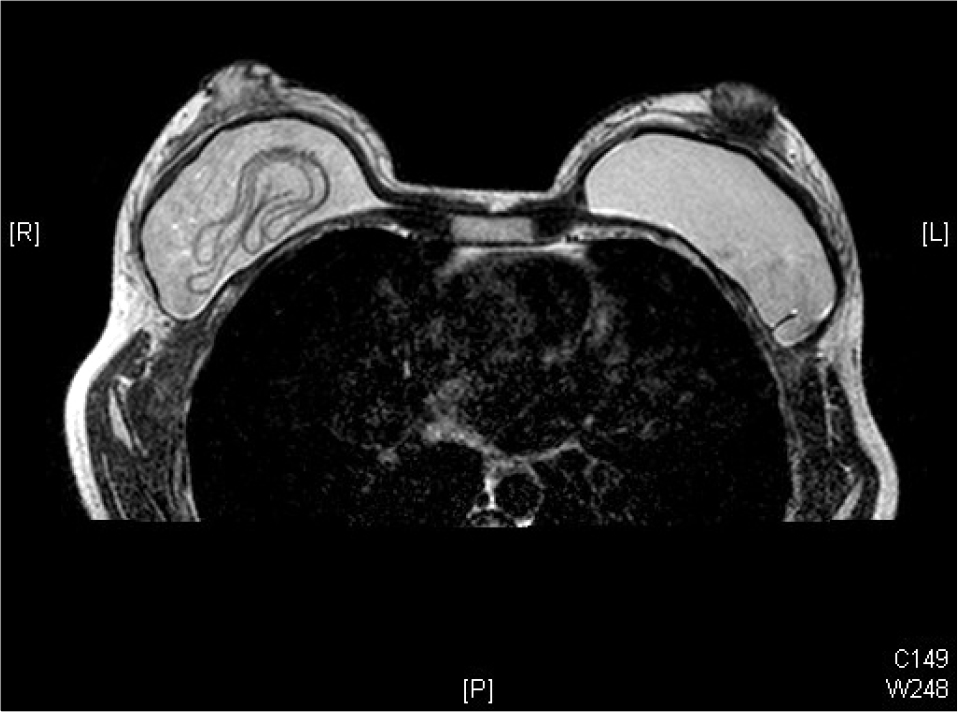 | Fig. 2.MRI showed the presence of multiple curvilinear low-sig-nal-intensity lines seen within the high-signal-intensity silicone gel (linguine sign). |
Table 1.
Demography of patients
| Case No. | Age (year) | Time to rupture (month) | US* | MRI† | Contracture | Implant volume (cc) | Simultaneous capsulectomy | Rupture type |
|---|---|---|---|---|---|---|---|---|
| 1 | 28 | 12 | Yes | Yes | Yes | 225 | Yes | Intracapsular |
| 2 | 26 | 10 | Yes | No | No | 225 | No | Intracapsular |
| 3 | 25 | 14 | Yes | No | No | 250 | No | Intracapsular |
| 4 | 41 | 12 | Yes | No | Yes | 250 | Yes | Intracapsular |
| 5 | 29 | 12 | Yes | No | No | 275 | No | Intracapsular |
| 6 | 34 | 5 | Yes | No | Yes | 250 | Yes | Intracapsular |
| 7 | 32 | 24 | Yes | No | Yes | 300 | Yes | Intracapsular |
| 8 | 37 | 3 | Yes | No | Yes | 225 | Yes | Intracapsular |
| 9 | 30 | 44 | Yes | No | Yes | 250 | Yes | Intracapsular |
| 10 | 24 | 11 | Yes | No | Yes | 190 | Yes | Intracapsular |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download