Abstract
Case summary
A 66-year-old male presented with a foreign body sensation in his right eye. He had no systemic or ocular inflammatory diseases. He had undergone penetrating keratoplasty following amniotic membrane transplantation for corneal perforation at 1 day after pterygium excision in his left eye. The uncorrected visual acuity (UCVA) and intraocular pressure (IOP) in his right eye were 0.8 and 2 mmHg, respectively. Active leakage of an aqueous humor on the head of the pterygium was found on slit lamp examination. With the impression of spontaneous corneal perforation combined with primary pterygium, emergent amniotic membrane transplantation was performed. Diffuse corneal thinning around the 2 mm-sized perforation site was found 3mm away from the limbus in the 5 o'clock after removing the pterygium. The perforation site was covered with amniotic membrane after applying fibrin glue. UCVA and IOP in his right eye were 1.0 and 9 mmHg, respectively, at postoperative 6 months. No definite recurrence of pterygium or additional corneal perforation was found.
Figures and Tables
Figure 1
Anterior segment photographs and schematic figure. (A, B) Nasal pterygium and active leakage of aqueous humor on the head of pterygium were found in the right eye (arrows). (C) Bullous keratopathy after penetrating keratoplasty for instant corneal perforation following pterygium excision on his left eye. (D) Schematic figure of corneal perforation and pterygium on the right eye. (E) The perforation site was covered with amniotic membrane in postoperative 3 weeks. (F) Corneal surface was smooth and mild stromal opacity was found at the previous amniotic membrane transplantation site in postoperative 9 months.
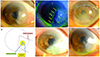
Figure 2
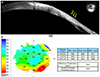
Anterior segment optical coherence tomography and Pachymetry map finding in postoperative 9 months. (A) Previous corneal perforation site was well repaired after amniotic membrane transplantation (yellow arrows: previous corneal perforation site). (B) Mild thinning around previous perforation site was found and other peripheral cornea revealed no definite thinning. OD = oculus dexter; Min. = minimum; Avg. = average; Max. = maximum.
References
1. Lim CY, Kim SH, Chuck RS, et al. Risk factors for pterygium in Korea: The Korean National Health and Nutrition Examination Survey V, 2010-2012. Medicine (Baltimore). 2015; 94:e1258.
2. Liu T, Liu Y, Xie L, et al. Progress in the pathogenesis of pterygium. Curr Eye Res. 2013; 38:1191–1197.
3. Dougherty PJ, Hardten DR, Lindstrom RL. Corneoscleral melt after pterygium surgery using a single intraoperative application of mitomycin-C. Cornea. 1996; 15:537–540.
4. Fujitani A, Hayasaka S, Shibuya Y, Noda S. Corneoscleral ulceration and corneal perforation after pterygium excision and topical mitomycin C therapy. Ophthalmologica. 1993; 207:162–164.
5. Kassir MS. Corneal perforation after excision of pterygium and use of 002% mitomycin eyedrops. J Fr Ophtalmol. 1999; 22:776–779.
6. Hagen KB, Waring GO 3rd, Johnson-Wint B. Progressive non-ulcerative paracentral keratolysis associated with elevated corneal metalloproteinases. Cornea. 1997; 16:486–492.
7. Lekhanont K, Patarakittam T, Mantachote K, et al. Progressive keratolysis with pseudopterygium associated with erythema elevatum diutinum. Ophthalmology. 2011; 118:927–933.
8. Tsai YY, Chiang CC, Yeh KT, et al. Effect of TIMP-1 and MMP in pterygium invasion. Invest Ophthalmol Vis Sci. 2010; 51:3462–3467.
9. Di Girolamo N, Chui J, Coroneo MT, Wakefield D. Pathogenesis of pterygia: role of cytokines, growth factors, and matrix metalloproteinases. Prog Retin Eye Res. 2004; 23:195–228.
10. Di Girolamo N, Wakefield D, Coroneo MT. Differential expression of matrix metalloproteinases and their tissue inhibitors at the advancing pterygium head. Invest Ophthalmol Vis Sci. 2000; 41:4142–4149.
11. Gomez DE, Alonso DF, Yoshiji H, Thorgeirsson UP. Tissue inhibitors of metalloproteinases: structure, regulation and biological functions. Eur J Cell Biol. 1997; 74:111–122.
12. Fukuda K, Chikama T, Nakamura M, Nishida T. Differential distribution of subchains of the basement membrane components type IV collagen and laminin among the amniotic membrane, cornea, and conjunctiva. Cornea. 1999; 18:73–79.
13. Tseng SC, Li DQ, Ma X. Suppression of transforming growth factor-beta isoforms, TGF-beta receptor type II, and myofibroblast differentiation in cultured human corneal and limbal fibroblasts by amniotic membrane matrix. J Cell Physiol. 1999; 179:325–335.
14. Kim HK, Park HS. Fibrin glue-assisted augmented amniotic membrane transplantation for the treatment of large noninfectious corneal perforations. Cornea. 2009; 28:170–176.
15. Solomon A, Meller D, Prabhasawat P, et al. Amniotic membrane grafts for nontraumatic corneal perforations, descemetoceles, and deep ulcers. Ophthalmology. 2002; 109:694–703.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download