Abstract
Purpose
To report a case of recovery of bilateral cortical blindness in a patient with posterior reversible encephalopathy syndrome.
Case summary
A 46-year-old female visited the ophthalmology department due to abrupt visual acuity decrease. Ten days earlier, she had received conservative management due to anemia caused by menorrhagia and uterine prolapse. She underwent a gynecological operation to remove a uterine myoma two days previously, and was given a blood transfusion postoperatively because of excessive bleeding. After the transfusion, she complained of acute blurred vision. Her best corrected visual acuity (BCVA) was hand motion 10 cm in both eyes. There were no abnormal specific findings except retinal dot hemorrhage at the temporal side in the left eye on fundus examination. Her pupillary light reflex was normal and optical coherence tomography examination was unremarkable in both eyes. However, flash visual evoked potential findings showed reduced P100 amplitude in both eyes and she was diagnosed with posterior reversible encephalopathy syndrome based on brain magnetic resonance imaging. After close observation without any treatment, the symptoms gradually improved. Finally, her BCVA recovered to 1.0 and P1 and P100 amplitudes were restored to normal range in both eyes at 16 weeks from the first diagnosis.
Go to : 
References
1. Hinchey J, Chaves C, Appignani B, et al. A reversible posterior leukoencephalopathy syndrome. N Engl J Med. 1996; 334:494–500.

2. Pavlakis SG, Frank Y, Kalina P, et al. Occipital-parietal encephalopathy: a new name for an old syndrome. Pediatr Neurol. 1997; 16:145–8.

3. Kahana A, Rowley HA, Weinstein JM. Cortical blindess: abdominal and radiologic findings in reversible posterior leukoencephalopathy syndorme: case report and review of the literature. Ophthalmology. 2005; 112:e7–e11.
4. Honkaniemi J, Kähärä V, Dastidar P, et al. Reversible posterior leukoencephalopathy after combination chemotherapy. Neuroradiology. 2000; 42:895–9.

5. Rubin AM, Kang H. Cerebral blindness and encephalopathy with cyclosporin A toxicity. Neurology. 1987; 37:1072–6.

6. Shin RK, Stern JW, Janss AJ, et al. Reversible posterior leukoencephalopathy during the treatment of acute lymphoblastic leukemia. Neurology. 2001; 56:388–91.

7. Do DV, Rismondo V, Nguyen QD. Reversible cortical blindness in preeclampsia. Am J Ophthalmol. 2002; 134:916–8.

8. Schwartz RB, Feske SK, Polak JF, et al. Preeclampsia-eclampsia: clinical and neuroradiographic correlates and insights into the pathogenesis of hypertensive encephalopathy. Radiology. 2000; 217:371–6.

9. Ito Y, Niwa H, Iida T, et al. Post-transfusion reversible posterior leukoencephalopathy syndrome with cerebral vasoconstriction. Neurology. 1997; 49:1174–5.

10. Ikeda M, Ito S, Hataya H, et al. Reversible posterior leukoencephalopathy in a patient with minimal-change nephrotic syndrome. Am J Kidney Dis. 2001; 37:E30.

11. Fugate JE, Rabinstein AA. Posterior reversible encephalopathy syndrome: clinical and radiological manifestations, pathophysiology, and outstanding questions. Lancet Neurol. 2015; 14:914–25.

12. Antunes NL, Small TN, George D, et al. Posterior leukoencephalopathy syndrome may not be reversible. Pediatr Neurol. 1999; 20:241–3.

13. Lindsberg PJ, Sirén AL, Hallenbeck JM. Microvascular perfusion during focal vasogenic brain edema: a scanning laser fluorescence microscopy study. Microvasc Res. 1997; 53:92–103.

14. Byrom FB. The pathogenesis of hypertensive encephalopathy and its relation to the malignant phase of hypertension; experimental evidence from the hypertensive rat. Lancet. 1954; 267:201–11.

15. Park SK, Rha JH, Han SR, et al. Immunosuppressants induced reversible posterior dominant encephalopathy-a capillary leak syndrome. J Korean Neurol Assoc. 1997; 15:1125–35.
16. Karuppannasamy D, Vikrant K, Raghuram A, Kumaar TM. Cortical visual loss in posterior reversible encephalopathy abdominal in late postpartum eclampsia: case series. Indian J Ophthalmol. 2014; 62:635–8.
17. Niyadurupola N, Burnett CA, Allen LE. Reversible posterior leu-coencephalopathy syndrome: a cause of temporary cortical blindness. Br J Ophthalmol. 2005; 89:924–5.

18. Garg RK. Postpartum posterior leukoencephalopathy syndrome. J Assoc Physicians India. 2003; 51:211–3.
19. Kauntia R, Valsalan R, Seshadri S, et al. Late postpartum preeclampsia with posterior reversible encephalopathy syndrome. Indian J Med Sci. 2009; 63:508–11.

20. Boughammoura A, Touzé E, Oppenheim C, et al. Reversible angiopathy and encephalopathy after blood transfusion. J Neurol. 2003; 250:116–8.

21. Huang YC, Tsai PL, Yeh JH, Chen WH. Reversible posterior leukoencephalopathy syndrome caused by blood transfusion: a case report. Acta Neurol Taiwan. 2008; 17:258–62.
22. Singh K, Gupta R, Kamal H, et al. Posterior reversible encephalopathy syndrome secondary to blood transfusion. J Clin Neurosci. 2015; 22:592–4.

23. Ay H, Buonanno FS, Schaefer PW, et al. Posterior leukoencephalopathy without severe hypertension: utility of diffusion-weighted MRI. Neurology. 1998; 51:1369–76.

24. Norman JK, Parke JT, Wilson DA, McNall-Knapp RY. Reversible posterior leukoencephalopathy syndrome in children undergoing induction therapy for acute lymphoblastic leukemia. Pediatr Blood Cancer. 2007; 49:198–203.

25. Kim KI, Choe BK, Kim HS, et al. A case of reversible posterior leukoencephalopathy in a child with acute lymphoblastic leukemia. Clin Pediatr Hematol Oncol. 2007; 14:78–82.
Go to : 
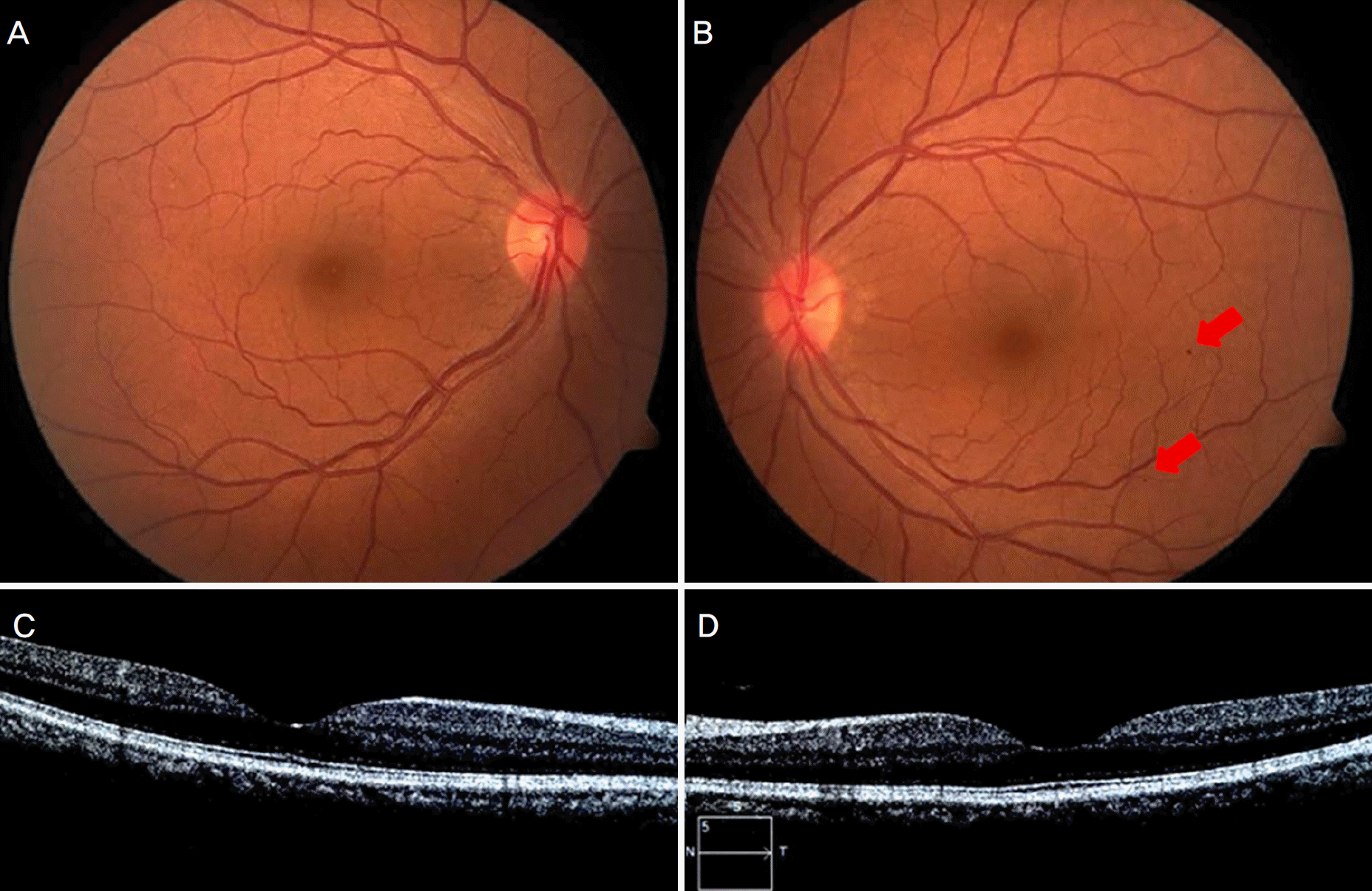 | Figure 1.At the first visit, fundus color photographs and optical coherence tomography images in both eyes. Fundus color photographs showed normal fundus of right eye (A) and dot retinal hemorrhage (red arrows) on left eye (B). Optical coherence tomography image showed no macular abnormalities on right eye (C) and left eye (D). |
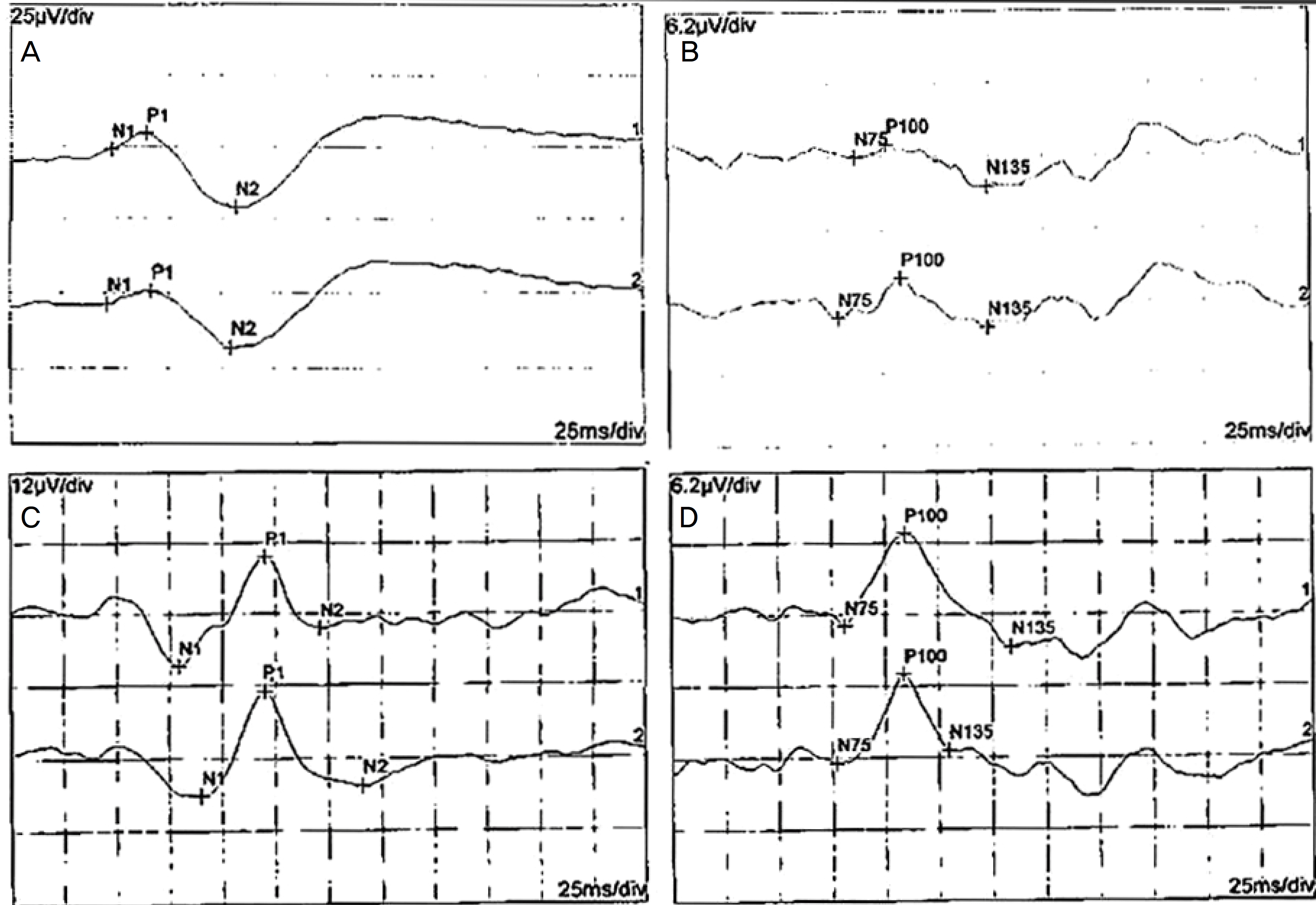 | Figure 2.Serial changes of flash visual evoked potential (VEP) and pattern VEP in both eyes. Flash VEP findings showed reduced P1 amplitude in both eyes at the first visit (A) and pattern VEP findings showed reduced P100 amplitude in both eyes at 5 weeks later (B). Flash VEP recovered normal P100 amplitude at 16 weeks later (C) and pattern VEP recovered normal P100 amplitude at 16 weeks later, too (D). |
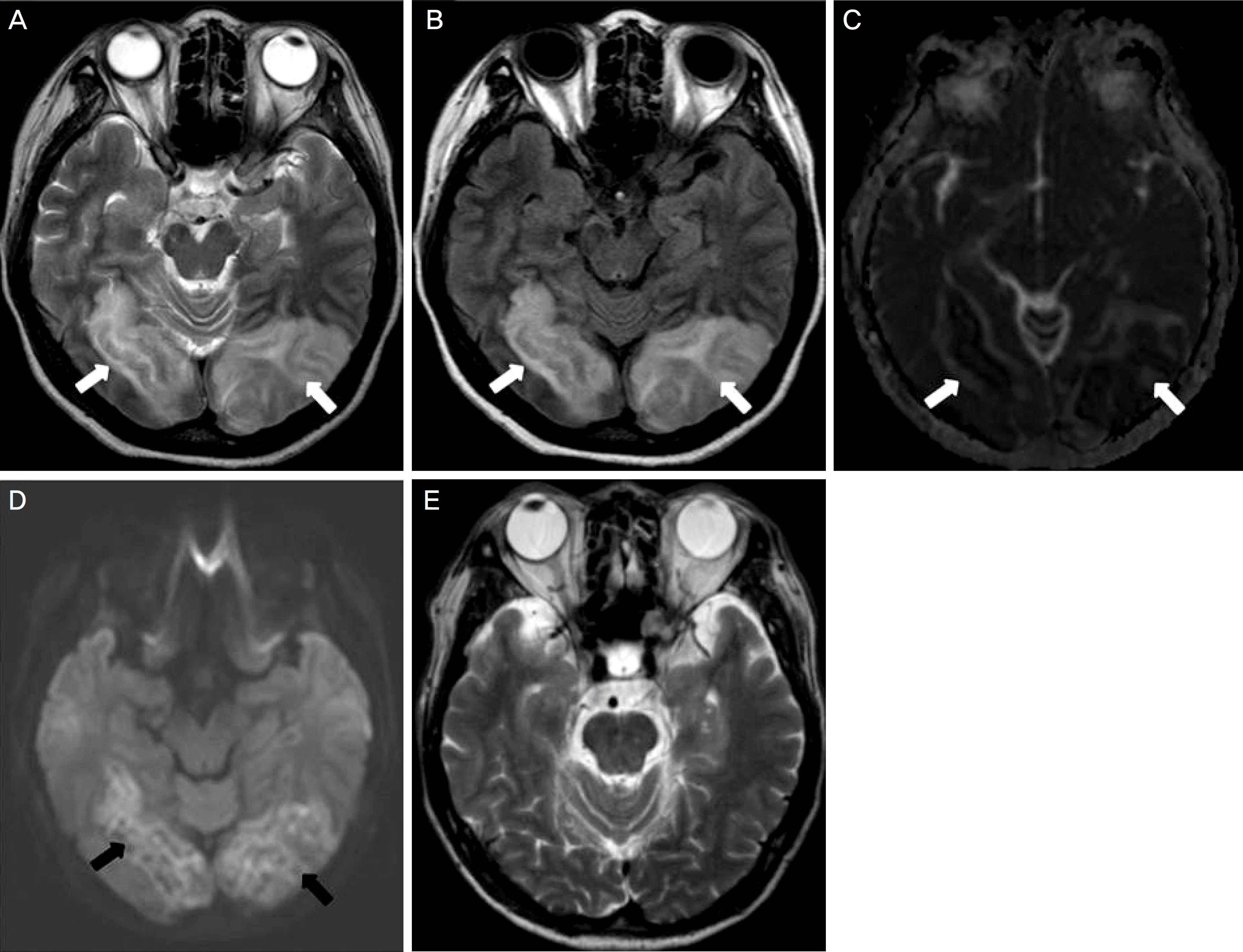 | Figure 3.Brain magnetic resonance imaging (MRI) at the 4th day after first symptom showed features of posterior reversible encephalopathy syndrome. Bilateral parieto-occipital lesions with a white matter predominance seen on T2-weighted MRI (A, white arrows) and fluid attenuated inversion recovery image (B, white arrows). Apparent diffusion coefficient maps (C, white arrows), which show a vasogenic pattern of edema in white matter. The diffusion-weighted image sequence shows slight heterogenous hyperintensity (D, black arrows). T2-weighted MRI taken 18 weeks later reveals resolution of previous lesion (E). |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download