초록
Purpose:
We report three cases of esotropia after botulinum toxin A injection in the eyelid for esthetic purposes.
Case summary:
Esotropia was observed in three patients presented with diplopia after botulinum toxin A application in the eyelid for esthetic purposes. (Case 1) A 51-year-old woman developed diplopia, mild ptosis and hyperemia in both eyes after periocular cosmetic use of botulinum toxin A 6 weeks before. In the primary position, the angle of esotropia was 10 prism diopters (PD) at distance and 6 PD at near. She had slightly limited abduction and mild ptosis on both eyes. (Case 2) A 50-year-old woman developed diplopia after botulinum toxin A injection in the upper eyelid 15 days prior. In primary position, the angle of esotropia was 18 PD at distance and 12 PD at near. She had slightly limited abduction and mild ptosis in both eyes. (Case 3) A 40-year-old woman was examined with diplopia after botulinum toxin A injection in the crow’s feet and lower lid 6 days earlier. In primary position, the angle of esotropia was 30-35 PD at distance and 18 PD at near. She had slightly limited abduction. Prism glasses were prescribed for patients in case 1 and case 3. All three patients were resolved in symptoms of diplopia, gaze limitation and ptosis after 1 or 2 months.
Conclusions:
Botulinum toxin A could spread out from the lateral canthus to both lateral rectus muscles and cause a transient paresis of the muscles. This was presented as esotropia and the mild abduction deficits on both eyes. We suggest that patients presenting with diplopia should be asked if they have a history of injection of botulinum toxin A and patients should be fully informed about the complications of botulinum toxin A before injection.
Go to : 
References
1. Scott AB, Rosenbaum A, Collins CC. Pharmacologic weakening of extraocular muscles. Invest Ophthalmol. 1973; 12:924–7.
2. Carruthers A, Carruthers J. Botulinum toxin type A: history and current cosmetic use in the upper face. Semin Cutan Med Surg. 2001; 20:71–84.

3. Ahn KY, Park MY, Park DH, Han DG. Botulinum toxin A for the treatment of facial hyperkinetic wrinkle lines in Koreans. Plast Reconstr Surg. 2000; 105:778–84.

4. Cot TR, Mohan AK, Polder JA. . Botulinum toxin type A injections: adverse events reported to the US Food and Drug Administration in therapeutic and cosmetic cases. J Am Acad Dermatol. 2005; 53:407–15.
6. Lachkar Y, Bouassida W. Drug-induced acute angle closure glaucoma. Curr Opin Ophthalmol. 2007; 18:129–33.

7. Wutthiphan S, Kowal L, O'Day J. . Diplopia following subcutaneous injections of botulinum A toxin for facial spasms. J Pediatr Ophthalmol Strabismus. 1997; 34:229–34.

8. Aristodemou P, Watt L, Baldwin C, Hugkulstone C. Diplopia associated with the cosmetic use of botulinum toxin a for facial rejuvenation. Ophthal Plast Reconstr Surg. 2006; 22:134–6.

9. Lipham WJ. Cosmetic and clinical applications of botox and dermal fillers. 2nd ed.Thorofare NJ: SLACK Incorporated;2008. p. 5–9.
10. Chen CS, Miller NR. Botulinum toxin injection causing lateral rectus palsy. Br J Ophthalmol. 2007; 91:843.
11. Isaac CR, Chalita MR, Pinto LD. Botox (R) after Botox (R) - a new approach to treat diplopia secondary to cosmetic botulinic toxin use: case reports. Arq Bras Oftalmol. 2012; 75:213–4.
12. Lange DJ, Brin MF, Warner CL. . Distant effects of local injection of botulinum toxin. Muscle Nerve. 1987; 10:552–5.

14. Sohn MA, Lee TD. A case of cyclic esotropia cured by botulinum toxin injection. J Korean Ophthalmol Soc. 1999; 40:2645–50.
Go to : 
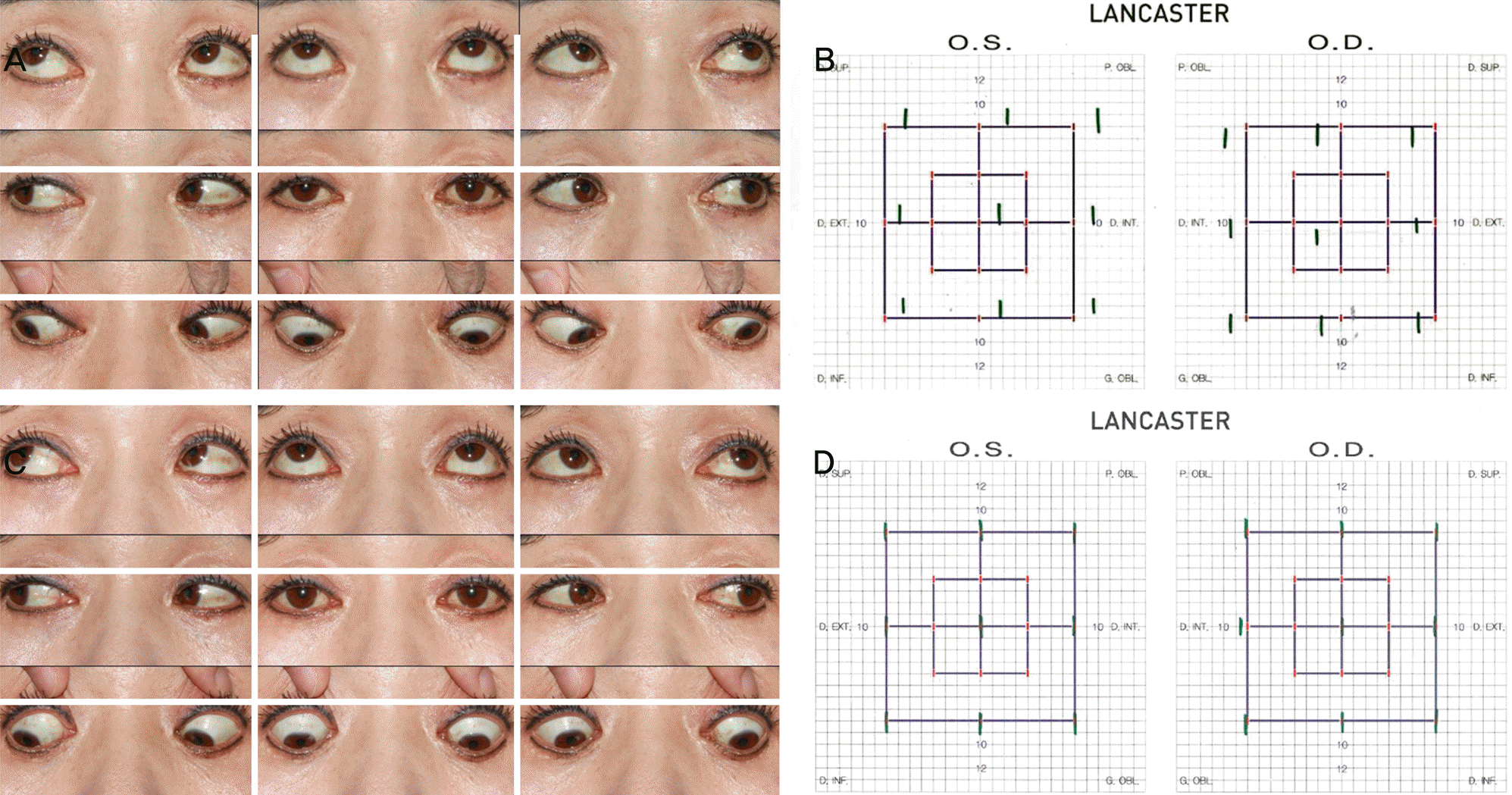 | Figure 1.The patient of case 1 showed esotropia after botulinum toxin A injection for crow’s feet treatment and resolved in symptoms after 2 months. (A) Nine cardinal gaze photographs taken 6 weeks after injection of botulinum toxin A showing approximately 10 prism diopters (PD) of esotropia. (B) Lancaster red-green test showed slightly limited abduction on both eyes. (C) Nine cardinal gaze photographs taken 2 months after using the 3 PD base out prism on her both spectacle lenses showing normal ocular motility. (D) There was no gaze limitation on both eyes after 2 months after injection of botulinum toxin A. O.S. = left eye; O.D. = right eye. |
Table 1.
Cases description, start of diplopia, performed treatment and recovery of symptoms
| Start of diplopia* | Injection site of botulinum toxin A | Eyelid ptosis | Treatment performed | Improvement of diplopia† | |
|---|---|---|---|---|---|
| Case 1 | 42 days | Crow’s feet, lower lid | Present | Base-out 3 PD glasses | 2 months |
| Case 2 | 15 days | Upper lid | Present | Observation | 1 month |
| Case 3 | 6 days | Crow’s feet, lower lid | Absent | Base-out 5 PD glasses | 2 months |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download