초록
Purpose:
To evaluate cross-sectional areas of conjunctiva and tear meniscus of conjunctivochalasis using Fourier-Domain RTVue-100 optical coherence tomography (OCT) before and after conjunctivochalasis surgery.
Methods:
Thirty-one patients (33 eyes) with symptomatic conjunctivochalasis were recruited for this study between June 2013 and April 2014. All patients underwent crescent-shaped conjunctiva resection and amniotic membrane transplantation. Anterior segment OCT (AS-OCT) imaging was performed and tear break-up time was evaluated prior to and 3 months after the conjunctivochalasis surgery. Cross-sectional areas of conjunctiva and tear meniscus of conjunctivochalasis at 7 locations (1 center, 3 nasal and 3 temporal areas) were measured in all patients.
Results:
The mean age of patients was 66.3 ± 10.8 years. Cross-sectional areas of conjunctivochalasis at all locations significantly decreased from 0.487 ± 0.42 mm2 to 0.007 ± 0.011 mm2 ( p < 0.001), whereas no significant changes in cross-sectional areas of tear meniscus at all 7 locations were observed after the surgery. Mean tear break-up time significantly increased from 2.26 ± 0.69 sec to 3.81 ± 1.22 sec following the surgery.
Go to : 
References
1. Meller D, Tseng SC. Conjunctivochalasis: literature review and possible pathophysiology. Surv Ophthalmol. 1998; 43:225–32.
2. Wang Y, Dogru M, Matsumoto Y, et al. The impact of nasal conjunctivochalasis on tear functions and ocular surface findings. Am J Ophthalmol. 2007; 144:930–7.

3. Watanabe A, Yokoi N, Kinoshita S, et al. Clinicopathologic study of conjunctivochalasis. Cornea. 2004; 23:294–8.

4. Jordan DR, Pelletier CR. Conjunctivochalasis. Can J Ophthalmol. 1996; 31:192–3.
5. Krachmer JH, Mannis MJ, Holland EJ. Cornea. 3rd ed.St. Louis: Mosby;2011. v. 1:p. 635–9.
7. Tseng SC, Prabhasawat P, Lee SH. Amniotic membrane transplantation for conjunctival surface reconstruction. Am J Ophthalmol. 1997; 124:765–74.

8. Meller D, Maskin SL, Pires RT, Tseng SC. Amniotic membrane transplantation for symptomatic conjunctivochalasis refractory to medical treatments. Cornea. 2000; 19:796–803.

9. Kheirkhah A, Casas V, Blanco G, et al. Amniotic membrane transplantation with fibrin glue for conjunctivochalasis. Am J Ophthalmol. 2007; 144:311–3.

10. Lim HJ, Lee JK, Park DJ. Conjunctivochalasis surgery: amniotic membrane transplantation with fibrin glue. J Korean Ophthalmol Soc. 2008; 49:195–204.

11. Nam K, Jo YJ, Lee SB. The efficacy of fibrin glue in surgical treatment of conjunctivochalasis with epiphora. J Korean Ophthalmol Soc. 2010; 51:498–503.

12. Oh SJ, Byon DS. Treatment of conjunctivochalasis using bipolar cautery. J Korean Ophthalmol Soc. 1999; 40:707–11.
13. Youm DJ, Kim JM, Choi CY. Simple surgical approach with high-frequency radio-wave electrosurgery for conjunctivochalasis. Ophthalmology. 2010; 117:2129–33.

14. Shin KH, Hwang JH, Kwon JW. New approach for conjunctivochalasis with argon laser photocoagulation. Can J Ophthalmol. 2012; 47:380–2.

16. Blumenthal EZ, Williams JM, Weinreb RN, et al. Reproducibility of nerve fiber layer thickness measurements by use of optical coherence tomography. Ophthalmology. 2000; 107:2278–82.
17. Chen TC, Cense B, Pierce MC, et al. Spectral domain optical coherence tomography: ultra-high speed, ultra-high resolution ophthalmic imaging. Arch Ophthalmol. 2005; 123:1715–20.
18. González-García AO, Vizzeri G, Bowd C, et al. Reproducibility of RTVue retinal nerve fiber layer thickness and optic disc measurements and agreement with Stratus optical coherence tomography measurements. Am J Ophthalmol. 2009; 147:1067–74. 1074.e1.

19. Menke MN, Dabov S, Knecht P, Sturm V. Reproducibility of retinal thickness measurements in patients with age-related macular degeneration using 3D Fourier-domain optical coherence tomography (OCT) (Topcon 3D-OCT 1000). Acta Ophthalmol. 2011; 89:346–51.
20. Jittpoonkuson T, Garcia PM, Rosen RB. Correlation between fluorescein angiography and spectral-domain optical coherence tomography in the diagnosis of cystoid macular edema. Br J Ophthalmol. 2010; 94:1197–200.

21. Sakata LM, Lavanya R, Friedman DS, et al. Comparison of gonio-scopy and anterior segment ocular coherence tomography in de-tecting angle closure in different quadrants of the anterior chamber angle. Ophthalmology. 2008; 115:769–74.

22. Doors M, Tahzib NG, Eggink FA, et al. Use of anterior segment optical coherence tomography to study corneal changes after collagen cross-linking. Am J Ophthalmol. 2009; 148:844–51.e2.

23. Wang J, Aquavella J, Palakuru J, Chung S. Repeated measurements of dynamic tear distribution on the ocular surface after instillation of artificial tears. Invest Ophthalmol Vis Sci. 2006; 47:3325–9.

24. Chen Q, Wang J, Shen M, et al. Lower volumes of tear menisci in contact lens wearers with dry eye symptoms. Invest Ophthalmol Vis Sci. 2009; 50:3159–63.

25. Singh M, Aung T, Aquino MC, Chew PT. Utility of bleb imaging with anterior segment optical coherence tomography in clinical de-cision-making after trabeculectomy. J Glaucoma. 2009; 18:492–5.

26. Ciancaglini M, Carpineto P, Agnifili L, et al. Filtering bleb functionality: a clinical, anterior segment optical coherence tomography and in vivo confocal microscopy study. J Glaucoma. 2008; 17:308–17.

27. Gumus K, Crockett CH, Pflugfelder SC. Anterior segment optical coherence tomography: a diagnostic instrument for conjunctivochalasis. Am J Ophthalmol. 2010; 150:798–806.

Go to : 
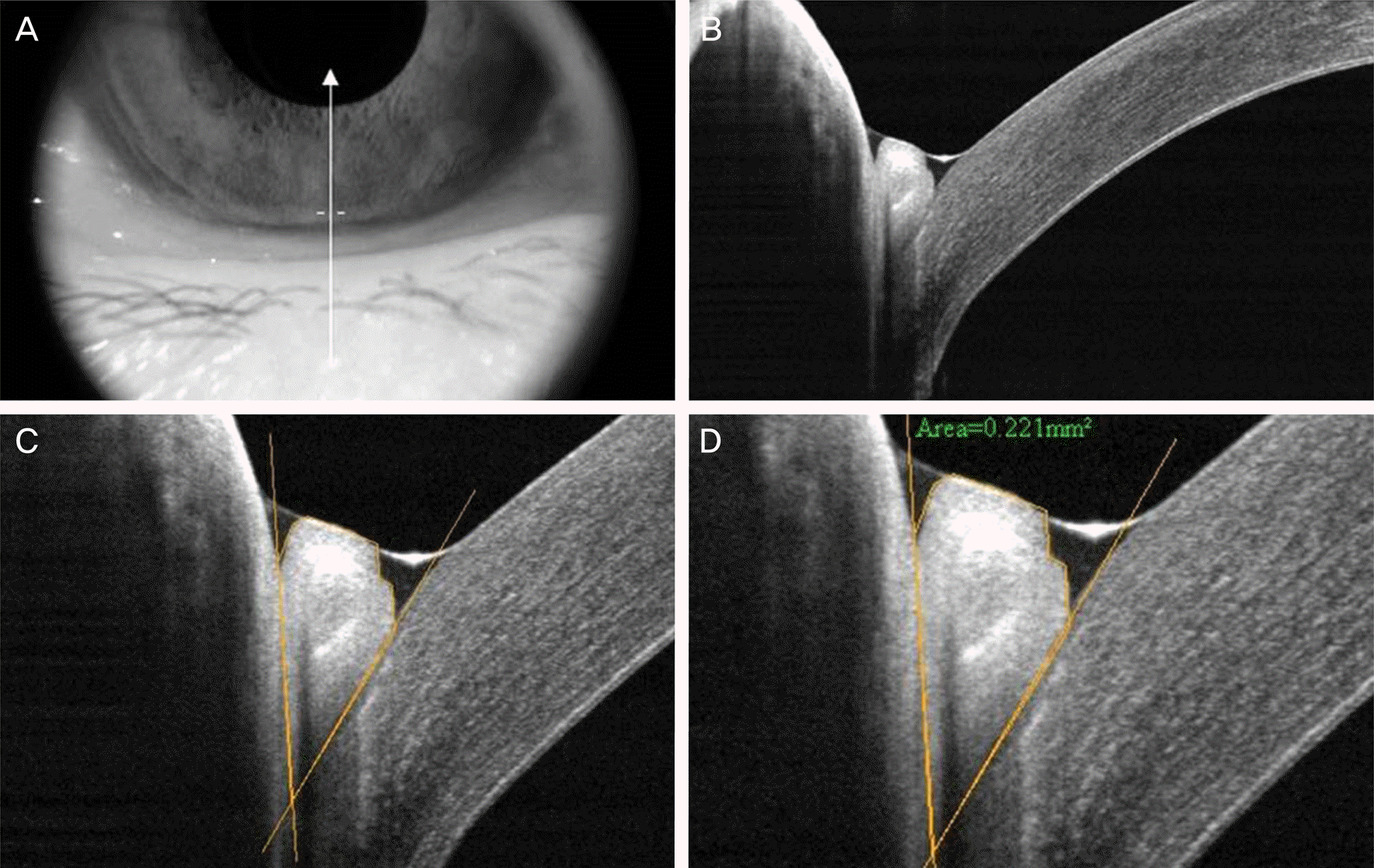 | Figure 2.Measurement of cross-sectional conjunctivochalasis area using RTVue-100. (A) Photograph of lower eyelid and cornea where the vertical scans were taken. Arrow means the location and direction scanned by RTvue. (B) The obtained scan image. (C) After the image was digitally magnified, tissue boundaries among the lower lid, pro-lapsed redundant conjunctiva, and bulbar conjunctiva/inferior part of the cornea were discriminated from each based on the different levels the brightness between tissues. (D) Cross-sectional area of the outlined conjunctivochalasis was measured as 0.221 mm2 using the instrument's software. |
 | Figure 3.Measurement of cross-sectional tear meniscus using RTVue-100. (A) Photograph of lower eyelid and cornea where the vertical scans were taken. Arrow means the location and direction scanned by RTvue. (B) After the image was digitally magnified. (C) Triangular-shaped tear meniscus is seen between the lower cornea and lower lid. (D) Cross-sectional area of the outlined tear meniscus was measure as 0.060 mm 2 using the instrument's software. |
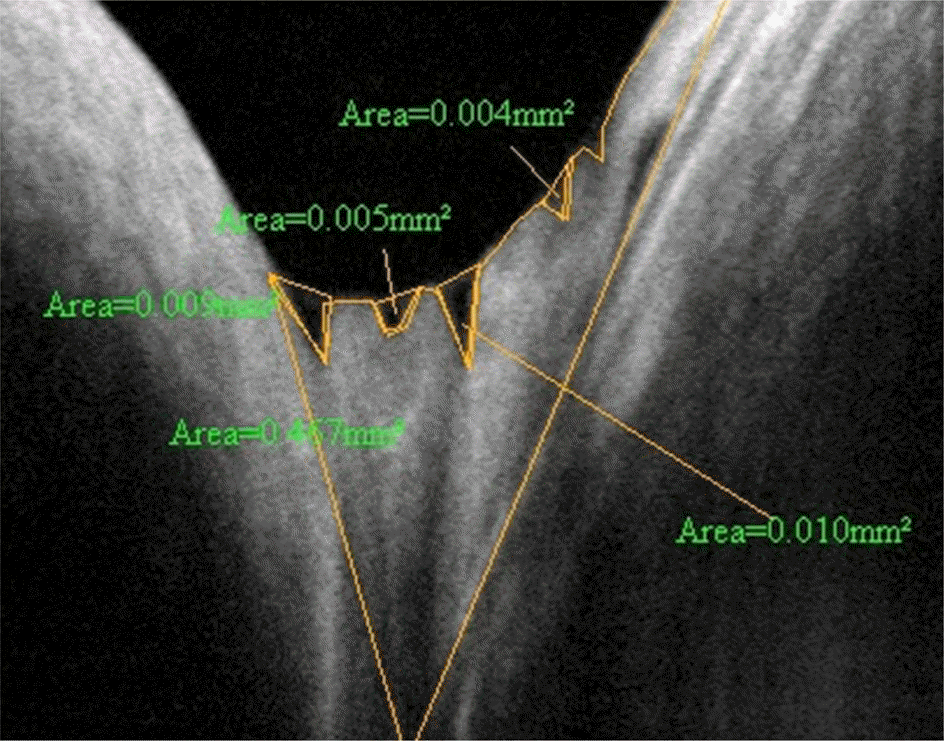 | Figure 4.Measurement of cross-sectional tear meniscus using RTVue-100. In case of many tear meniscus caused by tears dispersed among the conjunctival folds, each tear meniscus area was measured separately and then the total meniscus area was determined by summation of each tear meniscus area. |
 | Figure 5.Schematic design for conjunctivochalasis surgery. After resection of the conjunctivochalasis area (inside of dot-ted line) including the pingueculae (arrows), amniotic membrane transplantations was done in the exposed scleral area. |
Table 1.
Cross-sectional conjunctivochalasis area at baseline and following the surgery
| Location | Preoperative (mm2) | Postoperative (mm2) | p-value* |
|---|---|---|---|
| Center | 0.091 ± 0.134 | 0.001 ± 0.001 | <0.05 |
| Nasal 1 | 0.229 ± 0.195 | 0.001 ± 0.001 | <0.05 |
| Nasal 2 | 0.452 ± 0.275 | 0.005 ± 0.006 | <0.05 |
| Nasal 3 | 0.542 ± 0.409 | 0.012 ± 0.011 | <0.05 |
| Nasal average | 0.427 ± 0.254 | 0.007 ± 0.007 | <0.05 |
| Temporal 1 | 0.203 ± 0.155 | 0.001 ± 0.002 | <0.05 |
| Temporal 2 | 0.343 ± 0.192 | 0.007 ± 0.011 | <0.05 |
| Temporal 3 | 0.288 ± 0.139 | 0.015 ± 0.018 | <0.05 |
| Temporal average | 0.281 ± 0.149 | 0.008 ± 0.011 | <0.05 |
Table 2.
Cross-sectional tear meniscus area at baseline and following the surgery
| Location | Preoperative (mm2) | Postoperative (mm2) | p-value* |
|---|---|---|---|
| Center | 0.03 ± 0.02 | 0.01 ± 0.00 | 0.073 |
| Nasal 1 | 0.02 ± 0.02 | 0.04 ± 0.04 | 0.415 |
| Nasal 2 | 0.02 ± 0.02 | 0.02 ± 0.02 | 0.789 |
| Nasal 3 | 0.02 ± 0.02 | 0.03 ± 0.04 | 0.545 |
| Nasal average | 0.02 ± 0.02 | 0.03 ± 0.04 | 0.513 |
| Temporal 1 | 0.04 ± 0.03 | 0.02 ± 0.01 | 0.377 |
| Temporal 2 | 0.02 ± 0.03 | 0.01 ± 0.01 | 0.689 |
| Temporal 3 | 0.02 ± 0.03 | 0.07 ± 0.05 | 0.211 |
| Temporal average | 0.03 ± 0.03 | 0.04 ± 0.04 | 0.104 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download