Abstract
Methods
We analyzed filamentary keratitis in 11 eyes with corneal occlusion due to blepharospasm, ptosis and paralytic strabismus, 11 eyes with keratoconjunctivitis sicca, and 13 eyes with both corneal occlusion and keratoconjunctivitis sicca. The following protocols were used for filament analyses: slit lamp examination for counting filament numbers, impression cytology for grading mucin content, and histopathology (hematoxylin and eosin staining) and immunohistochemistry (with AE1/AE3, MUC1, 5AC, 16) for histological analysis.
Results
Slit lamp biomicrosopy showed a significantly smaller number of filaments in the corneal occlusion group (11 eyes, 2.7 ± 1.4) than the keratoconjunctivitis sicca group (11 eyes, 5.1 ± 2.2, p = 0.009). In impression cytology, the standard photograph grade of mucin content was significantly higher in the corneal occlusion group (2.7 ± 0.9) than the keratoconjunctivitis sicca group (1.9 ± 0.8, p = 0.034). On pathological exam, filaments showed a central filament with peripheral mucin. The central filaments were composed of core and cortex, and the core contained multiple deformed nuclei. However, the cortex was not stained by AE1/AE3, MUC1, 5AC, 16.
Go to : 
References
1. Beetham WP. Filamentary Keratitis. Trans Am Ophthalmol Soc. 1935; 33:413–35.
2. Wright P. Filamentary keratitis. Trans Ophthalmol Soc U K. 1975; 95:260–6.
3. Albietz J, Sanfilippo P, Troutbeck R, Lenton LM. Management of filamentary keratitis associated with aqueous-deficient dry eye. Optom Vis Sci. 2003; 80:420–30.

5. Kakizaki H, Zako M, Mito H, Iwaki M. Filamentary keratitis improved by blepharoptosis surgery: two cases. Acta Ophthalmol Scand. 2003; 81:669–71.

6. Tanioka H, Yokoi N, Komuro A, et al. Investigation of the corneal filament in filamentary keratitis. Invest Ophthalmol Vis Sci. 2009; 50:3696–702.

7. Choi SK, Paek NH, Hong WS. Clinical observation of the filamentary keratitis. J Korean Ophthalmol Soc. 1974; 15:323–6.
8. Unal M, Sarici A. Filamentary keratopathy caused by corneal occlusion by large-angle paralytic strabismus. Cornea. 2006; 25:1105–6.
9. Scott AB, Kennedy RA, Stubbs HA. Botulinum A toxin injection as a treatment for blepharospasm. Arch Ophthalmol. 1985; 103:347–50.

10. Berke RN. Congenital ptosis; a classification of 200 cases. Arch Ophthal. 1949; 41:188–97.
11. Gilbard JP, Farris RL, Santamaria J 2nd. Osmolarity of tear micro-volumes in keratoconjunctivitis sicca. Arch Ophthalmol. 1978; 96:677–81.

12. Gao J, Schwalb TA, Addeo JV, et al. The role of apoptosis in the pathogenesis of canine keratoconjunctivitis sicca: the effect of topical Cyclosporin A therapy. Cornea. 1998; 17:654–63.
13. Fox RI, Chan R, Michelson JB, et al. Beneficial effect of artificial tears made with autologous serum in patients with keratoconjunctivitis sicca. Arthritis Rheum. 1984; 27:459–61.

14. Baum JL. The Castroviejo Lecture. Prolonged eyelid closure is a risk to the cornea. Cornea. 1997; 16:602–11.
15. Good WV, Whitcher JP. Filamentary keratitis caused by corneal occlusion in large-angle strabismus. Ophthalmic Surg. 1992; 23:66.

16. Merjava S, Neuwirth A, Tanzerova M, Jirsova K. The spectrum of cytokeratins expressed in the adult human cornea, limbus and per-ilimbal conjunctiva. Histol Histopathol. 2011; 26:323–31.
18. Argüeso P, Spurr-Michaud S, Russo CL, et al. MUC16 mucin is expressed by the human ocular surface epithelia and carries the H185 carbohydrate epitope. Invest Ophthalmol Vis Sci. 2003; 44:2487–95.
19. Inatomi T, Spurr-Michaud S, Tisdale AS, Gipson IK. Human corneal and conjunctival epithelia express MUC1 mucin. Invest Ophthalmol Vis Sci. 1995; 36:1818–27.
20. Inatomi T, Spurr-Michaud S, Tisdale AS, et al. Expression of secre-tory mucin genes by human conjunctival epithelia. Invest Ophthalmol Vis Sci. 1996; 37:1684–92.
21. Absolon MJ, Brown CA. Acetylcysteine in keratoconjunctivitis sicca. Br J Ophthalmol. 1968; 52:310–6.

23. Fayet B, Bernard JA, Ammar J, et al. Treatment of dry eye by tem-porary punctum plug. Comparison with a control group. J Fr Ophtalmol. 1990; 13:123–33.
24. Tuberville AW, Frederick WR, Wood TO. Punctal occlusion in tear deficiency syndromes. Ophthalmology. 1982; 89:1170–2.

25. Gumus K, Lee S, Yen MT, Pflugfelder SC. Botulinum toxin injection for the management of refractory filamentary keratitis. Arch Ophthalmol. 2012; 130:446–50.

26. Kitazawa K, Yokoi N, Watanabe A, et al. Eyelid surgery for refractory filamentary keratitis. Nihon Ganka Gakkai Zasshi. 2011; 115:693–8.
Go to : 
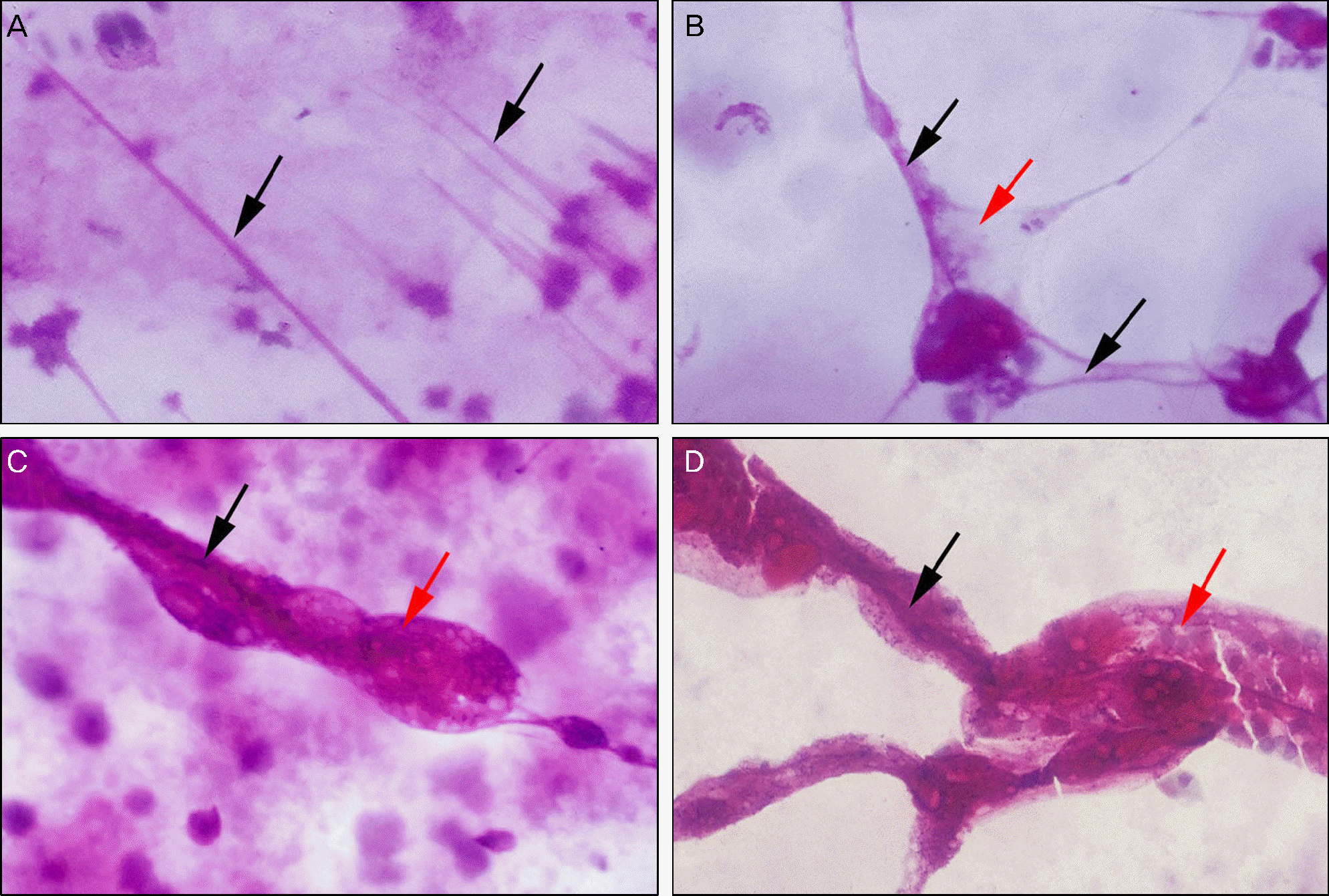 | Figure 1.Standard photographs of mucin debridement amount. (A) Grade I: filament (black arrow) without mucin debridement. (B) Grade II: filaments with scanty mucin (red arrow) debridement. (C) Grade III: filament with moderate amount of mucin debridement (D) Grade IV: filaments with large amount of mucin debridement (PAS stain, ×100). |
 | Figure 2.The number of filaments observed on silt lamp examination. The corneal occlusion group shows signigicantly less number of filaments than other groups (p < 0.05). KCS = keratoconjunctivitis sicca group; CO = corneal occlusion group. * Mann-Whitney U-test; p < 0.05 was considered statistically significant. |
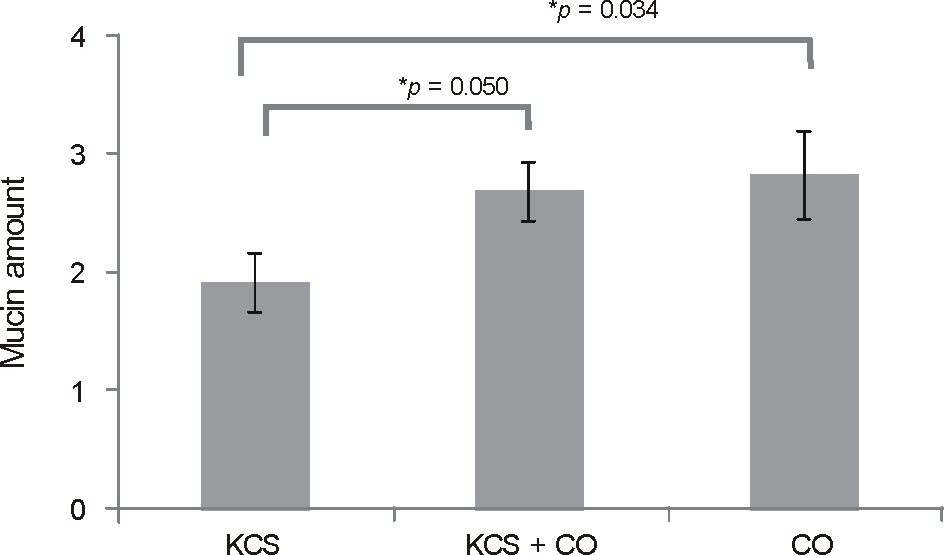 | Figure 3.The grade of mucin amount by standard photograph observed on impression cytology. The keratoconjunctivitis sicca group shows signigicantly less amount of mucin than other groups (p < 0.05). KCS = keratoconjunctivitis sicca group; CO = corneal occlusion group. * Mann-Whitney U-test; p < 0.05 was considered statistically significant. |
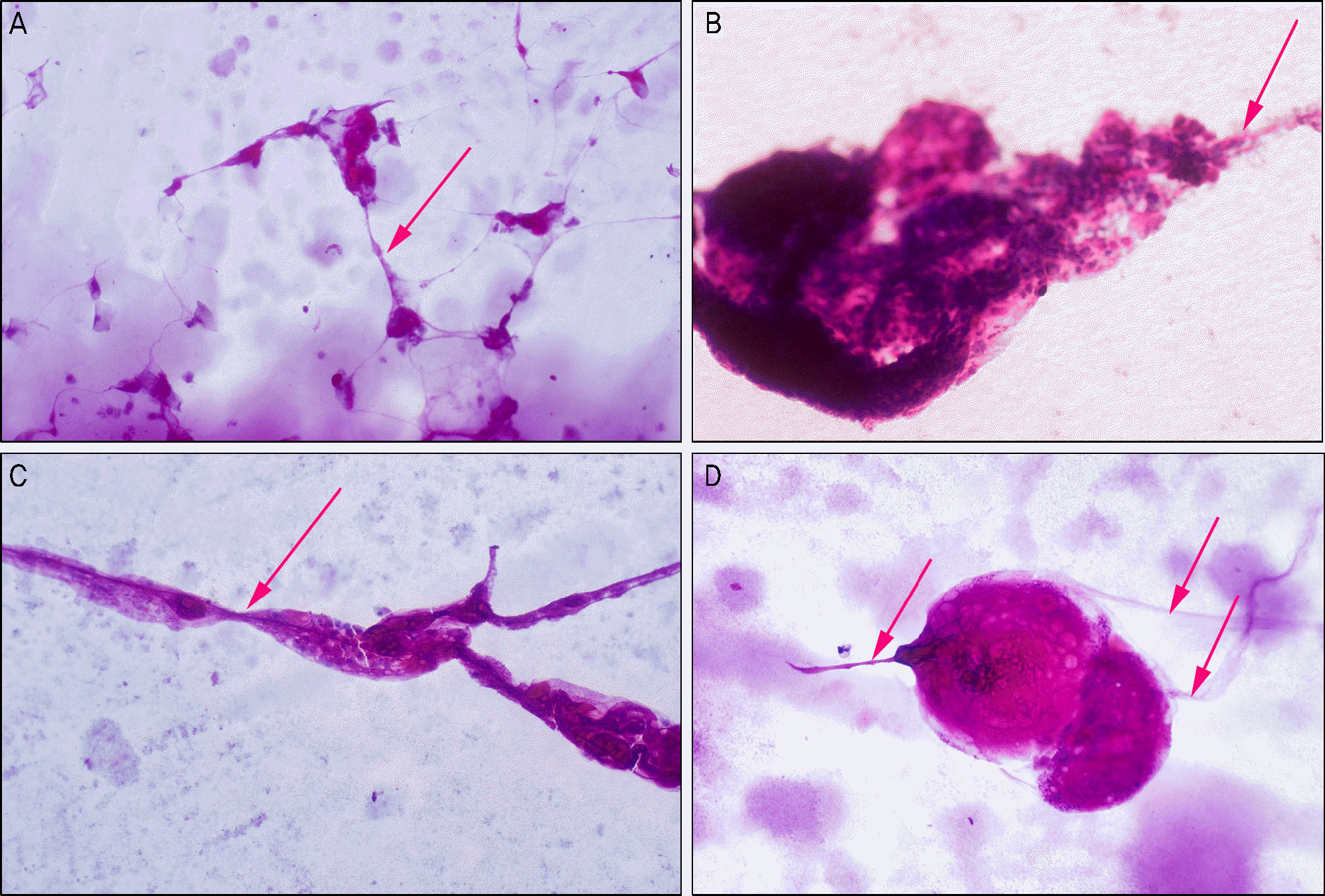 | Figure 4.Photographs of various filiament forms examined by impression cytology. The presence of central fiber (red arrows) are commonly observed (A, B, C, D: PAS stain, ×40). |
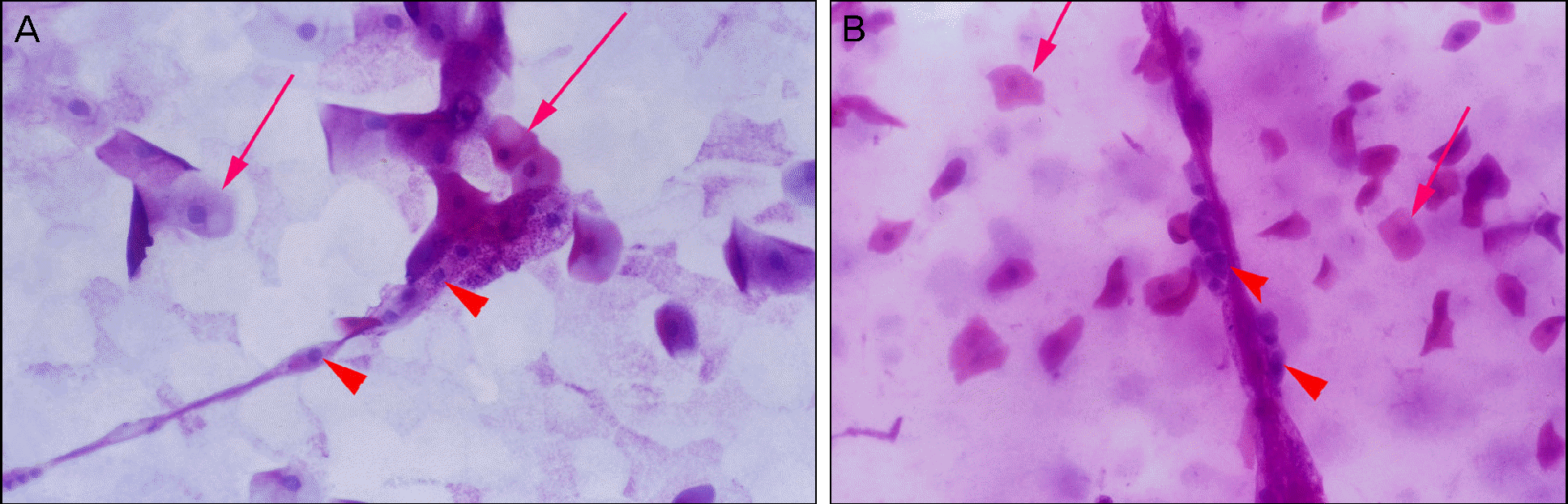 | Figure 5.Photographs obtained by impression cytology near the base of filaments. They show homogeneous cells without important nuclear alterations, with eosinophilic cytoplasm and a N:C ratio of 1:8 (arrow). In the base of filaments, there were metaplastic cells with large N:C ratio, meta-chromatic cytoplasm and frequent nuclear alterations such as twin-nuclei (arrow head) (A, B: PAS stain, ×200). |
 | Figure 6.Pathologic specimens stained with H & E. Central fibers were definitely confirmed (black arrows). The deformed nuclei were aligned like bricks within the central fibers (red arrows). The vicinity of the central fibers was composed of atypical substances with mucin and dead cells (A: ×400 longitudinal sectio; B: ×400 transverse section; C: ×1000 longitudinal section; D: ×1000 transverse section, H & E stain). |
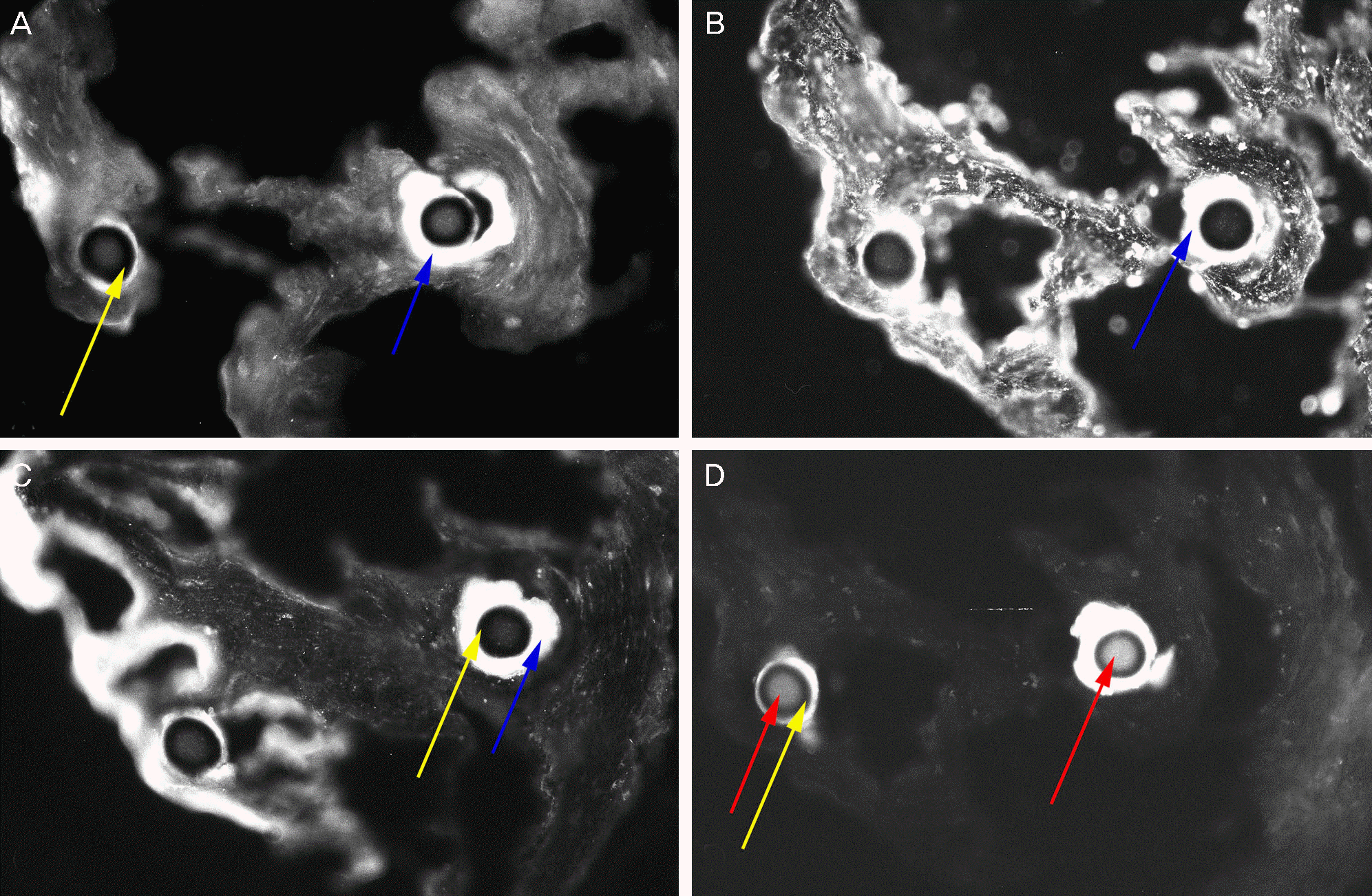 | Figure 7.In immunostaining, excluding the central fiber, the peripheral area is stained with MUC1 (A), MUC5AC (B), and MUC16 (C) (blue arrows). The core of central fibers is stained with the pan-cytokeratin marker AE1/AE3 (D) (red arrows), but the cortex of central fibers (yellow arrows) is not staind with MUC1, 5AC, 16 or AE1/AE3 (×400). |
Table 1.
Demographics of study population
Table 2.
The cause and treatment of 35 eyes of patients with filamentary keratitis patients examined




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download