Abstract
Purpose
To report a case of Phoma glomerata keratitis occurring in recurrent herpes simplex keratitis cicatrix.
Case summary
A 63-year-old male patient was admitted to our hospital with complaints of abrupt visual deterioration and ocular pain in his left eye. He was treated for recurrent herpes simplex keratitis in the same eye 12 years prior. Because central desmatocele was observed as a result of advanced corneal stromal melting, Gram staining, Potassium Hydroxide (KOH) mount, and culture were performed in corneal scrape specimens. On microbiological evaluation, a Phoma species was detected and Phoma glomerata was diagnosed using DNA sequencing method. Two consecutive amniotic membrane transplantations were performed with topical antifungal agents. The lesion was not improved when using topical amphotericin B and natamycin eyedrops, thus fluconazole eyedrops were used additionally. The corneal infection was resolved with central thick opacification.
Conclusions
In the present case, herpetic keratitis was the main underlying causative factor because the patient had no past history of trauma. When diverse appearances of keratitis occur in herpes simplex keratitis patients, clinicians need to consider the concurrence of fungal infection, especially Phoma glomerata, a rare fungal organism.
References
1. Zaitz C, Heins-Vaccari EM, de Freitas RS, et al. Subcutaneous phaeohyphomycosis caused by Phoma cava. Report of a case and review of the literature. Rev Inst Med Trop Sao Paulo. 1997; 39:43–8.
2. Baker JG, Salkin IF, Forgacs P, et al. First report of subcutaneous phaeohyphomycosis of the foot caused by Phoma minutella. J Clin Microbiol. 1987; 25:2395–7.

3. Bakerspigel A. The isolation of phoma hibernica from a lesion on a leg. Sabouraudia. 1970; 7:261–4.
4. Bakerspigel A, Lowe D, Rostas A. The isolation of phoma eupyrena from a human lesion. Arch Dermatol. 1981; 117:362–3.

5. Dooley DP, Beckius ML, Jeffery BS, et al. Phaeohyphomycotic cutaneous disease caused by Pleurophoma in a cardiac transplant patient. J Infect Dis. 1989; 159:503–7.

6. Gordon MA, Salkin IF, Stone WB. Phoma (Peyronellaea) as zoopathogen. Sabouraudia. 1975; 13:329–33.
7. Hirsh AH, Schiff TA. Subcutaneous phaeohyphomycosis caused by an unusual pathogen: Phoma species. J Am Acad Dermatol. 1996; 34:679–80.

8. Oh CK, Kwon KS, Lee JB, et al. Subcutaneous pheohyphomycosis caused by Phoma species. Int J Dermatol. 1999; 38:874–6.
10. Rosen T, Rinaldi MJ, Tschen JA, et al. Cutaneous lesions due to Pleurophoma (Phoma) complex. South Med J. 1996; 89:431–3.

11. Shukla NP, Rajak RK, Agarwal GP, Gupta DK. Phoma minutispora as a human pathogen. Mykosen. 1984; 27:255–8.

12. Stone MS, Rosen T, Clarridge J. Phaeohyphomycosis due to coelomycetes organisms. Int J Dermatol. 1988; 27:404–5.

13. Young NA, Kwon-Chung KJ, Freeman J. Subcutaneous abscess caused by Phoma sp. resembling Pyrenochaeta romeroi: unique fungal infection occurring in immunosuppressed recipient of renal allograft. Am J Clin Pathol. 1973; 59:810–6.
15. Errera MH, Barale PO, Nourry H, et al. Usefulness of voriconazole in treatment of Phoma glomerata after penetrating injury. J Fr Ophtalmol. 2008; 31:62–6.
16. Malecha MA, Tarigopula S, Malecha MJ. Successful treatment of Paecilomyces lilacinus keratitis in a patient with a history of herpes simplex virus keratitis. Cornea. 2006; 25:1240–2.

Figure 1.
Slit lamp photography of Phoma glomerata keratitis. (A) At initial presentation, descemetocele due to progressive stromal melting was seen at the inferotemporal cornea. Deep and wide circular sulcus was observed around the descemetocele (arrow). (B) and (C) Postoperative 4 days after transplantation of amniotic membrane, dark-brown pigmentations appeared around the corneal lesion (arrow heads). Thinned and protruded cornea were obvious by slitbeam observations. (D) Postoperative 3 months, the lesion was completely healed, but central corneal pigmentation was remained (asterisk).
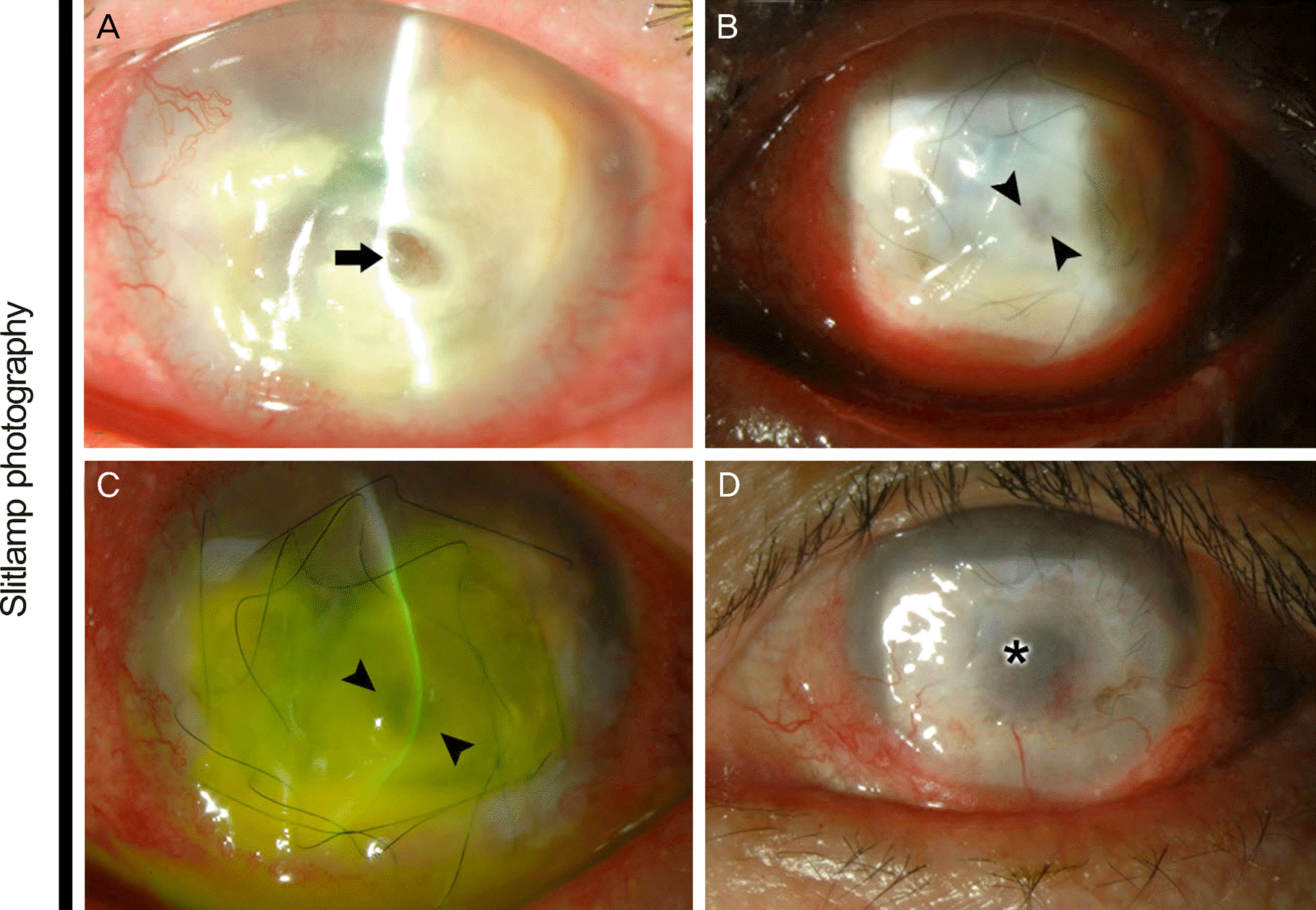
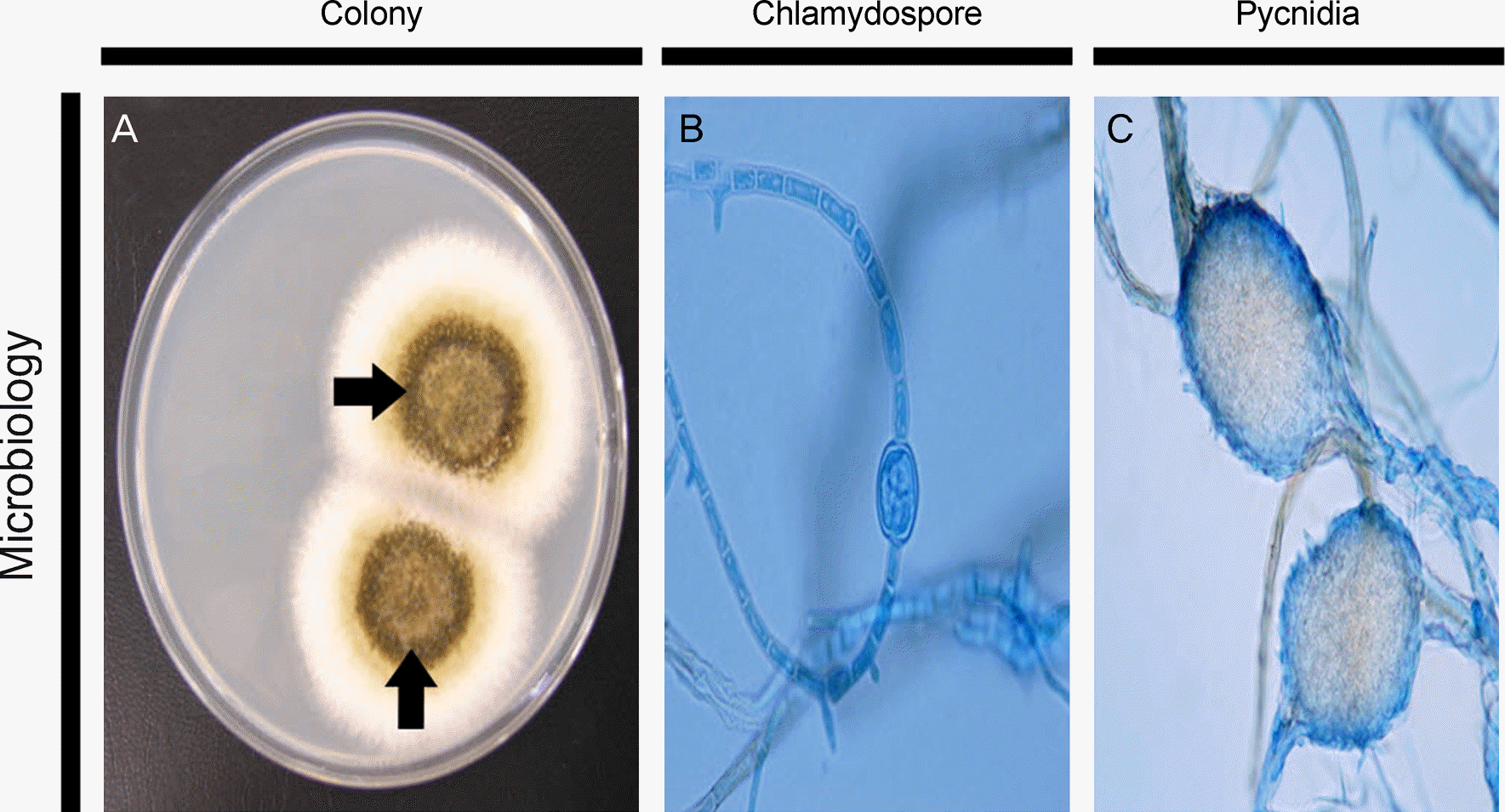
Figure 2.
Microbiological findings of Phoma glomerata keratitis. (A) The colony of Phoma glomerata on Sabouraud dextrose agar at 25°C for 6 days. Grayish-brown, broadly spreading colonies with sparse aerial mycelium were seen (arrows). (B) Microscopic finding of Phoma glomerata (lactophenol cotton blue stain, × 1000) Chlamydospore, (C) pycnidia.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download