Abstract
Purpose
To determine predictive factors associated with visual outcome after treatment for myopic choroidal neovascularization (mCNV).
Methods
Medical records of the patients who underwent photodynamic therapy (PDT), intravitreal anti-vascular endothelial growth factor (Anti-VEGF) injection, or combination therapy of PDT and Anti-VEGF for myopic CNV, and followed up for more than a year, were reviewed retrospectively. Multiple linear regression analyses were performed to evaluate the predictive factors significantly associated with the visual outcome at 1 year after the treatment.
Results
Mean best-corrected visual acuity (BCVA) of 45 eyes of 45 patients showed statistically significant improvement 1 year after the treatment with a mean of 3.5 line improvement (p < 0.01, Wilcoxon signed rank test). Age, 1-month BCVA after treatment and treatment type appeared to be associated with the 1-year visual outcome after treatment for mCNV (p = 0.033, p < 0.001, and p = 0.044, respectively, multivariate linear regression analysis).
Conclusions
Younger age (less than 40 years), better 1-month BCVA after treatment, intravitreal Anti-VEGF monotherapy were associated with improved visual outcome after treatment for mCNV. In particular, 1-month BCVA after treatment is a useful indicator to predict therapeutic response after treatment for mCNV.
References
2. Hotchkiss ML, Fine SL. Pathologic myopia and choroidal neovascularization. Am J Ophthalmol. 1981; 91:177–83.

3. Ohno-Matsui K, Yoshida T, Futagami S, et al. Patchy atrophy and lacquer cracks predispose to the development of choroidal neovascularisation in pathological myopia. Br J Ophthalmol. 2003; 87:570–3.

4. Bottoni F, Tilanus M. The natural history of juxtafoveal and subfoveal choroidal neovascularization in high myopia. Int Ophthalmol. 2001; 24:249–55.
5. Yoshida T, Ohno-Matsui K, Yasuzumi K, et al. Myopic choroidal neovascularization: a 10-year follow-up. Ophthalmology. 2003; 110:1297–305.
6. Yoshida T, Ohno-Matsui K, Ohtake Y, et al. Long-term visual prognosis of choroidal neovascularization in high myopia: a comparison between age groups. Ophthalmology. 2002; 109:712–9.
7. Blinder KJ, Blumenkranz MS, Bressler NM, et al. Verteporfin therapy of subfoveal choroidal neovascularization in pathologic myopia: 2-year results of a randomized clinical trial - VIP report no. 3. Ophthalmology. 2003; 110:667–73.
8. Lam DS, Chan WM, Liu DT, et al. Photodynamic therapy with verteporfin for subfoveal choroidal neovascularisation of pathologic myopia in Chinese eyes: a prospective series of 1 and 2 year follow up. Br J Ophthalmol. 2004; 88:1315–9.

9. Schnurrbusch UE, Jochmann C, Wiedemann P, Wolf S. Quantitative assessment of the long-term effect of photodynamic therapy in patients with pathologic myopia. Graefes Arch Clin Exp Ophthalmol. 2005; 243:829–33.

10. Pece A, Isola V, Vadalà M, Matranga D. Photodynamic therapy with verteporfin for subfoveal choroidal neovascularization secondary to pathologic myopia: long-term study. Retina. 2006; 26:746–51.
11. Chen YS, Lin JY, Tseng SY, et al. Photodynamic therapy for Taiwanese patients with pathologic myopia: a 2-year follow-up. Retina. 2007; 27:839–45.
12. Cohen SY. Anti-VEGF drugs as the 2009 first-line therapy for choroidal neovascularization in pathologic myopia. Retina. 2009; 29:1062–6.

13. Byeon SH, Kwon OW, Lee SC, et al. Indocyanine green angio-graphic features of myopic subfoveal choroidal neovascularization as a prognostic factor after photodynamic therapy. Korean J Ophthalmol. 2006; 20:18–25.

14. Kuo JZ, Ong FS, Yeung L, et al. Predictive factors for visual outcome to intravitreal bevacizumab in young Chinese patients with myopic choroidal neovascularization. Retina. 2011; 31:1835–40.

15. Yoon JU, Kim YM, Lee SJ, et al. Prognostic factors for visual outcome after intravitreal anti-vegf injection for naive myopic choroidal neovascularization. Retina. 2012; [Epub ahead of print].

16. Axer-Siegel R, Ehrlich R, Yassur Y, et al. Photodynamic therapy for age-related macular degeneration in a clinical setting: visual results and angiographic patterns. Am J Ophthalmol. 2004; 137:258–64.

17. Hampton GR, Kohen D, Bird AC. Visual prognosis of disciform degeneration in myopia. Ophthalmology. 1983; 90:923–6.

18. Secrétan M, Kuhn D, Soubrane G, Coscas G. Long-term visual outcome of choroidal neovascularization in pathologic myopia: natural history and laser treatment. Eur J Ophthalmol. 1997; 7:307–16.

19. Brancato R, Pece A, Avanza P, Radrizzani E. Photocoagulation scar expansion after laser therapy for choroidal neovascularization in degenerative myopia. Retina. 1990; 10:239–43.

20. Pece A, Brancato R, Avanza P, et al. Laser photocoagulation of choroidal neovascularization in pathologic myopia: long-term results. Int Ophthalmol. 1994-1995; 18:339–44.

21. Ruiz-Moreno JM, Montero JA. Long-term visual acuity after argon green laser photocoagulation of juxtafoveal choroidal neovascularization in highly myopic eyes. Eur J Ophthalmol. 2002; 12:117–22.

22. Virgili G, Varano M, Giacomelli G, et al. Photodynamic therapy for nonsubfoveal choroidal neovascularization in 100 eyes with pathologic myopia. Am J Ophthalmol. 2007; 143:77–82.

23. Pece A, Vadalà M, Isola V, Matranga D. Photodynamic therapy with verteporfin for juxtafoveal choroidal neovascularization in pathologic myopia: a long-term follow-up study. Am J Ophthalmol. 2007; 143:449–54.

24. Gibson J. Photodynamic therapy with verteporfin for juxtafoveal choroidal neovascularisation secondary to pathological myopia. Eye (Lond). 2005; 19:829–30.

25. Lam DS, Liu DT, Fan DS, et al. Photodynamic therapy with verteporfin for juxtafoveal choroidal neovascularization secondary to pathologic myopia-1-year results of a prospective series. Eye (Lond). 2005; 19:834–40.

26. Gelisken F, Inhoffen W, Hermann A, et al. Verteporfin photodynamic therapy for extrafoveal choroidal neovascularisation in pathologic myopia. Graefes Arch Clin Exp Ophthalmol. 2004; 242:926–30.

27. Chan WM, Lai TY, Wong AL, et al. Combined photodynamic therapy and intravitreal triamcinolone injection for the treatment of choroidal neovascularisation secondary to pathological myopia: a pilot study. Br J Ophthalmol. 2007; 91:174–9.

28. Marticorena J, Gomez-Ulla F, Fernandez M, et al. Combined photodynamic therapy and intravitreal triamcinolone acetonide for the treatment of myopic subfoveal choroidal neovascularization. Am J Ophthalmol. 2006; 142:335–7.

29. Rishi P, Rishi E, Venkataraman A, et al. Photodynamic mono-therapy or combination treatment with intravitreal triamcinolone acetonide, bevacizumab or ranibizumab for choroidal neovascularization associated with pathological myopia. Indian J Ophthalmol. 2011; 59:242–6.

30. Manzano RP, Peyman GA, Khan P, Kivilcim M. Testing intravitreal toxicity of bevacizumab (Avastin). Retina. 2006; 26:257–61.

31. Gharbiya M, Giustolisi R, Allievi F, et al. Choroidal neovascularization in pathologic myopia: intravitreal ranibizumab versus bevacizumab-a randomized controlled trial. Am J Ophthalmol. 2010; 149:458–64.

32. Matsuo M, Honda S, Matsumiya W, et al. Comparison between anti-vascular endothelial growth factor therapy and photodynamic therapy for myopic choroidal neovascularization. Eur J Ophthalmol. 2012; 22:210–5.

33. Ikuno Y, Nagai Y, Matsuda S, et al. Two-year visual results for older Asian women treated with photodynamic therapy or bevacizumab for myopic choroidal neovascularization. Am J Ophthalmol. 2010; 149:140–6.

34. Peiretti E, Vinci M, Fossarello M. Intravitreal bevacizumab as a treatment for choroidal neovascularisation secondary to myopia: 4-year study results. Can J Ophthalmol. 2012; 47:28–33.

35. Axer-Siegel R, Ehrlich R, Weinberger D, et al. Photodynamic therapy of subfoveal choroidal neovascularization in high myopia in a clinical setting: visual outcome in relation to age at treatment. Am J Ophthalmol. 2004; 138:602–7.

36. Ikuno Y, Sayanagi K, Soga K, et al. Intravitreal Bevacizumab for choroidal neovascularization attributable to pathological myopia: one-year results. Am J Ophthalmol. 2009; 147:94–100.

Figure 1.
Mean best corrected visual acuity (log MAR) changes of all eyes after the first treatment. M ean best-corrected visual acuity (BCVA) of forty five eyes of 45 patients showed statistically significant improvement at each time points after the treatment (p < 0.01, Wilcoxon signed rank test).
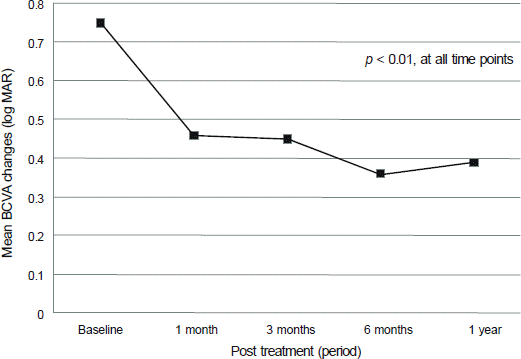
Figure 2.
Mean best corrected visual acuity (BCVA, log MAR) changes at 1 year after the first treatment of the groups. M ean BCVA of all groups improved significantly at 1 year after the treatment (p = 0.028, p < 0.01, p = 0.037, respectively, in group A, B and C, W ilcoxon signed rank test).
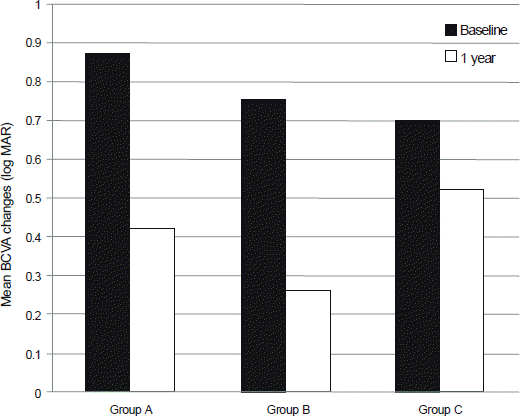
Table 1.
Demographics and Ocular Characteristics of the Study Population
Table 2.
Results of the linear regression models for the single and multiple covariate on the 1 year BCVA (log M AR) after 1st treatment
Table 3.
Demographics of each treatment group
| PDT (Group A) | Anti-VEGF (Group B) | PDT+ Anti-VEGF (Group C) | p-value* | |
|---|---|---|---|---|
| Number of patients (eyes) | 7 | 21 | 17 | - |
| Mean age (years) | 41 | 43.5 | 35.2 | 0.063 |
| Gender (M:F) | 2 : 5 | 2 : 19 | 9 : 8 | 0.429 |
| Mean refractive error (diopter) | -10.8 | -10.9 | -11.6 | 0.603 |
| CNV location (subfoveal : juxtafoveal) | 7 : 0 | 4 : 17 | 6 : 11 | 0.391 |
| CNV size (μm) | 1291 | 1282 | 1374 | 0.418 |
| Mean baseline CMT (μm) | 253 | 284 | 291 | 0.486 |
| Mean baseline BCVA (log MAR) | 0.87 ± 0.41 | 0.75 ± 0.35 | 0.70 ± 0.43 | 0.401 |
| Mean post Tx 1 year BCVA (log MAR) | 0.32 ± 0.27 | 0.26 ± 0.25 | 0.52 ± 0.45 | 0.044 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download