Abstract
Purpose
To investigate the effect of ROCK inhibitor Y27632 on the human corneal endothelial cell proliferation in vitro and in vivo.
Methods
Using corneal endothelial cells isolated and cultured from human donor cornea, we compared the effect of Y27632 (10 μM) on the proliferation in vitro by flow cytometry analysis. For the evaluation of the effect of Y27632 (10 mM) in vivo, corneal thickness and wound area were analyzed for the corneal endothelial wound rabbit model induced by transcorneal freezing.
Results
Ki67 positive cells were increased in the Y27632 group (9.1 ± 4.1 %) than the control group (8.0 ± 5.9 %), whereas annexin V positive cells in the Y27632 group (2.9 ± 1.0 %) were decreased compared to the control group (4.2 ± 2.2 %). However these were not statistically significant. Wound area after Y27632 application in animal model is concerned, the control group showed significant smaller area (45.6 ± 0.6 mm2) compared to the Y27632 group (49.3 ± 0.8 mm2; p = 0.029, Mann-Whitney U test), however, these were not significantly different from the baseline. Corneal thickness was not different between the two groups.
Go to : 
References
1. Bourne WM. Clinical estimation of corneal endothelial pump function. Trans Am Ophthalmol Soc. 1998; 96:229–39. discussion 239-42.
2. Riley MV, Winkler BS, Starnes CA, et al. Regulation of corneal endothelial barrier function by adenosine, cyclic AMP, and protein kinases. Invest Ophthalmol Vis Sci. 1998; 39:2076–84.
3. Joyce NC. Proliferative capacity of the corneal endothelium. Prog Retin Eye Res. 2003; 22:359–89.

4. Zhu C, Joyce NC. Proliferative response of corneal endothelial cells from young and older donors. Invest Ophthalmol Vis Sci. 2004; 45:1743–51.

5. Miyata K, Drake J, Osakabe Y, et al. Effect of donor age on morphologic variation of cultured human corneal endothelial cells. Cornea. 2001; 20:59–63.

6. Engelmann K, Böhnke M, Friedl P. Isolation and long-term cultivation of human corneal endothelial cells. Invest Ophthalmol Vis Sci. 1988; 29:1656–62.
7. Okumura N, Ueno M, Koizumi N, et al. Enhancement on primate corneal endothelial cell survival in vitro by a ROCK inhibitor. Invest Ophthalmol Vis Sci. 2009; 50:3680–7.

8. Okumura N, Koizumi N, Ueno M, et al. Enhancement of corneal endothelium wound healing by Rho-associated kinase (ROCK) inhibitor eye drops. Br J Ophthalmol. 2011; 95:1006–9.

9. Riento K, Ridley AJ. Rocks: multifunctional kinases in cell behaviour. Nat Rev Mol Cell Biol. 2003; 4:446–56.

10. Moore M, Marroquin BA, Gugliotta W, et al. Rho kinase inhibition initiates apoptosis in human airway epithelial cells. Am J Respir Cell Mol Biol. 2004; 30:379–87.

11. Shibata R, Kai H, Seki Y, et al. Rho-kinase inhibition reduces neointima formation after vascular injury by enhancing Bax expression and apoptosis. J Cardiovasc Pharmacol. 2003; 42:S43–7.

12. Svoboda KK, Moessner P, Field T, Acevedo J. ROCK inhibitor (Y27632) increases apoptosis and disrupts the actin cortical mat in embryonic avian corneal epithelium. Dev Dyn. 2004; 229:579–90.

13. Matsubara M, Tanishima T. Wound-healing of the corneal endothelium in the monkey: a morphometric study. Jpn J Ophthalmol. 1982; 26:264–73.
14. Van Horn DL, Hyndiuk RA. Endothelial wound repair in primate cornea. Exp Eye Res. 1975; 21:113–24.

15. Leung T, Chen XQ, Manser E, Lim L. The p160 RhoA-binding kinase ROK alpha is a member of a kinase family and is involved in the reorganization of the cytoskeleton. Mol Cell Biol. 1996; 16:5313–27.

16. Lee JG, Kay EP. FGF-2-mediated signal transduction during endothelial mesenchymal transformation in corneal endothelial cells. Exp Eye Res. 2006; 83:1309–16.

17. Lee JG, Song JS, Smith RE, Kay EP. Human corneal endothelial cells employ phosphorylation of p27(Kip1) at both ser10 and Thr187 sites for FGF-2-mediated cell proliferation via PI 3-kinase. Invest Ophthalmol Vis Sci. 2011; 52:8216–23.
18. Chevrier V, Piel M, Collomb N, et al. The Rho-associated protein kinase p160ROCK is required for centrosome positioning. J Cell Biol. 2002; 157:807–17.

19. Kosako H, Yoshida T, Matsumura F, et al. Rho-kinase/ROCK is involved in cytokinesis through the phosphorylation of myosin light chain and not ezrin/radixin/moesin proteins at the cleavage furrow. Oncogene. 2000; 19:6059–64.

20. Coleman ML, Marshall CJ, Olson MF. RAS and RHO GTPases in G1-phase cell-cycle regulation. Nat Rev Mol Cell Biol. 2004; 5:355–66.

21. Hu W, Bellone CJ, Baldassare JJ. RhoA stimulates p27(Kip) degradation through its regulation of cyclin E/CDK2 activity. J Biol Chem. 1999; 274:3396–401.

22. Olson MF, Ashworth A, Hall A. An essential role for Rho, Rac, and Cdc42 GTPases in cell cycle progression through G1. Science. 1995; 269:1270–2.
23. Welsh CF, Roovers K, Villanueva J, et al. Timing of cyclin D1 expression within G1 phase is controlled by Rho. Nat Cell Biol. 2001; 3:950–7.

24. Sebbagh M, Renvoizé C, Hamelin J, et al. Caspase-3-mediated cleavage of ROCK I induces MLC phosphorylation and apoptotic membrane blebbing. Nat Cell Biol. 2001; 3:346–52.

25. Ongusaha PP, Kim HG, Boswell SA, et al. RhoE is a pro-survival p53 target gene that inhibits ROCK I-mediated apoptosis in response to genotoxic stress. Curr Biol. 2006; 16:2466–72.

26. Chang J, Xie M, Shah VR, et al. Activation of Rho-associated coiled-coil protein kinase 1 (ROCK-1) by caspase-3 cleavage plays an essential role in cardiac myocyte apoptosis. Proc Natl Acad Sci U S A. 2006; 103:14495–500.

27. Bharadwaj S, Thanawala R, Bon G, et al. Resensitization of breast cancer cells to anoikis by tropomyosin-1: role of Rho kinase-dependent cytoskeleton and adhesion. Oncogene. 2005; 24:8291–303.

28. Koyanagi M, Takahashi J, Arakawa Y, et al. Inhibition of the Rho/ROCK pathway reduces apoptosis during transplantation of embryonic stem cell-derived neural precursors. J Neurosci Res. 2008; 86:270–80.

29. Miñambres R, Guasch RM, Perez-Aragó A, Guerri C. The RhoA/ROCK-I/MLC pathway is involved in the ethanol-induced apoptosis by anoikis in astrocytes. J Cell Sci. 2006; 119((Pt 2)):271–82.
30. Coleman ML, Sahai EA, Yeo M, et al. Membrane blebbing during apoptosis results from caspase-mediated activation of ROCK I. Nat Cell Biol. 2001; 3:339–45.

31. Davies SP, Reddy H, Caivano M, Cohen P. Specificity and mechanism of action of some commonly used protein kinase inhibitors. Biochem J. 2000; 351((Pt 1)):95–105.

32. Ishizaki T, Uehata M, Tamechika I, et al. Pharmacological properties of Y-27632, a specific inhibitor of rho-associated kinases. Mol Pharmacol. 2000; 57:976–83.
33. Chen J, Guerriero E, Lathrop K, SundarRaj N. Rho/ROCK signaling in regulation of corneal epithelial cell cycle progression. Invest Ophthalmol Vis Sci. 2008; 49:175–83.

34. Jampel HD, Roche N, Stark WJ, Roberts AB. Transforming growth factor-beta in human aqueous humor. Curr Eye Res. 1990; 9:963–9.
35. Dalma-Weiszhauz J, Blumenkranz M, Hartzer M, Hernandez E. Intraocular extracellular cyclic nucleotide concentrations: the influence of vitreous surgery. Graefes Arch Clin Exp Ophthalmol. 1993; 231:184–6.
36. Van Horn DL, Sendele DD, Seideman S, Buco PJ. Regenerative capacity of the corneal endothelium in rabbit and cat. Invest Ophthalmol Vis Sci. 1977; 16:597–613.
37. Kim SD. Study of endothelial regeneration in rabbits. J Korean Ophthamol Soc. 1979; 20:3–7.
Go to : 
 | Figure 1.(A) Cylindrical dry-ice block was prepared using 7.75 mm sized corneal trephine. (B) Dry-ice block was applied on the corneal surface of the animal for 25 seconds (C, D) for the induction of transcorneal freezing injury. |
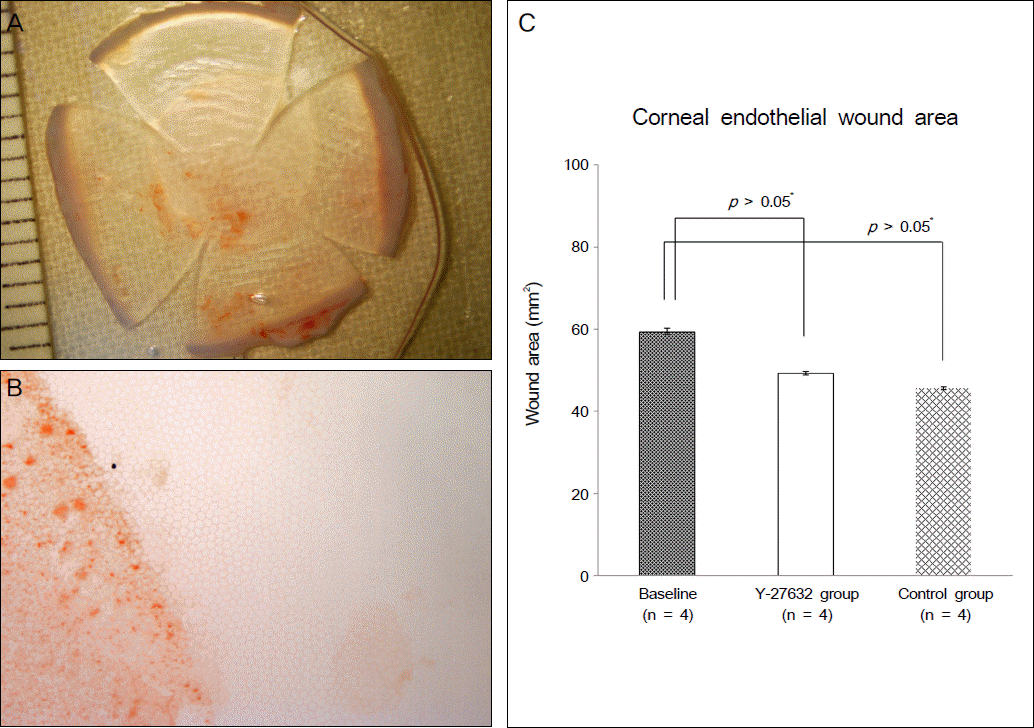 | Figure 2.(A) Alizarin red staining was performed using the cornea isolated 48 hours after corneal endothelial wound induction. Gross appearance and (B) microscopic appearance shows the area of the induced corneal endothelial wound. (C) There was no significant difference between Y-27632 group and control group for the wound area change from the baseline (*M ann-W hitney U test). |
 | Figure 3.(A) M orphological analysis of cultured corneal endothelial cells showed relatively uniform and similar sized appearance. Until the second passage cells were kept hexagonal confluent single cell layers and showed less phenotypical change due to the replicative scenecence. (B, C) Cells during primary culture and subcultures were stained positively by both the COL8A2 and Na/K ATPase. Different from intracellular homogenous distribution of COL8A2, Na/K ATPase was distinctively found around the cellular membrane. |
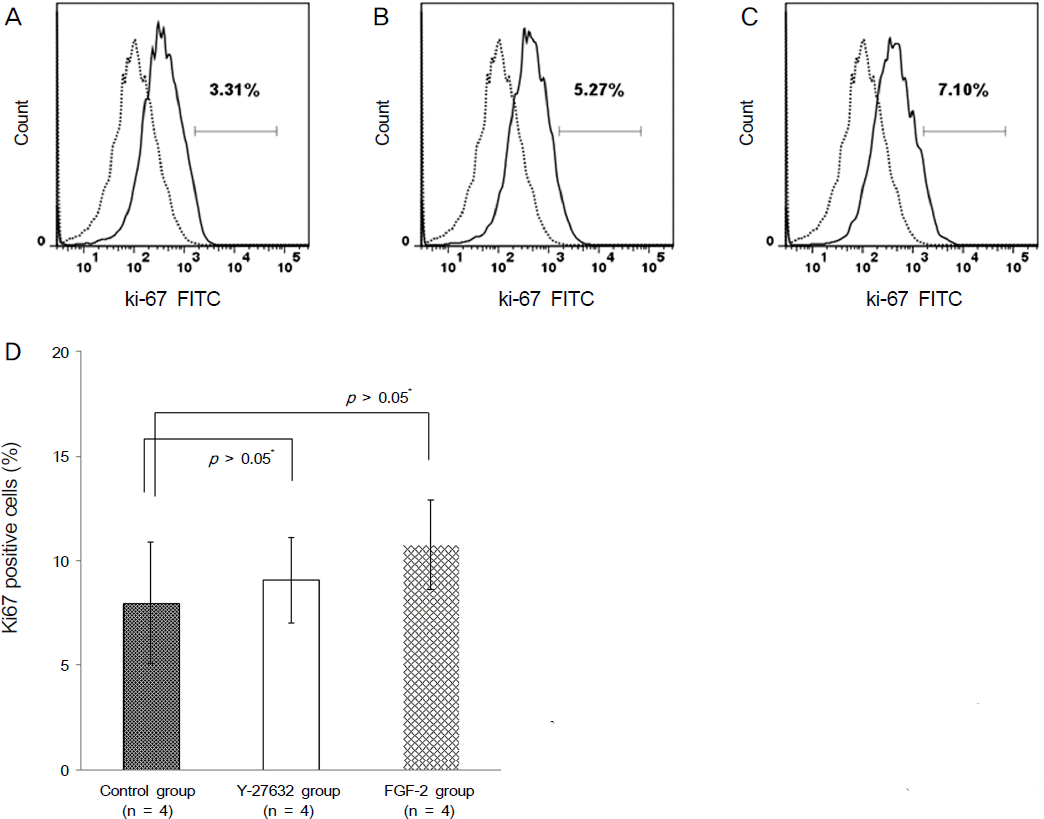 | Figure 4.(A-C) The number of Ki67 positive cells were analyzed using flow cytometry. (A: Control group, B: Y-27632 group, C: FGF-2 group) (D) Ki67 positive cells were increased in the Y-27632 group compared to the control group, however there was no statistical significance (*M ann-W hitney U test). |
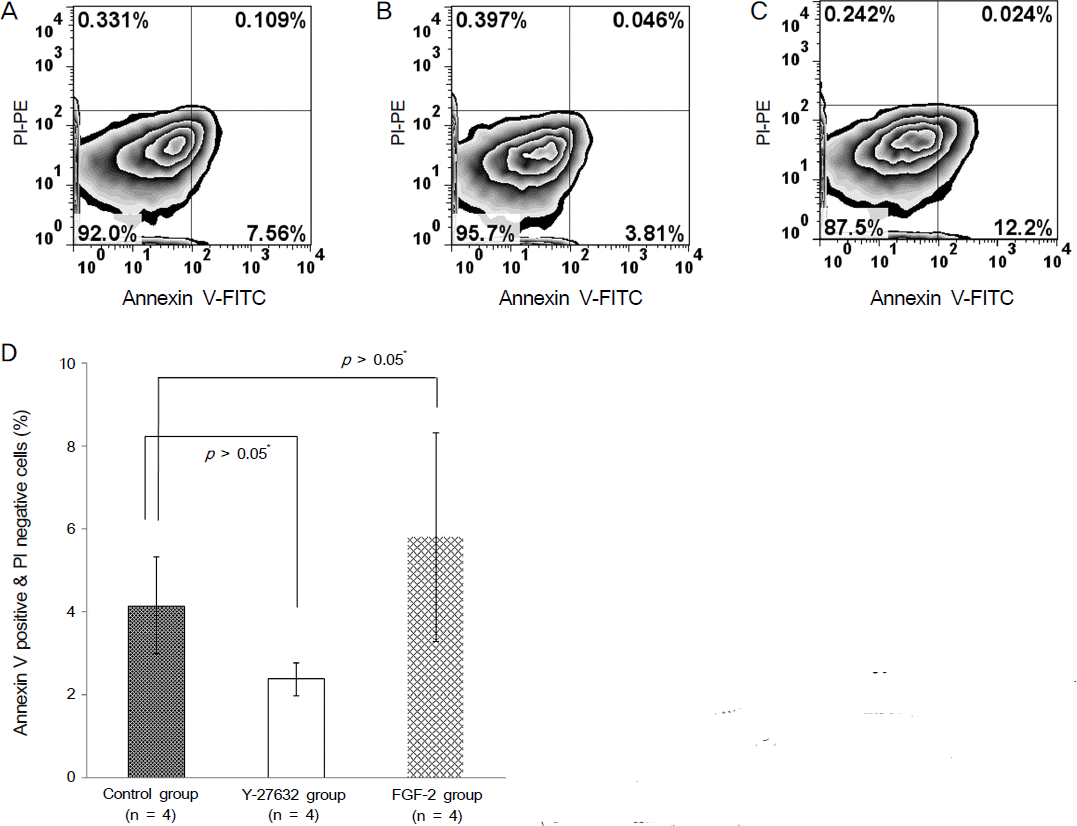 | Figure 5.(A-C) The number of annexin V positive cells were analyzed using flow cytometry. (A: Control group, B: Y-27632 group, C: FGF-2 group) (D) Annexin V positive and propidium iodide (PI) negative cells were decreased in the Y-27632 group compared the the control group with no statistical significance (*M ann-W hitney U test). |
 | Figure 6.Baseline endothelial cell profile analyzed with specular microscope was not different between the Y-27632 group (A) and the control group (B). |
 | Figure 7.(A, B) As far as corneal transparency and edema are concerned, there was no significant difference between the Y-27632 group and the control group 48 after corneal endothelial wound induction. However, Y-27632 group showed relatively more surface irregularity compared to the control group.(A: Y-27632 group, B: Control group) (C) Corneal thickness confirmed by ultrasound pachymetry 48 after corneal endothelial wound induction revealed no significant difference between the two groups (*Mann-W hitney U test). |
Table 1.
Baseline characteristics of corneal endothelial cells before wound induction
| Y-27632 group (n = 4) | Control group (n = 4) | p | |
|---|---|---|---|
| Specular microscopy | |||
| Cell Density | 2692 ± 293.3 | 2415 ± 330.1 | 0.400* |
| Coefficient of Variance | 32.8 ± 8.8 | 31.7 ± 3.7 | 0.857* |
| Hexagonality | 65.5 ± 10.4 | 54.7 ± 8.3 | 0.229* |
| Pachymetry | |||
| Corneal thickness (μm) | 341.6 ± 16.3 | 341.1 ± 28.5 | 0.857* |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download