Abstract
Purpose
To assess long-term comparisions of intravitreal bevacizumab injection (IVB group) with natural course (Natural course group) in patients with macular edema after branch retinal vein occlusion (BRVO).
Methods
Fifty-seven patients were included in this retrospective study and were followed-up for at least 12 months. Patients in the Natural course group (27 eyes of 27 patients) were enrolled before February 2007, and patients in the IVB group (30 eyes of 30 patients) underwent intravitreal bevacizumab injection as-needed after February 2007. The main effi-cacy outcome was reported as the mean change from baseline best corrected visual acuity (BCVA), and all patients were measured at the initial visit and after 1, 3, 6, and 12 months. Retreatment criteria included central subfield macular thick-ness (CSMT) greater than 300 μ m or increased CSMT of at least 100 μ m between visits.
Results
There were no statistically significant differences in age or initial BCVA between the 2 groups. Mean changes of BCVA at initial visit and at 3 and 12 months were 0.61 ± 0.48, 0.44 ± 0.46 and 0.34 ± 0.40 in the Natural course group and 0.67 ± 0.40, 0.31 ± 0.26 and 0.27 ± 0.25 in the IVB group, respectively. Improvement in BCVA was observed in both groups at 12 months, although the IVB group improved significantly more at 3 months and not at 12 months ( p = 0.018, p = 0.187, respectively).
References
1. Patz A. Argon laser photocoagulation for macular edema in branch vein occlusion. Am J Ophthalmol. 1984; 98:374–5.

2. Cugati S, Wang JJ, Knudtson MD, et al. Retinal vein occlusion and vascular mortality: pooled data analysis of 2 population-based cohorts. Ophthalmology. 2007; 114:520–4.
3. Mitchell P, Smith W, Chang A. Prevalence and associations of reti-nal vein occlusion in Australia. The Blue Mountains Eye Study. Arch Ophthalmol. 1996; 114:1243–7.

4. Greer DV, Constable IJ, Cooper RL. Macular oedema and retinal branch vein occlusion. Aust J Ophthalmol. 1980; 8:207–9.

5. Channa R, Smith M, Campochiaro PA. Treatment of macular ede-ma due to retinal vein occlusions. Clin Ophthalmol. 2011; 5:705–13.
6. Ryan SJ, Oick SB, Jampol LH, Haller JA. Retina. 3rd ed,. St. Louis: Mosby Co;2001. p. chap. 55–7.
7. Aiello LP, Avery RL, Arrigg PG, et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med. 1994; 331:1480–7.

8. Boyd SR, Zachary I, Chakravarthy U, et al. Correlation of in-creased vascular endothelial growth factor with neovascularization and permeability in ischemic central vein occlusion. Arch Ophthalmol. 2002; 120:1644–50.

9. Funk M, Kriechbaum K, Prager F, et al. Intraocular concentrations of growth factors and cytokines in retinal vein occlusion and the ef-fect of therapy with bevacizumab. Invest Ophthalmol Vis Sci. 2009; 50:1025–32.

10. Pe'er J, Folberg R, Itin A, et al. Vascular endothelial growth factor upregulation in human central retinal vein occlusion. Ophthalmology. 1998; 105:412–6.
11. Kriechbaum K, Michels S, Prager F, et al. Intravitreal Avastin for macular oedema secondary to retinal vein occlusion: a prospective study. Br J Ophthalmol. 2008; 92:518–22.

12. Rabena MD, Pieramici DJ, Castellarin AA, et al. Intravitreal bev-acizumab (Avastin) in the treatment of macular edema secondary to branch retinal vein occlusion. Retina. 2007; 27:419–25.

13. Campochiaro PA, Heier JS, Feiner L, et al. Ranibizumab for mac-ular edema following branch retinal vein occlusion: six-month pri-mary end point results of a phase III study. Ophthalmology. 2010; 117:1102–12.e1.
14. The Branch Vein Occlusion Study Group. Argon laser photo-coagulation for macular edema in branch vein occlusion. Am J Ophthalmol. 1984; 98:271–82.
15. Haller JA, Bandello F, Belfort R Jr, et al. Randomized, sham-con-trolled trial of dexamethasone intravitreal implant in patients with macular edema due to retinal vein occlusion. Ophthalmology. 2010; 117:1134–46.e3.

16. Jager RD, Aiello LP, Patel SC, Cunningham ET Jr. Risks of intra-vitreous injection: a comprehensive review. Retina. 2004; 24:676–98.

17. Krohne TU, Eter N, Holz FG, Meyer CH. Intraocular pharmacoki-netics of bevacizumab after a single intravitreal injection in humans. Am J Ophthalmol. 2008; 146:508–12.

19. Song IJ, Kim HW, Yun IH. Long-term effects of intravitreal bev-acizumab injection for macular edema secondary to branch retinal vein occlusion. J Korean Ophthalmol Soc. 2012; 53:283–90.

20. Magargal LE, Kimmel AS, Sanborn GE, Annesley WH Jr. Temporal branch retinal vein obstruction: a review. Ophthalmic Surg. 1986; 17:240–6.

21. Stefánsson E. The therapeutic effects of retinal laser treatment and vitrectomy. A theory based on oxygen and vascular physiology. Acta Ophthalmol Scand. 2001; 79:435–40.
22. Larsson J, Bauer B, Andréasson S. The 30-Hz flicker cone ERG for monitoring the early course of central retinal vein occlusion. Acta Ophthalmol Scand. 2000; 78:187–90.

23. Kim YG, Kim ES, Kim MS, et al. Early and late intravitreal bev-acizumab injection in macular edema due to branch retinal vein occlusion. J Korean Ophthalmol Soc. 2009; 50:1527–30.

Figure 1.
BCVA changes in each group. Vertical solid bars and vertical dotted bars are standard deviation of the IVB group and natural course group, respectively. There was a stat-istically signi fi cant group difference at month 3 (* p = 0.018). † Intravitreal bevacizumab injections within 7 days from first diagnosed day; ‡ Observation without any treatment.
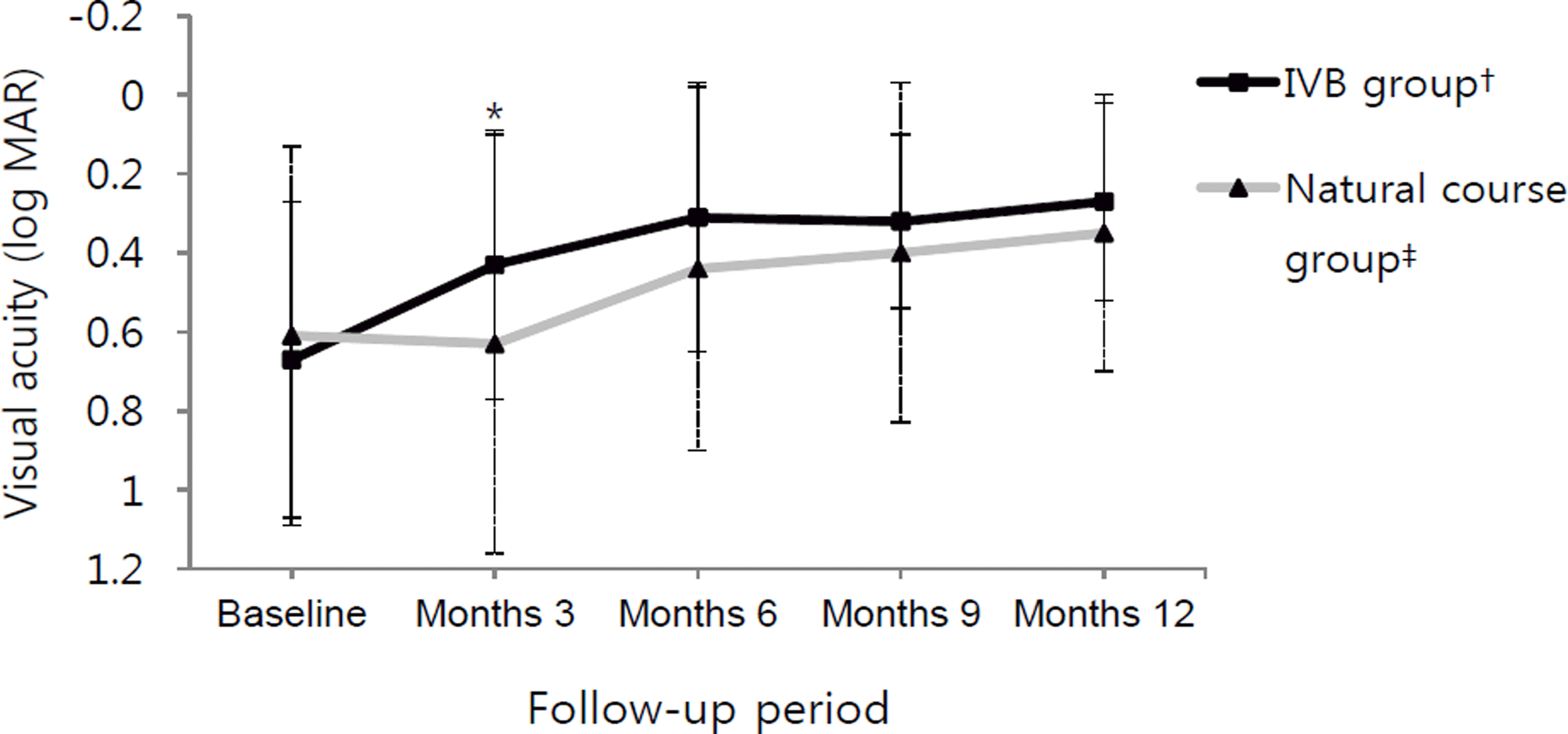
Figure 2.
Graph showing changes in central subfield macular thickness (CSMT) after intravitreal bevacizumab injection. The CSMT in the IVB group decreased at month 3, 6, and 12, and it was statistically different from baseline ( p = 0.002, p < 0.001, p = 0.001, respectively). CSMT = central subfield mac-ular thickness.
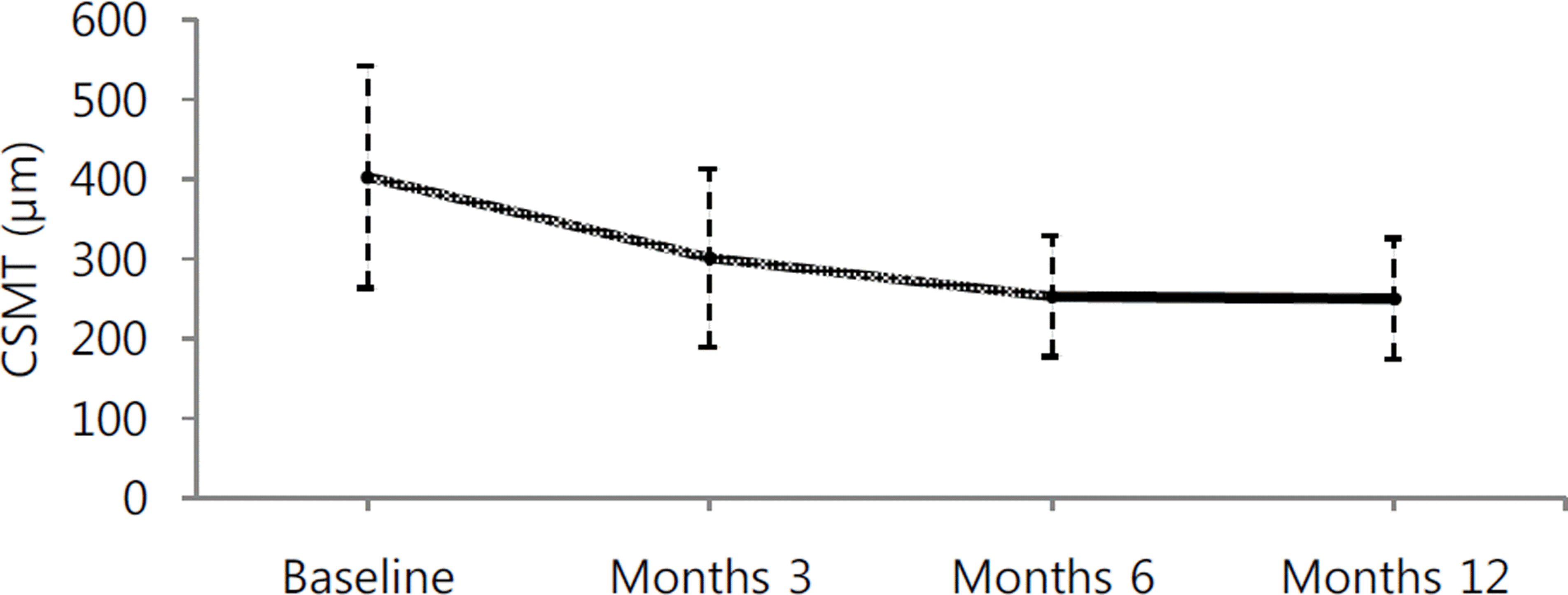
Table 1.
Demographic and clinical characteristics of each group
| Groups | Natural course group* | IVB group† | p-value‡ | |
|---|---|---|---|---|
| Baseline characteristics | ||||
| Number of eyes | 27 | 30 | ||
| Gender (male : female) | 3:24 | 11:19 | 0.03§ | |
| Eye (right : left) | 11:16 | 13:17 | 0.85Π | |
| Age (years) | 60.0 ± 11.3 | 61.7 ± 10.2 | 0.56# | |
| Systemic disease | Diabetes mellitus | 4 (15%) | 1 (3%) | 0.04# |
| Hypertension | 14 (52%) | 16 (55%) | 0.91# | |
| Duration of symptoms (days) | 34.1 ± 36.8 | 23.6 ± 24.4 | 0.23# | |
| BCVA (log MAR) | 0.61 ± 0.48 | 0.67 ± 0.40 | 0.33# |
Table 2.
Changes in BCVA in each groups
| BCVA | Baseline | Months 3 | Months 6 | Months 9 | Months 12 |
|---|---|---|---|---|---|
| IVB group* | 0.67 ± 0.40 | 0.43 ± 0.34 | 0.31 ± 0.34 | 0.32 ± 0.22 | 0.27 ± 0.25 |
| Mean changes from baseline (log MAR) | -0.24 ± 0.29 | -0.37 ± 0.28 | -0.36 ± 0.34 | -0.40 ± 0.31 | |
| p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | ||
| Natural course group† | 0.61 ± 0.48 | 0.63 ± 0.53 | 0.44 ± 0.46 | 0.40 ± 0.43 | 0.35 ± 0.40 |
| Mean changes from baseline (log MAR) | 0.02 ± 0.41 | -0.17 ± 0.44 | -0.21 ± 0.42 | -0.27 ± 0.46 | |
| p = 1.00 | p = 0.065 | p = 0.016 | p = 0.011 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download