Abstract
Purpose
To evaluate the response and cellular changes of cultured human corneal epithelium according to the concentration of topical cyclosporine.
Methods
Human corneal epithelial cells were exposed to cyclosporine A concentrations of 1 ug/ml (0.0001%), 10 ug/ml (0.001%), 100 ug/ml (0.01%), and 500 ug/ml (0.05%) for 5 and 10 minutes. An MTT-based colorimetric assay was performed to assess the metabolic activity of cellular proliferation and a lactate dehydrogenase (LDH) leakage assay was used to determine cellular toxicity. The modulations of extracellular matrix proteins such as PIP and laminin were evaluated. The levels of proinflammatory cytokine, TNF-α, and IL-1 were evaluated by ELISA kits.
Results
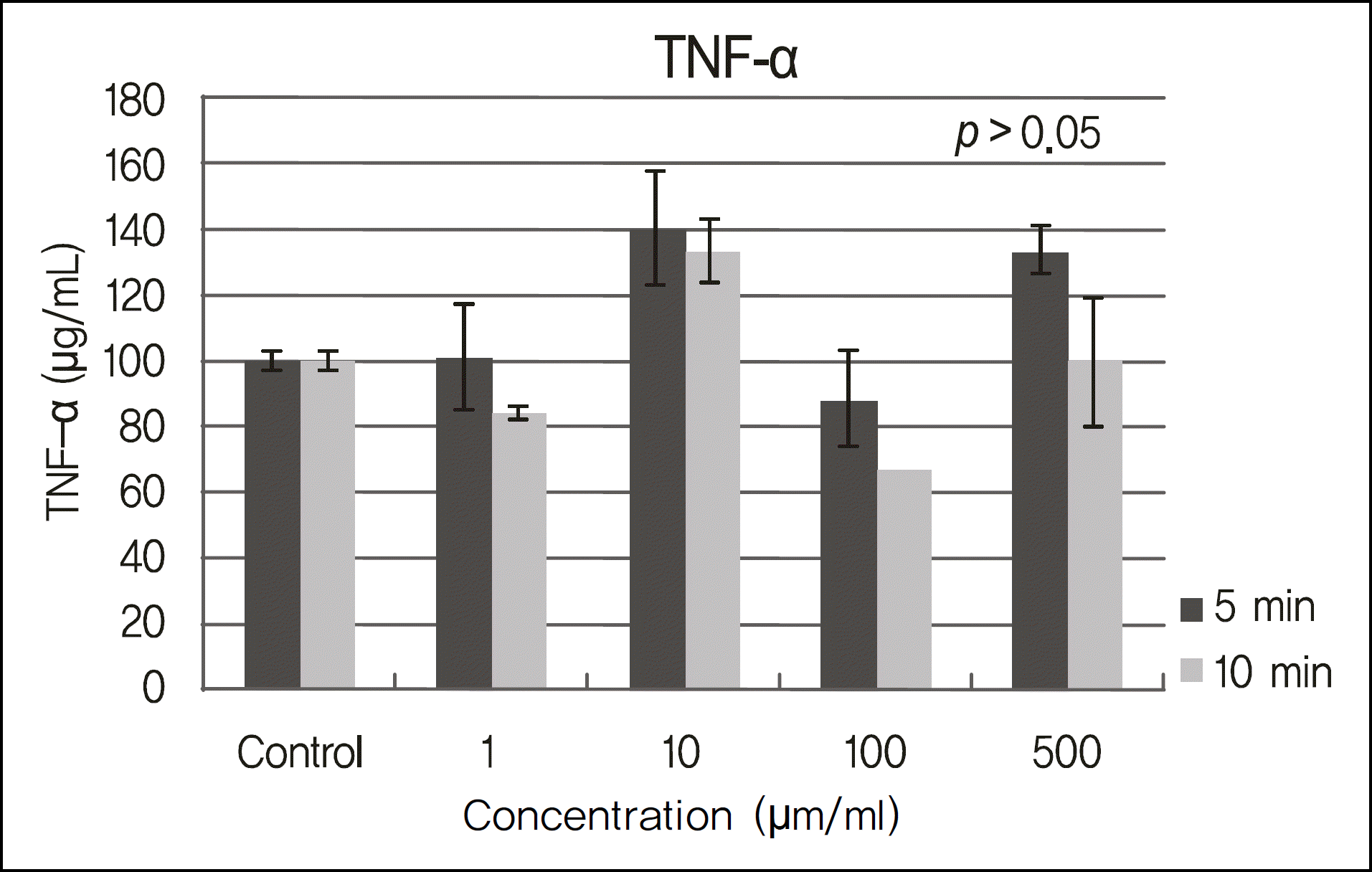
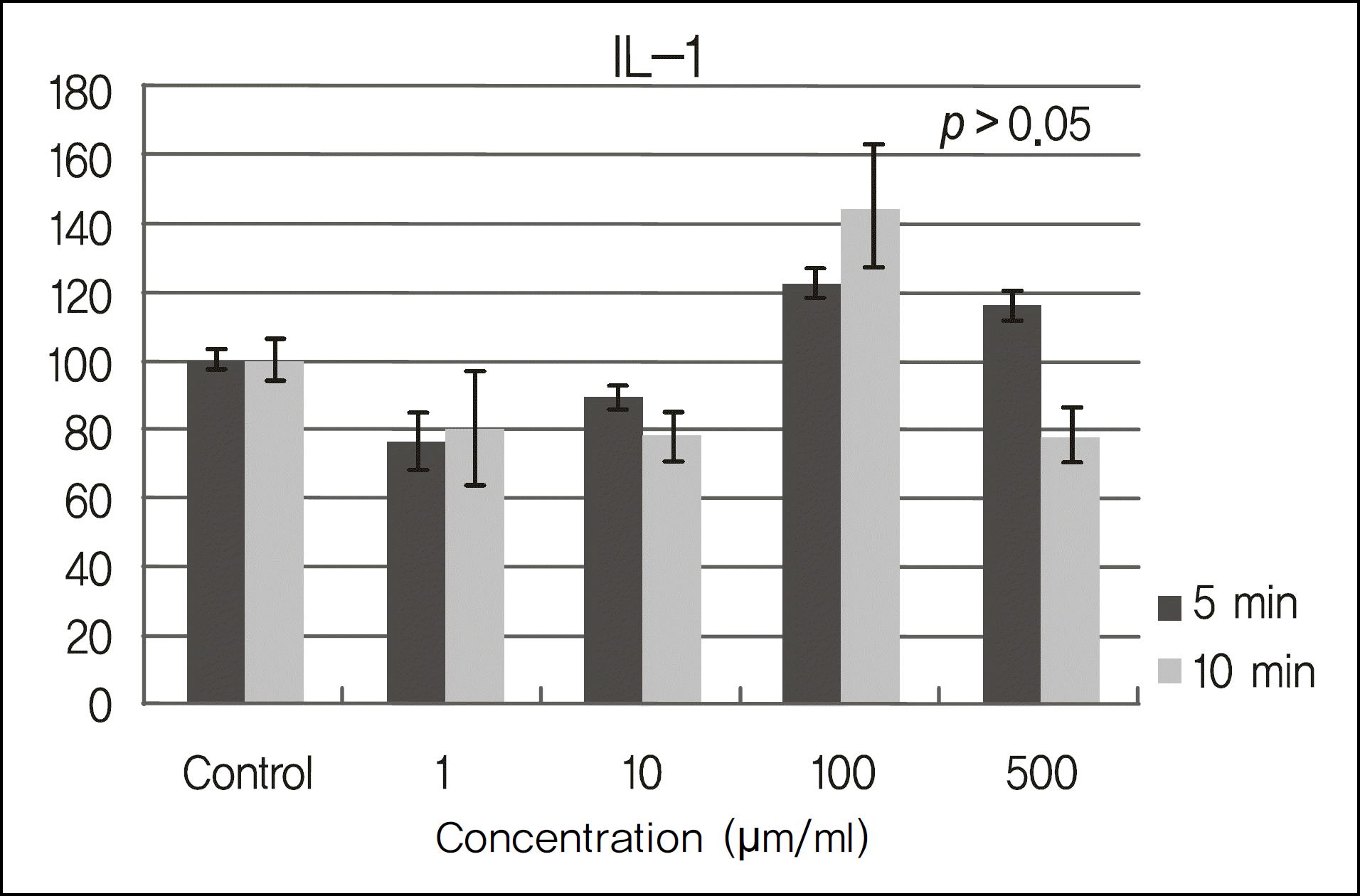
The inhibitory effects of human corneal epithelial cellular proliferation did not show a concentration- or exposure time-dependent response. Activity of LDH did not show a statistically significant difference for the different concentrations and exposure times. The inhibitory effects of extracellular matrix proteins such as PIP or laminin synthesized from human corneal epithelial cells were compared with those in the control group and showed a statistically significant difference at a cyclosporine concentration greater than 0.01%. Released TNF-α and IL-1 from human corneal epithelial cells did not show any difference according to concentration or cyclosporine exposure time.
Conclusions
To modulate human corneal epithelial cellular proliferation and the levels of extracellular matrix proteins such as PIP and laminin, a concentration of cyclosporine A greater than 0.01% for longer than 5 minutes of exposure is needed. There were little effects of cyclosporine A on proinflammatory cytokine secreted from corneal epithelial cells.
References
2. Dick AD, Azim M, Forrester JV. Immunosuppressive therapy for chronic uveitis: optimising therapy with steroids and cyclosporine A. Br J Ophthalmol. 1997; 81:1107–12.
3. Masuda K, Nakajima A, Urayama A, et al. Double-masked trial of cyclosporin versus colchicine and long-term open study of cyclosporin in Behcet's disease. Lancet. 1989; 1:1093–6.
4. Le Hoang P, Girard B, Deray G, et al. Cyclosporin A in the treatment of birdshot retinochoroidopathy. Transplant Proc. 1988; 20:128–30.
5. Stepkowski SM. Molecular targets for existing and novel immunosuppressive drugs. Expert Rev Mol Med. 2000; 2:1–23.

6. Minguez E, Tiestos MT, Cristobal JA, et al. Intraocular absorption of cyclosporin A eyedrops. J Fr Ophthalmol. 1992; 15:263–7.
7. Oh C, Apel AJ, Saville BA, et al. Local efficacy of cyclosporine in corneal transplant therapy. Curr Eye Res. 1994; 13:337–43.
8. BenEzra D, Matamoros N, Cohen E. Treatment of severe vernal keratoconjunctivitis with cyclosporine A eyedrops. Transplant Proc. 1988; 20:644–9.
9. Calonge M. The treatment of dry eye. Surv Opthahlmol. 2001; 45(suppl. 2):S227–39.
10. Díaz-Valle D, Benítez del Castillo JM, Castillo A, et al. Immunologic and clinical evaluation of postsurgical necrotizing sclerocorneal ulceration. Cornea. 1998; 17:371–5.

11. Sall K, Stevenson OD, Mundorf TK, Reis BL. Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. CsA Phase 3 Study Group. Ophthalmology. 2000; 107:631–9.
12. Nussenblatt RB, Palestine AG. Cyclosporin: immunology, pharmacology and therapeutic uses. Surv Ophthalmol. 1986; 31:159–69.
13. Power WJ, Mullaney P, Farrel M, et al. Effect of topical cyclosporin A on conjunctival T cells in patients with secondary Sjögren's syndrome. Cornea. 1993; 12:507–11.

14. Donnenfeld E, Pflugfelder SC. Topical ophthalmic cyclosporine: pharmacology and clinical uses. Surv Ophthalmol. 2009; 54:321–38.
15. Tatlipinar S, Akpek EK. Topical ciclosporin in the treatment of ocular surface disorder. Br J Ophthalmol. 2005; 89:1363–7.
16. Wiederholt M, Kössendrup D, Schulz W, Hoffmann F. Pharmacokinetic of topical cyclosporin A in the rabbit eye. Invest Ophthalmol Vis Sci. 1986; 27:519–24.
17. Acheampong AA, Shcakleton M, Tang-Liu DD, et al. Distrubution of cyclosporin A in ocular tissues after topical administration to albino rabbits and beagle dogs. Curr Eye Res. 1999; 18:91–103.
18. Lee LE, Shin WB, Lee JS. Effect of cyclosporine A 0.05% on human corneal epithelial cells. J Korean Ophthalmol. 2007; 48:1399–1409.

19. Garweg JG, Wegmann-Burns M, Goldblum D. Effects of daunor-ubicin, mitomycin C, azathiprine and cyclosporin A on human retinal pigmented epithelial, corneal endothelial and conjunctival cell lines. Graefes Arch Clin Exp Ophthalmol. 2006; 244:382–9.
20. Mauger TF, Craig EL. Havener's Ocular Pharmacology. 6th ed.St. Lousi: Mosby;1994. p. 28.
21. Singh G, Lindstrom RL, Doughman DJ. Cyclosporin A on human corneal endothelium. Cornea. 1984; 3:272–7.

22. BenEzra D, Antebe I, Maftzir G. Differential effect of cyclosporin A on lymphocyte and keratocyte proliferation. Invest Ophthalmol Vis Sci. 1987; 28:42.
23. Tonoe O, Yamanaka O, Okada Y, et al. Collagen biosynthesis and cellular proliferation after filtering surgery in rabbits. Folia Ophthalmol Jpn. 1995; 46:242–5.
24. Berman B, Duncan MR. Pentoxifylline inhibits normal hyman dermal fibroblast in vitro proliferation, collagen, glycosaminoglycan, and fibronectin production, and increases collagenase activity. J Invest Dermatol. 1989; 92:605–10.
25. Leonardi A, DeFranchis G, Fregona IA, et al. Effects of cyclosporine A on human conjunctival fibroblasts. Arch Ophthalmol. 2001; 119:1512–7.
26. Bankers-Fulbright JL, Kalli KR, McKean DJ. Interleukin-1 signal transduction. Life Sciences. 1996; 59:61–83.

27. Li DQ, Lokeshwar BL, Solomon A, et al. Regulation of MMP-9 production by human corneal epithelial cells. Exp Eye Res. 2001; 73:449–59.

28. Bargagna-Mohan P, Strissel KJ, Fini ME. Regulation of gelatinase B production in corneal cells is independent of autocrine IL-1alpha. Invest Ophthalmol Vis Sci. 1999; 40:784–9.
Figure 1.
The absorption rate of the water-insolubale formazan dye in corneal epithelial cell exposed in 1 ug/ml (0.0001%), 10 ug/ml (0.001%), 100 ug/ml (0.01%), 500 ug/ml (0.05%) cyclosporine by a scanning spectrometer (ELISA reader). Cellular viabilities were not significantly different in proportion to concentration increase. As exposure time lengthened, cell viabilities were not significantly different (p > 0.05).
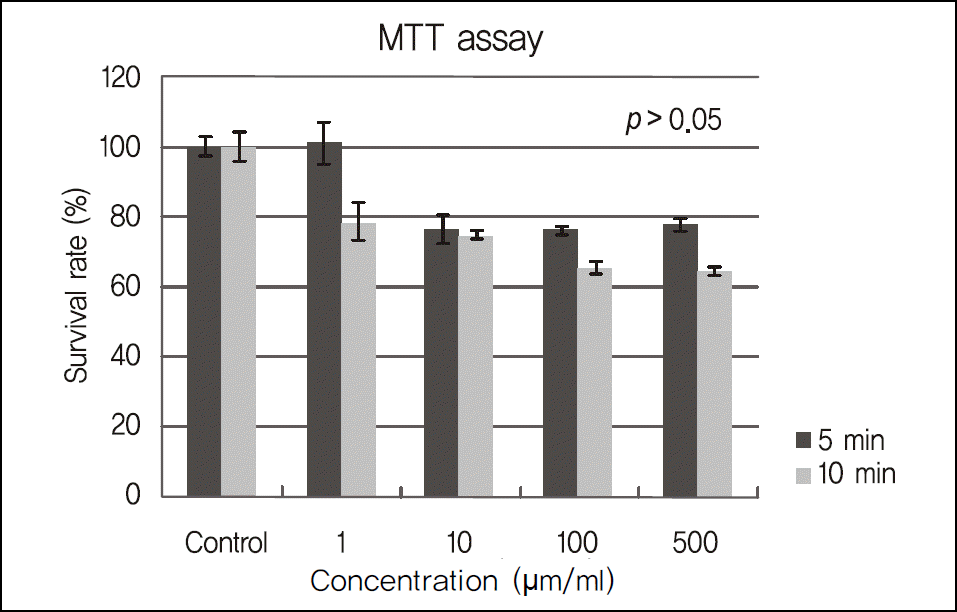
Figure 2.
LDH titers of cultured corneal epithelial cells exposed in cyclosporine. LDH titer significantly increased in all cases when exposed to 1 ug/ml (0.0001%), 10 ug/ml (0.001%), 100 ug/ml (0.01%), 500 ug/ml (0.05%) cyclosporine (p < 0.017) compared with the control group, but there were no significant differences according to exposure time within the concentrations (p > 0.05).
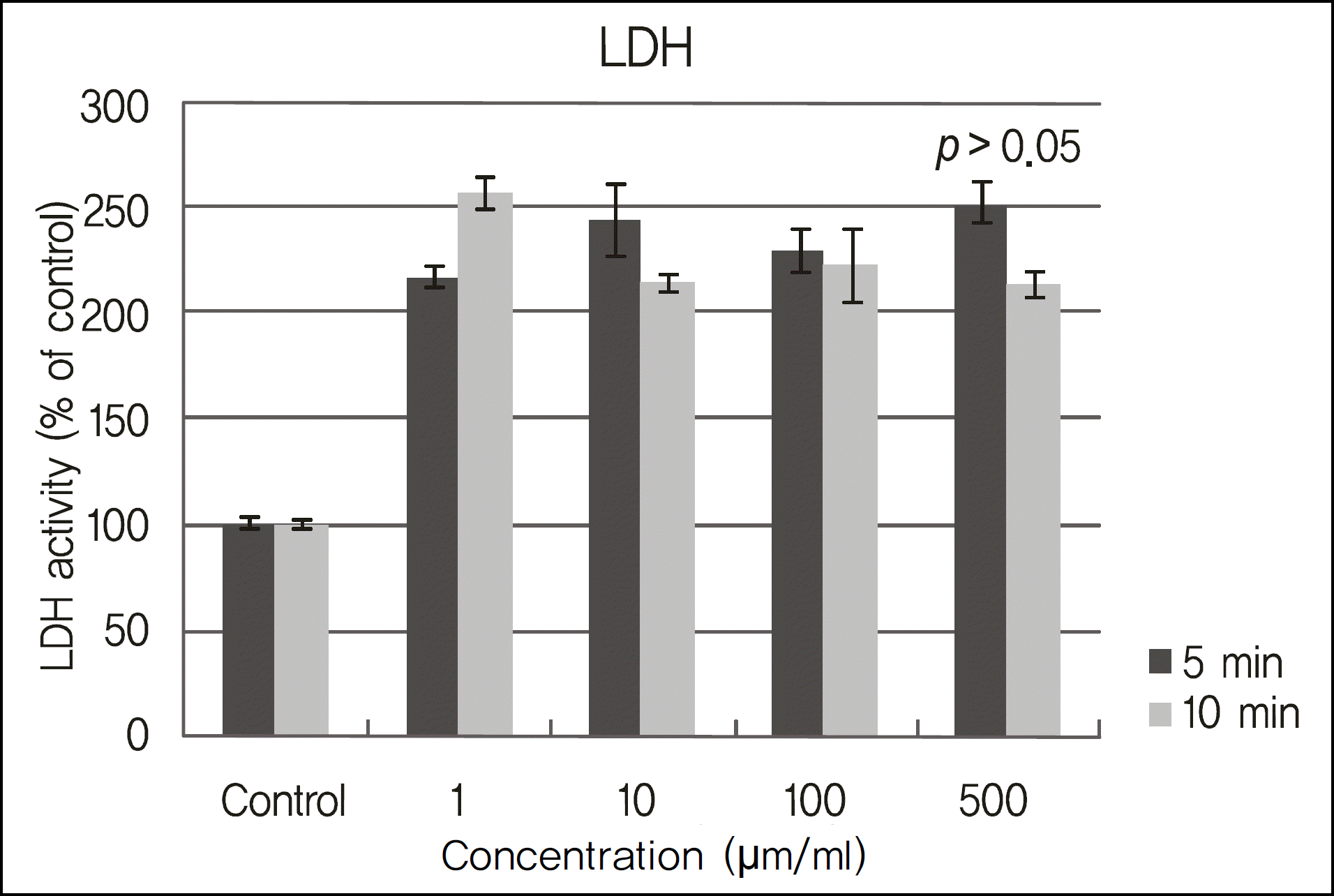
Figure 3.
PIP level of cultured corneal epithelial cells exposed to cyclosporin. In contrast to the control group, there were significant differences between the high concentration (100 ug/ml, 500 ug/ml) group and control group (p < 0.028) (*), but there were no significant differences between the low concentration (1 ug/ml, 10 ug/ml) group and the control group (p > 0.05).
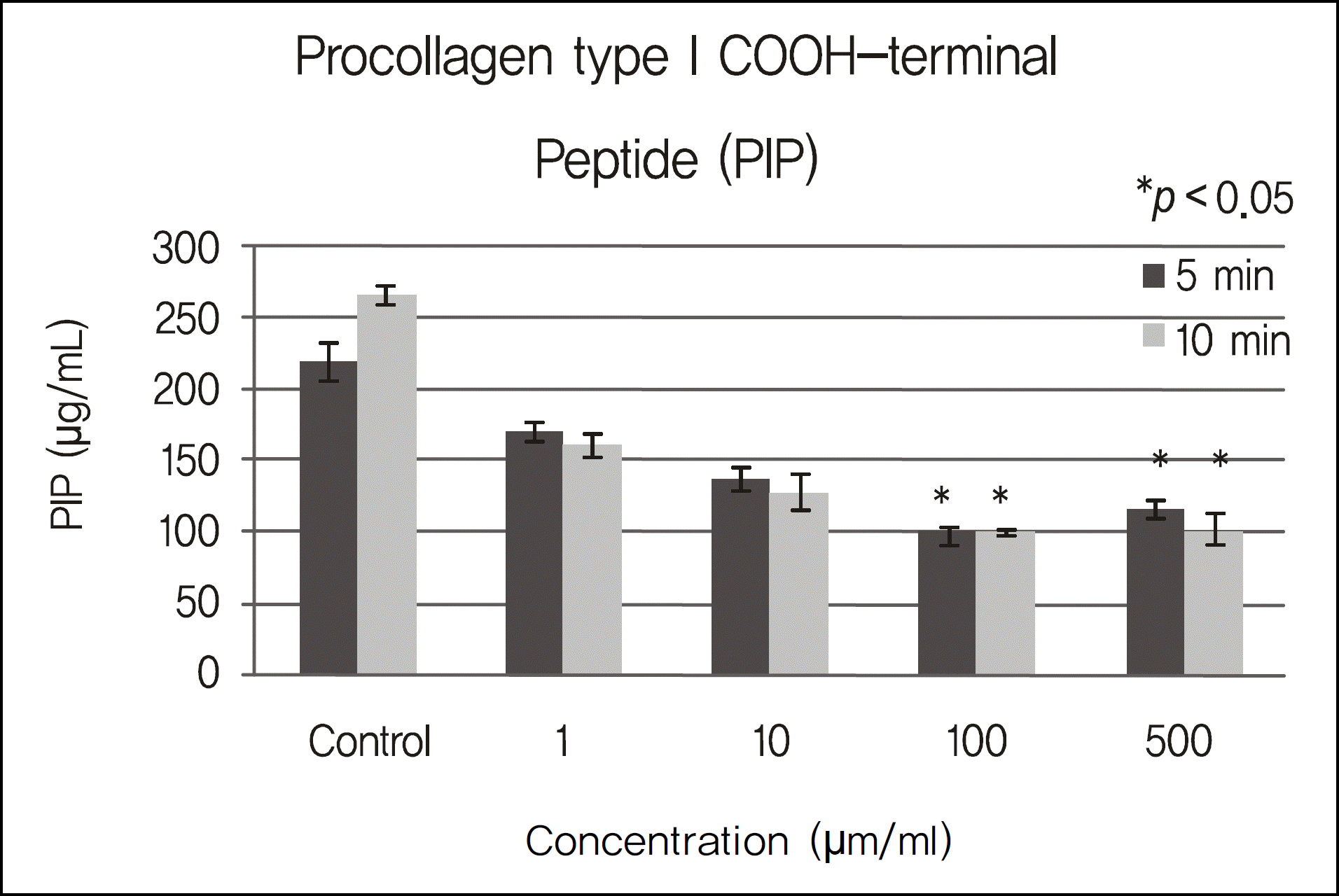
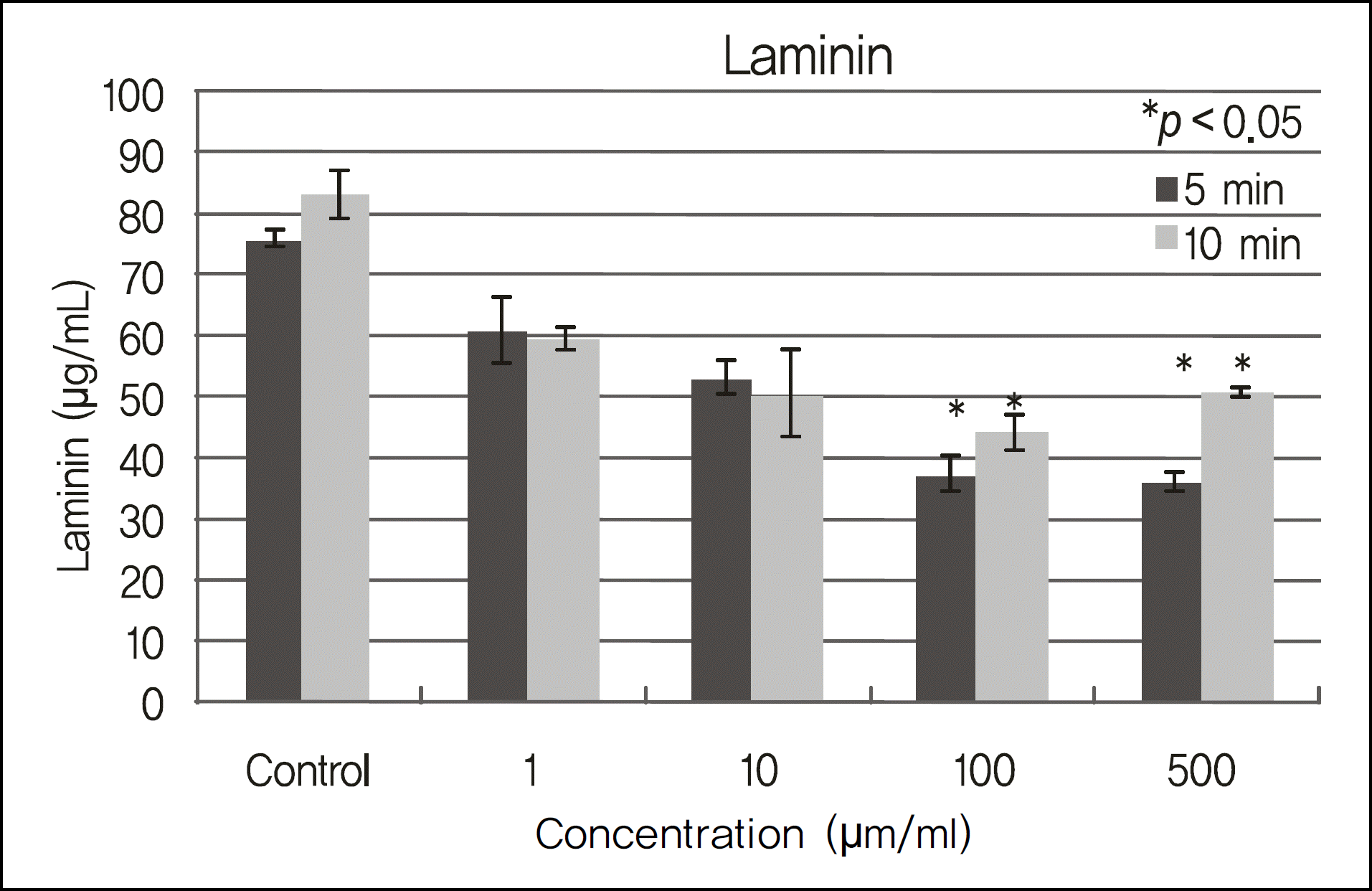
Figure 4.
Laminin levels of cultured corneal epithelial cells exposed to cyclosporin. Laminin levels were suppressed compared to the control group. There were significant differences between the high concentration (100 ug/ml, 500 ug/ml) group and control group (p < 0.028) (*), however, there were no significant differences between the low concentration (1 ug/ml, 10 ug/ml) group and the control group (p > 0.05).




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download