Abstract
Purpose
To evaluate posterior sub-Tenon injection of triamcinolone acetonide (TA) for recurrent diabetic macular edema (DME) after repeated intravitreal bevacizumab (IVB).
Methods
This is a retrospective interventional case series comprised of 35 eyes (32 patients) with recurrent clinically significant DME after two or more IVB treatments. All patients were administered TA 40 mg injections. Best-corrected visual acuity (BCVA), central macular thickness (CMT), and intraocular pressure (IOP) were reevaluated at months 1, 3, and 6.
Results
The baseline OCT showed five eyes with diffuse retinal thickening, 18 eyes with cystoid macular edema, and 12 eyes with serous retinal detachment. Mean BCVA improved at months 1 and 3, and mean CMT decreased at months 1 and 3. However, mean BCVA and CMT at six months was not different from baseline. Each DME pattern showed significant improvements in BCVA and CMT at three months. Mean IOP at months 1, 3, and 6 did not show any significant difference from baseline. No eyes showed complications including cataract progression or endophthalmitis.
Go to : 
References
1. Early Treatment Diabetic Retinopathy Study Research Group. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol. 1985; 103:1796–806.
2. Nguyen QD, Tatlipinar S, Shah SM, et al. Vascular endothelial growth factor is a critical stimulus for diabetic macular edema. Am J Ophthalmol. 2006; 142:961–9.

3. Lee CM, Olk RJ. Modified grid laser photocoagulation for diffuse diabetic macular edema. Long-term visual results. Ophthalmology. 1991; 98:1594–602.
4. Isaac DL, Abud MB, Frantz KA, et al. Comparing intravitreal triamcinolone acetonide and bevacizumab injections for the treatment of diabetic macular oedema: a randomized double-blind study. Acta Ophthalmol. 2009 Dec 16. [Epub ahead of print].

5. Paccola L, Costa RA, Folgosa MS, et al. Intravitreal triamcinolone versus bevacizumab for treatment of refractory diabetic macular oedema (IBEME study). Br J Ophthalmol. 2008; 92:76–80.

6. Shimura M, Nakazawa T, Yasuda K, et al. Comparative therapy evaluation of intravitreal bevacizumab and triamcinolone acetonide on persistent diffuse diabetic macular edema. Am J Ophthalmol. 2008; 145:854–61.

7. Oh SB, Moon JW, Kim HC. Comparison of effects of intravitreal triamcinolone and bevacizumab in the treatment of diabetic macular edema. J Korean Ophthalmol Soc. 2009; 50:1190–6.

8. Gillies MC, Sutter FK, Simpson JM, et al. Intravitreal triamcinolone for refractory diabetic macular edema: two-year results of a double-masked, placebo-controlled, randomized clinical trial. Ophthalmology. 2006; 113:1533–8.
9. Bhavsar AR, Ip MS, Glassman AR. The risk of endophthalmitis following intravitreal triamcinolone injection in the DRCRnet and SCORE clinical trials. Am J Ophthalmol. 2007; 144:454–6.

10. Ozdek S, Bahceci UA, Gurelik G, Hasanreisoglu B. Posterior subtenon and intravitreal triamcinolone acetonide for diabetic macular edema. J Diabetes Complications. 2006; 20:246–51.
11. Cellini M, Pazzaglia A, Zamparini E, et al. Intravitreal vs. subtenon triamcinolone acetonide for the treatment of diabetic cystoid macular edema. BMC Ophthalmol. 2008; 8:5–12.

12. Otani T, Kishi S, Maruyama Y. Patterns of diabetic macular edema with optical coherence tomography. Am J Ophthalmol. 1999; 127:688–93.

13. Chylack LT Jr, Wolfe JK, Singer DM, et al. The Lens Opacities Classification System III. The Longitudinal Study of Cataract Study Group. Arch Ophthalmol. 1993; 111:831–6.
14. Kang SW, Park CY, Ham DI. The correlation between fluorescein angiographic and optical coherence tomographic features in clinically significant diabetic macular edema. Am J Ophthalmol. 2004; 137:313–22.

15. Kim BY, Smith SD, Kaiser PK. Optical coherence tomographic patterns of diabetic macular edema. Am J Ophthalmol. 2006; 142:405–12.

16. Kim NR, Kim YJ, Chin HS, Moon YS. Optical coherence tomographic patterns in diabetic macular oedema: prediction of visual outcome after focal laser photocoagulation. Br J Ophthalmol. 2009; 93:901–5.

17. Funatsu H, Yamashita H, Sakata K, et al. Vitreous levels of vascular endothelial growth factor and intercellular adhesion molecule 1 are related to diabetic macular edema. Ophthalmology. 2005; 112:806–16.

18. Arevalo JF, Fromow-Guerra J, Quiroz-Mercado H, et al. Primary intravitreal bevacizumab (Avastin) for diabetic macular edema: results from the Pan-American Collaborative Retina Study Group at 6-month follow-up. Ophthalmology. 2007; 114:743–50.
19. Arevalo JF, Sanchez JG, Wu L, et al. Primary intravitreal bevacizumab for diffuse diabetic macular edema: the Pan-American Collaborative Retina Study Group at 24 months. Ophthalmology. 2009; 116:1488–97.
20. Kook D, Wolf A, Kreutzer T, et al. Long-term effect of intravitreal bevacizumab (avastin) in patients with chronic diffuse diabetic macular edema. Retina. 2008; 28:1053–60.

21. Roh MI, Byeon SH, Kwon OW. Repeated intravitreal injection of bevacizumab for clinically significant diabetic macular edema. Retina. 2008; 28:1314–8.

22. Bakri SJ, Snyder MR, Reid JM, et al. Pharmacokinetics of intravitreal bevacizumab (Avastin). Ophthalmology. 2007; 114:855–9.

24. Artunay O, Yuzbasioglu E, Rasier R, et al. Incidence and management of acute endophthalmitis after intravitreal bevacizumab (Avastin) injection. Eye (Lond). 2009; 23:2187–93.

25. Biester S, Ziemssen F, Ulrich Bartz-Schmidt K, Gelisken F. Is intravitreal bevacizumab treatment effective in diffuse diabetic macular edema? Graefes Arch Clin Exp Ophthalmol. 2009; 247:1575–7.

26. Shima C, Sakaguchi H, Gomi F, et al. Complications in patients after intravitreal injection of bevacizumab. Acta Ophthalmol. 2008; 86:372–6.

27. Huang ZL, Lin KH, Lee YC, et al. Acute vision loss after intravitreal injection of bevacizumab (Avastin) associated with ocular ischemic syndrome. Ophthalmologica. 2009; 224:86–9.

28. Chen E, Hsu J, Park CH. Acute visual acuity loss following intravitreal bevacizumab for diabetic macular edema. Ophthalmic Surg Lasers Imaging. 2009; 40:68–70.

29. Song HJ, Sohn HJ, Lee DY, Nam DH. Tractional retinal detachment after intravitreal bevacizumab (Avastin®) injection in proliferative diabetic retinopathy. J Korean Ophthalmol Soc. 2009; 50:1751–4.

30. Funatsu H, Yamashita H, Ikeda T, et al. Vitreous levels of inter-leukin-6 and vascular endothelial growth factor are related to diabetic macular edema. Ophthalmology. 2003; 110:1690–6.

31. Aiello LP. The potential role of PKC beta in diabetic retinopathy and macular edema. Surv Ophthalmol. 2002; 47(Suppl 2):S263–9.
32. Kent D, Vinores SA, Campochiaro PA. Macular oedema: the role of soluble mediators. Br J Ophthalmol. 2000; 84:542–5.

33. Sander B, Larsen M, Moldow B, Lund-Andersen H. Diabetic macular edema: passive and active transport of fluorescein through the blood-retina barrier. Invest Ophthalmol Vis Sci. 2001; 42:433–8.
34. Bandi N, Kompella UB. Budesonide reduces vascular endothelial growth factor secretion and expression in airway (Calu-1) and alveolar (A549) epithelial cells. Eur J Pharmacol. 2001; 425:109–16.

35. Fischer S, Renz D, Schaper W, Karliczek GF. In vitro effects of dexamethasone on hypoxia-induced hyperpermeability and expression of vascular endothelial growth factor. Eur J Pharmacol. 2001; 411:231–43.

36. Tong JP, Lam DS, Chan WM, et al. Effects of triamcinolone on the expression of VEGF and PEDF in human retinal pigment epithelial and human umbilical vein endothelial cells. Mol Vis. 2006; 12:1490–5.
37. Floman N, Zor U. Mechanism of steroid action in ocular inflammation: Inhibition of prostaglandin production. Invest Ophthalmol Vis Sci. 1977; 16:69–73.
38. Kang BS, Chung EY, Yun YP, et al. Inhibitory effects of anti-inflammatory drugs on interleukin-6 bioactivity. Biol Pharm Bull. 2001; 24:701–3.

39. Umland SP, Nahrebne DK, Razac S, et al. The inhibitory effects of topically active glucocorticoids on IL-4, IL-5, and interferon-gamma production by cultured primary CD4+ T cells. J Allergy Clin Immunol. 1997; 100:511–9.
40. Wilson CA, Berkowitz BA, Sato Y, et al. Treatment with intravitreal steroid reduces blood-retinal barrier breakdown due to retinal photocoagulation. Arch Ophthalmol. 1992; 110:1155–9.

41. Bonini-Filho MA, Jorge R, Barbosa JC, et al. Intravitreal injection versus sub-Tenon's infusion of triamcinolone acetonide for refractory diabetic macular edema: a randomized clinical trial. Invest Ophthalmol Vis Sci. 2005; 46:3845–9.

42. Chung EJ, Roh MI, Kwon OW, Koh HJ. Effects of macular ischemia on the outcome of intravitreal bevacizumab therapy for diabetic macular edema. Retina. 2008; 28:957–63.

43. Kim HD, Choi KS, Lee SJ. Combined therapy of intravitreal bevacizumab and posterior subtenon triamcinolone acetonide injection in diabetic macular edema. J Korean Ophthalmol Soc. 2009; 50:1652–6.

44. Inoue M, Takeda K, Morita K, et al. Vitreous concentrations of triamcinolone acetonide in human eyes after intravitreal or subtenon injection. Am J Ophthalmol. 2004; 138:1046–8.

45. Audren F, Tod M, Massin P, et al. Pharmacokinetic-pharmacody-namic modeling of the effect of triamcinolone acetonide on central macular thickness in patients with diabetic macular edema. Invest Ophthalmol Vis Sci. 2004; 45:3435–41.

46. Kim YG, Yu SY, Kwak HW. The effect of intravitreal triamcinolone acetonide injection according to the diabetic macular edema. J Korean Ophthalmol Soc. 2006; 46:84–9.
47. Yoon SC, Lee DY, Nam DH. The effect of intravitreal triamcinolone injection according to the OCT patterns of diabetic macular edema. J Korean Ophthalmol Soc. 2008; 49:1611–8.

48. Jonas JB, Kreissig I, Degenring R. Intraocular pressure after intravitreal injection of triamcinolone acetonide. Br J Ophthalmol. 2003; 87:24–7.

49. Smithen LM, Ober MD, Maranan L, Spaide RF. Intravitreal triamcinolone acetonide and intraocular pressure. Am J Ophthalmol. 2004; 138:740–3.

50. Beck RW, Edwards AR, Aiello LP, et al. Three-year follow-up of a randomized trial comparing focal/grid photocoagulation and intravitreal triamcinolone for diabetic macular edema. Arch Ophthalmol. 2009; 127:245–51.

51. Moshfeghi DM, Kaiser PK, Scott IU, et al. Acute endophthalmitis following intravitreal triamcinolone acetonide injection. Am J Ophthalmol. 2003; 136:791–6.

52. Sohn HJ, Nam DH. Infectious endophthalmitis after intravitreal injection of triamcinolone acetonide. J Korean Ophthalmol Soc. 2006; 47:1865–70.
Go to : 
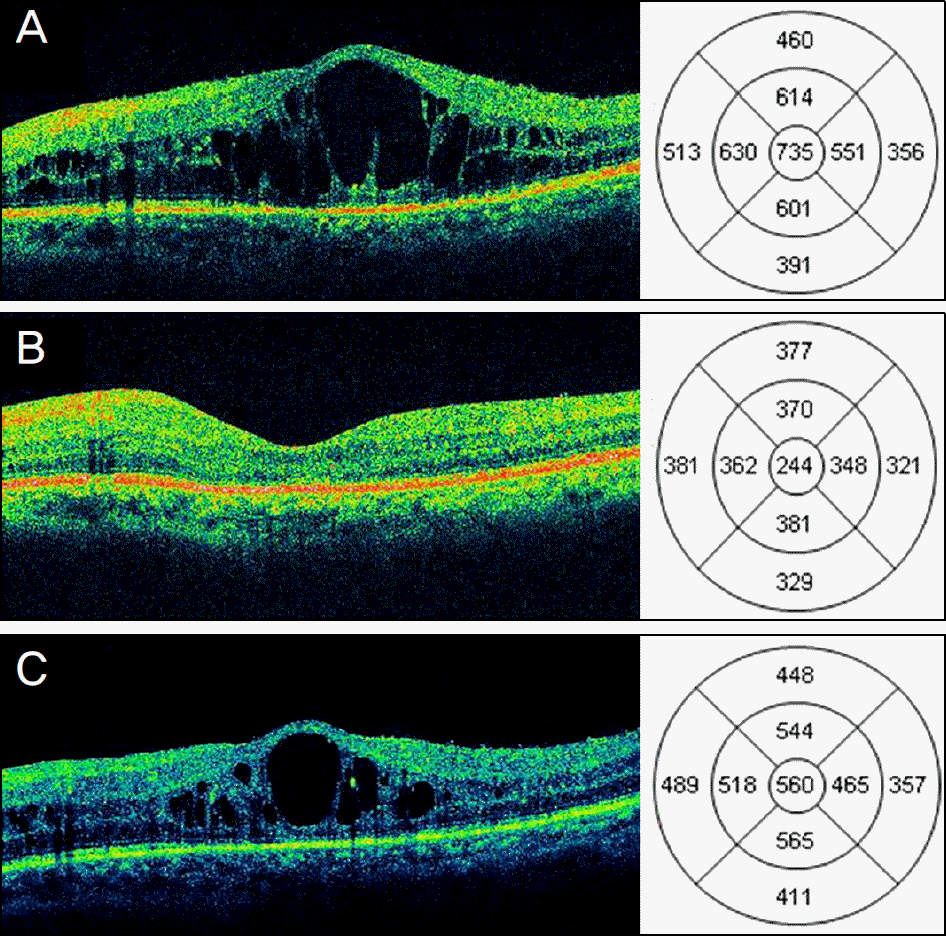 | Figure 1.Changes in central macular thickness (CMT) and best-corrected visual acuity (BCVA) of eyes with diabetic macular edema (DME) after posterior sub-Tenon triamcinolone acetonide (TA) 40 mg (1 ml) injection. (A, B, C) Horizontal optical coherence tomography (OCT) scan of a 67-year-old patient with recurrent DME despite 3 repeated intravitreal bevacizumab (IVB) injections. (A) At baseline, OCT showed cystoid macular edema with intraretinal cystoid spaces with 735 μ m of CMT and BCVA was 20/200. (B) Three months after TA injection, OCT showed complete resolution of intraretinal cystoid spaces with 244 μ m of CMT and BCVA improved to 20/50. (C) Six months after TA injection, CMT increased to 560 μ m and BCVA decreased to 20/100. |
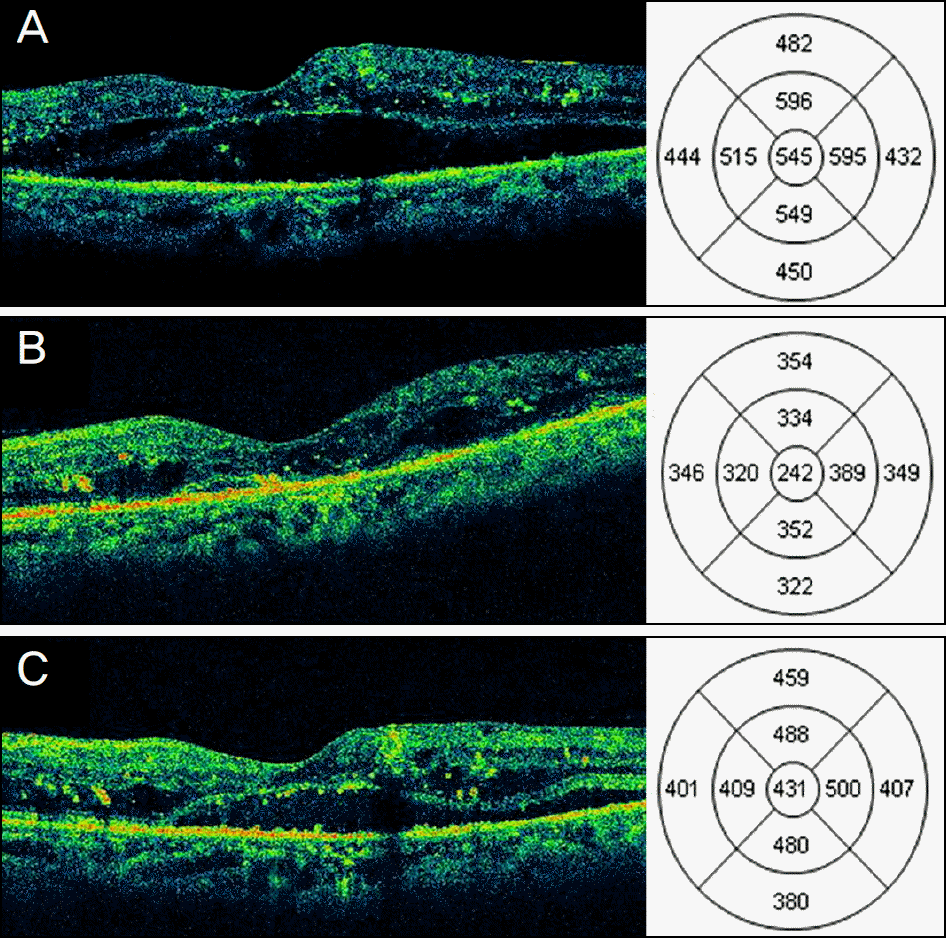 | Figure 2.Changes in central macular thickness (CMT) and best-corrected visual acuity (BCVA) of eyes with diabetic macular edema (DME) after posterior sub-Tenon triamcinolone acetonide (TA) 40 mg (1 ml) injection. (A, B C) Horizontal optical coherence tomography (OCT) scan of a 64-year-old patient with recurrent DME despite 3 repeated intravitreal bevacizumab (IVB) injections. (A) At baseline, OCT showed serous retinal detachment with low subretinal reflectivity with 545 μ m of CMT and BCVA was 20/125. (B) Three months after TA injection, OCT showed nearly complete resolution of subretinal fluid with 242 μ m of CMT and BCVA improved to 20/80. (C) Six months after TA injection, CMT increased to 431 μ m and BCVA decreased to 20/125. |
Table 1.
Demographics and clinical data
DME = diabetic macular edema; DR = diabetic retinopathy; IVB = intravitreal bevacizumab; NPDR = nonproliferative diabetic retinopathy; OCT = optical coherence tomography; PDR = proliferative diabetic retinopathy; PRP = panretinal photocoagulation; SD = standard deviation; TA = triamcinolone acetonide.
Table 2.
Changes in best-corrected visual acuity and central macular thickness
| | n = 35 | p-value |
|---|---|---|
| BCVA (log MAR) | | |
| Baseline | 0.77 ± 0.36 | |
| After posterior sub-Tenon TA injection | | |
| 1 month | 0.68 ± 0.33 | 0.017* |
| 3 months | 0.66 ± 0.30 | 0.001* |
| 6 months | 0.75 ± 0.38 | 1.0* |
| Improvement of BCVA ≥ 0.3 log MAR | 3 (8.6) | |
| Unchanged BCVA | 32 (91.4) | |
| Deterioration of BCVA ≥ 0.3 log MAR | 0 (0) | |
| CMT (μ m) | | |
| Baseline | 512 ± 134 | |
| After posterior sub-Tenon TA injection | | |
| 1 month | 356 ± 100 | <0.001* |
| 3 months | 323 ± 109 | <0.001* |
| 6 months | 456 ± 101 | 0.171* |
Table 3.
Changes in best-corrected visual acuity and central macular thickness according to the patterns of diabetic macular edema by optical coherence tomography
| | Baseline | 3 months | p-value |
|---|---|---|---|
| BCVA (log MAR) | | | |
| Diffuse retinal thickening (n = 5) | 0.78 ± 0.29 | 0.64 ± 0.26 | 0.031* |
| Cystoid macular edema (n = 18) | 0.76 ± 0.31 | 0.64 ± 0.25 | 0.008* |
| Serous retinal detachment (n = 12) | 0.77 ± 0.43 | 0.68 ± 0.40 | 0.036* |
| p-value | 0.532† | 0.982† | |
| CMT (μ m) | | | |
| Diffuse retinal thickening (n = 5) | 454 ± 144 | 319 ± 50 | 0.033* |
| Cystoid macular edema (n = 18) | 540 ± 127 | 322 ± 81 | <0.001* |
| Serous retinal detachment (n = 12) | 493 ± 140 | 327 ± 111 | 0.002* |
| p-value | 0.186† | 0.851† | |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download