Abstract
Purpose
To report retinal hemorrhage and retinal edema induced by the remaining triamcinolone acetonide (TA) used in vitrectomy for dyeing of vitreous and epiretinal membranes.
Case summary
The authors performed 23 gauze vitrectomies for macular epiretinal membranes. TA was used during the operation to enhance visualization of the vitreous and epiretinal membrane. At the end of the surgery, an amount of TA was left on the retinal surface. One day after the operation, the patient was maintained a semi-sitting position because of the floating TA particles in the vitreous cavity. The following day, the TA settled onto the inferior retina and retinal edema and hemorrhage occurred unexpectedly in the inferior retina. Vitrectomy was performed again and the fluid of the vitreous cavity was replaced with a fresh balanced salt solution and the settled TA was removed from retinal surface. Finally, argon laser photocoagulation was performed at the area where the retinal edema and hemorrhage had occurred. The retinal edema and hemorrhage disappeared after a few days, normal visual acuity was recovered in seven days, and a normal condition with no further ocular complications was maintained at six months.
Conclusions
When a TA vehicle, such as benzyl alcohol, and TA particles contained in the vehicle are left in the vitreous cavity and contact the retinal surface of a vitrectomized eye, retinal toxicity can be induced due to retinal edema and hemorrhage. The safest use of TA in vitrectomy would involve at least two rounds of sedimentation or filtration through a micofilter needle.
Go to : 
References
1. Enaida H, Hata Y, Ueno A, et al. Possible benefits of triamcinolone- assisted pars plana vitrectomy for retinal diseases. Retina. 2003; 23:764–70.
2. Peyman GA, Cheema R, Conway MD, et al. Triamcinolone acetonide as an aid to visualization of the vitreous and the posterior hyaloid during pars plana vitrectomy. Retina. 2000; 20:554–5.

3. Choi ES, Choi YR, Yoon HS. Comparison of outcomes of ILM peeling using triamcinolone and indocyanine green during idiopathic macular hole surgery. J Korean Ophthalmol Soc. 2006; 47:1589–96.
4. Park UC, Park KH, Yu YS, Chung H. Comparison of indocyanine green and triamcinolone acetonide for internal limiting membrane peeling in macular hole. J Korean Ophthalmol Soc. 2005; 46:1995–2003.
5. McCuen BW 2nd, Bessler M, Tano Y, et al. The lack of toxicity of intravitreally adminstered triamcinolone acetonide. Am J Ophthalmol. 1981; 91:785–8.
6. Sakamoto T, Miyazaki M, Hisatomi T, et al. Triamcinolone- assisted pars plana vitrectomy improves the surgical procedures and decreases the post-operative blood–ocular barrier breakdown. Graefes Arch Clin Exp Ophthalmol. 2002; 240:423–9.
7. Yu SI, Kim HW. The benefits of triamcinolone-assisted pars plana vitrectomy for proliferative diabetic retinopathy. J Korean Ophthalmol Soc. 2006; 47:1427–34.
8. Jonas JB, Hayler JK, Sofker A, Panda-Jonas S. Intravitreal injection of crystalline cortisone as adjunctive treatment of proliferative diabetic retinopathy. Am J Ophthalmol. 2001; 131:468–71.

9. Woods AC. Clinical and experimental observations on the use of ACTH and cortisone in ocular inflammatory disease. Am J Ophthalmol. 1950; 33:1325–49.
10. Schindler RH, Chandler D, Thresher R, Machemer R. The clearance of intravitreal triamcinolone acetonide. Am J Ophthalmol. 1982; 93:415–7.

11. Jonas JB, Söfker A, Degenring R. Intravitreal Triamcinolone acetonide as an additional tool in pars plana vitrectomy for proliferative diabetic retinopathy. Eur J Ophthalmol. 2003; 13:468–73.

12. Jaissle GB, Szurman P, Bartz-Schmidt KU. Ocular side effects and complications of intravitreal triamcinolone acetonide injection. Ophthalmologe. 2004; 101:121–8.
13. Beer PM, Bakri SJ, Singh RJ, et al. Intraocular concentraion and pharmacokitetics of triamcinolone acetonide after a single intravitreal injection. Ophthalmology. 2003; 110:681–6.
14. Nelson ML, Tennant MT, Sivalingam A, et al. Infectious and pre-sumed noninfectious endophthalmitis after intravitreal triamcino lone acetonide injection. Retina. 2003; 23:686–91.
15. Bakri SJ, Beer PM. The effect of intravitreal triamcinolone acetonide on intraocular pressure. Ophthalmic Surg Lasers Imaging. 2003; 34:386–90.

16. Gillies MC, Simpson JM, Billson FA, et al. Safety of an intravitreal injection of triamcinolone: results from a randomized clinical trial. Arch Ophthalmol. 2004; 122:336–40.
17. Shin JH, Kim DK, Yu SY, Kwak HW. A case of central retinal artery occlusion after intravitreal triamcinolone acetonide injection for diabetic macular edema in non-proliferative diabetic retinopathy. J Korean Ophthalmol Soc. 2006; 47:667–71.
18. Yeung CK, Chan KP, Chiang SW, et al. The toxic and stress responses of cultured human retinal pigment epithelium (ARPE19) and human glial cells (SVG) in the presence of triamcinolone. Invest Ophthalmol Vis Sci. 2003; 44:5293–300.

19. Kang SM, Park YS, Lee BR. The clear oil-drop residue in the vitreous cavity after intravitreal triamcinolone injection. J Korean Ophthalmol Soc. 2007; 48:665–70.
20. Morrison VL, Koh HJ, Cheng L, et al. Intravitreal toxicity of the kenalog vehicle (benzyl alcohol) in rabbits. Retina. 2006; 26:339–44.

21. Macky TA, Helmy D, El Shazly N. Retinal toxicity of triamcinolone' s vehicle (benzyl alcohol): an electrophysiologic and electron microscopic study. Graefes Arch Clin Exp Ophthalmol. 2007; 245:817–24.
Go to : 
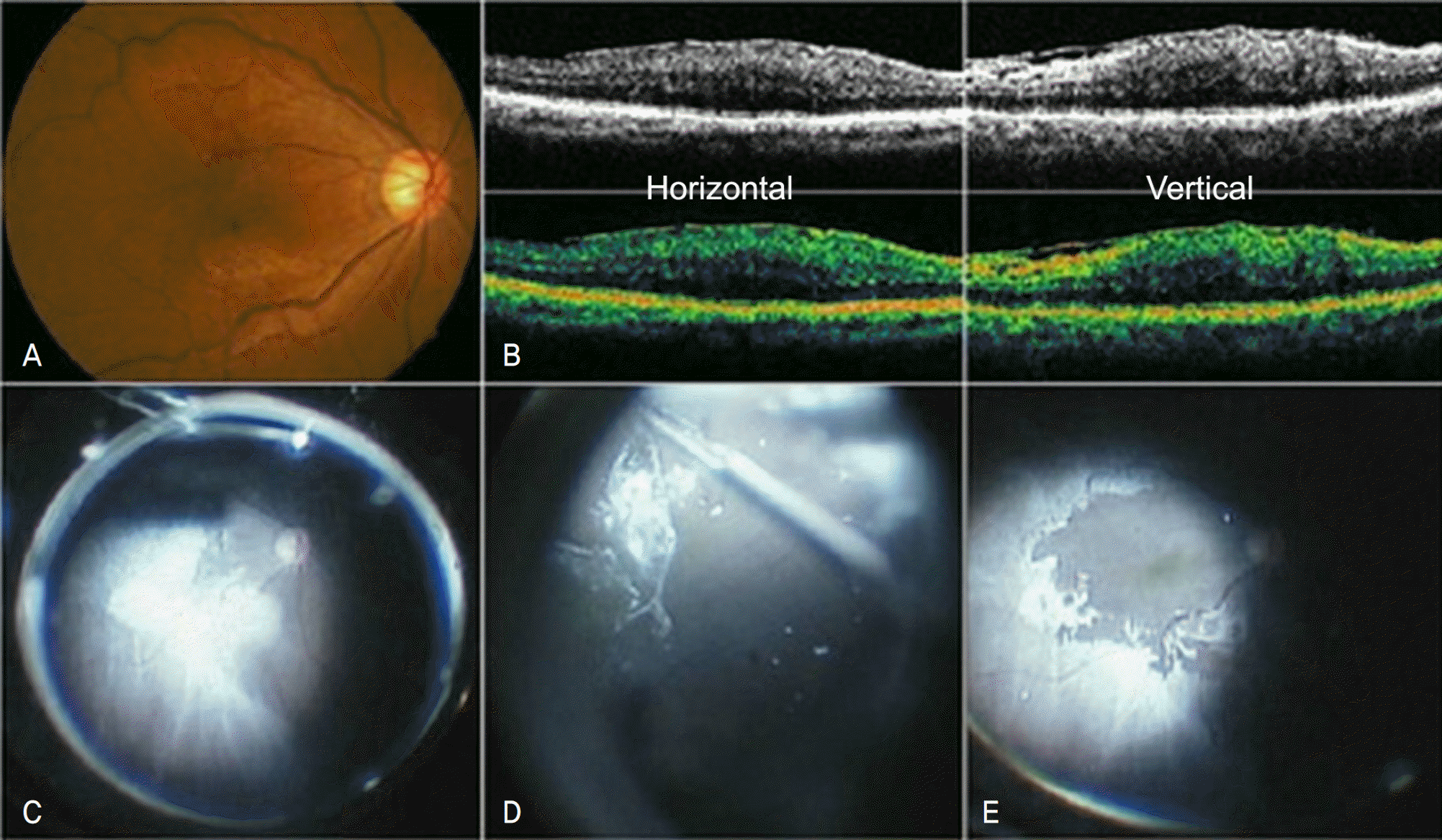 | Figure 1.The pre-operative fundus photograph (A) and OCT images (B). Intra-operative findings shows epiretinal membrane peeling using triamcinolone acetonide for enhanced visualization of the epiretinal membrane (C, D). The features of fundus at the end of vitrectomy after epiretinal membrane removal (E). |
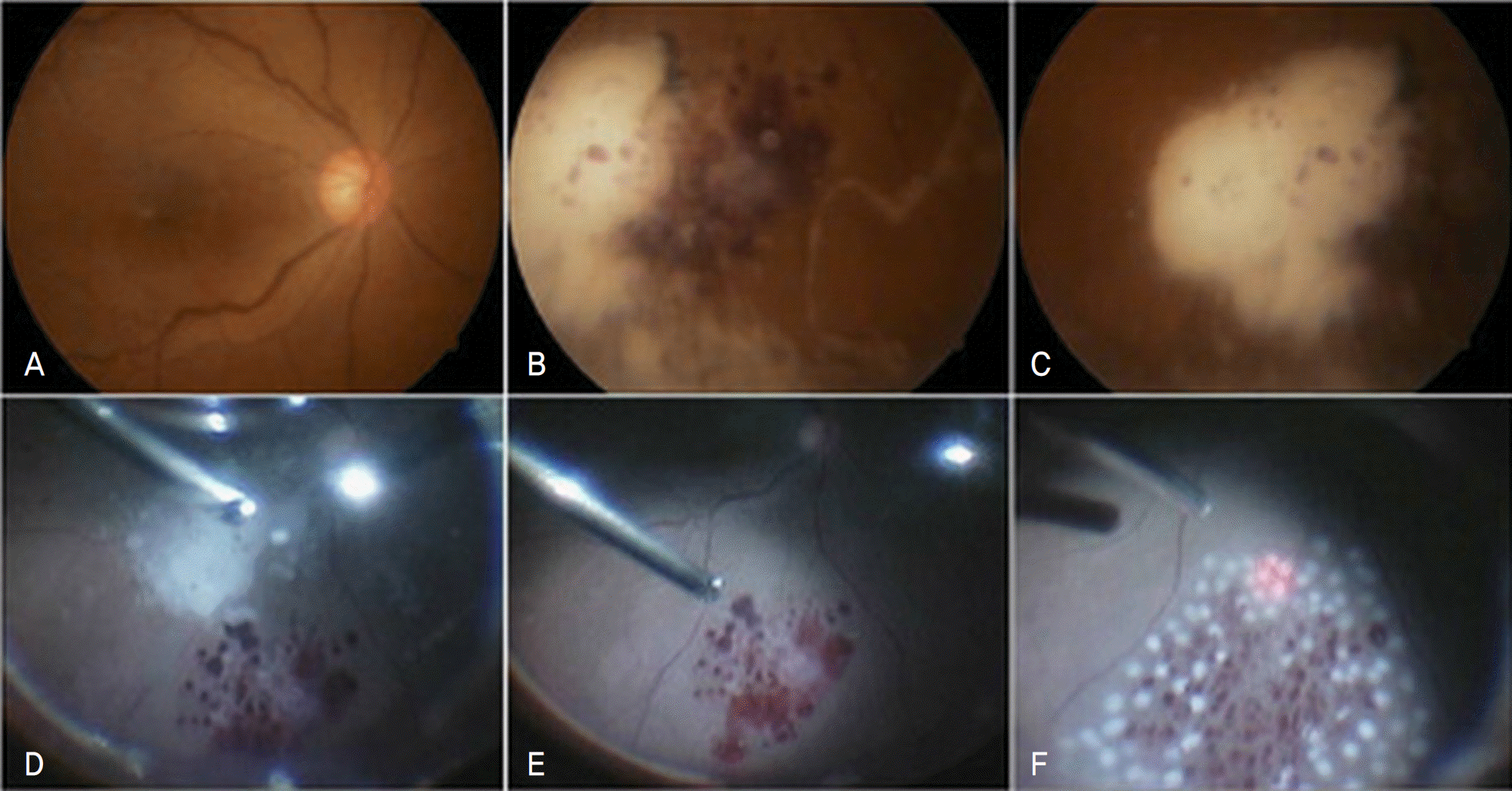 | Figure 2.The fundus photographs taken two day after vitrectomy show clear vitreous cavity and posterior pole (A). But, triamcinolone acetonide settled down in the inferior retina and focal retinal edema and hemorrhage were noted under the flake of triamcinolone acetonide (B, C). Intra-operative fundus findings at the time of the second vitrectomy: Layered TA particle was removed with an extrusion needle (D) note retinal edema and hemorrhage in inferior retinal surface significantly (E), argon laser photocoagulation was done at the hemorrhagic and edematous retinal region (F). |
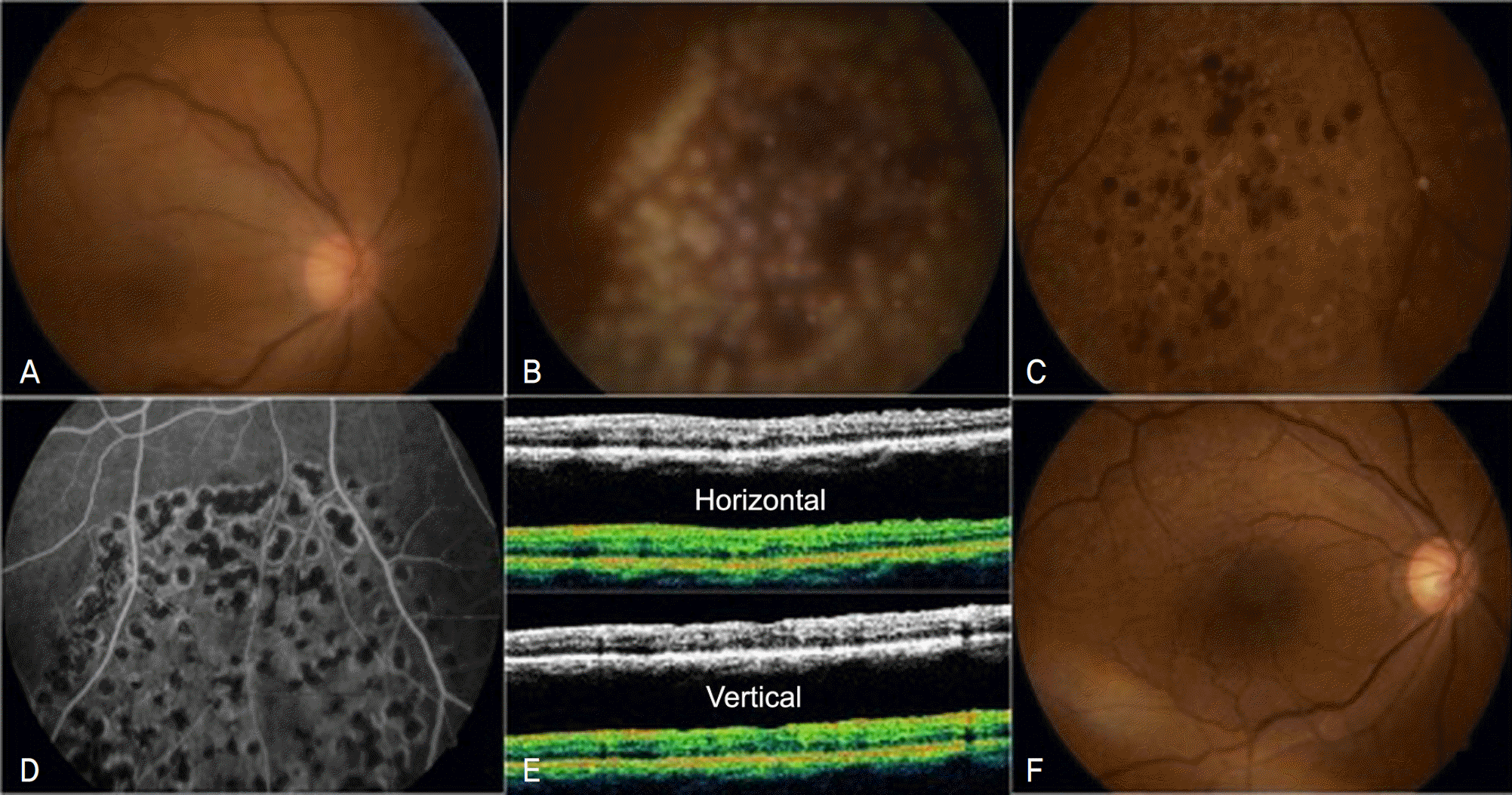 | Figure 3.The fundus photographs taken one day after the second vitrectomy (A, B). The fundus photograph at 7day after the second operation (C). The FAG finding at 2 months after the second operation (D). The OCT images at 2 months after vitrectomy show reduction of macular thickness and absence of epiretinal membrane (E). The fundus photographs of 6 months after the second operation: the retina remains stable without any other complication (F). |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download