Abstract
Purpose:
To investigate the effect of intravitreal bevacizumab injection in preventing panretinal photocoagulation (PRP)-induced macular edema and visual dysfunction in proliferative diabetic retinopathy.
Methods:
We conducted a prospective study of 40 consecutive eyes (20 patients) with proliferative diabetic retinopathy whose visual acuity (logMAR) was 0.2 or less, foveal thickness (µ m) was 280 or less, and retinopathy was bilaterally symmetrical. In Group 1 (20 eyes), PRP was performed with intravitreal bevacizumab injection 1 week before the initiation of PRP, but in Group 2 (20 eyes) PRP alone was performed. In all eyes, PRP was completed using the same technique. Best corrected visual acuity (BCVA) and foveal and parafoveal thicknesses were measured before treatment and 1 and 3 months after PRP.
Results:
Before treatment, the mean BCVA and foveal and parafoveal thicknesses were not statistically significantly different between the two groups (p >0.05). At 1 and 3 months after PRP, the mean BCVA improved and the foveal and parafoveal thicknesses were significantly smaller in Group 1 than in Group 2 (p <0.05).
References
1. Early Treatment Diabetic Retinopathy Study Research Group. Early photocoagulation for diabetic retinopathy. Early Treatment Diabetic Retinopathy Study Report No 9. Ophthalmology. 1991; 98:766–85.
2. The Diabetic Retinopathy Study Research Group. Photocoagu-lation treatment of proliferative diabetic retinopathy. Clinical application of Diabetic Retinopathy Study (DRS) findings, DRS Report Number 8. Ophthalmology. 1981; 88:583–600.
3. McDonald HR, Schatz H. Macular edema following panretinal photocoagulation. Retina. 1985; 5:5–10.

4. McDonald HR, Schatz H. Visual loss following panretinal photocoagulation for proliferative diabetic retinopathy. Ophthalmology. 1985; 92:388–93.

5. Higgins KE, Meyers SM, Jaffe MJ, et al. Temporary loss of foveal contrast sensitivity associated with panretinal photocoagulation. Arch Ophthalmol. 1986; 104:997–1003.

6. Meyers SM. Macular edema after scatter laser photocoagulation for proliferative diabetic retinopathy. Am J Ophthalmol. 1980; 90:210–6.

7. Kleiner RC, Elman MJ, Murphy RP, Ferris FL. Transient severe visual loss after panretinal photocoagulation. Am J Ophthalmol. 1988; 106:298–306.
8. Shimura M, Yasuda K, Nakazawa T, Tamai M. Visual dys-function after panretinal photocoagulation in patients with severe diabetic retinopathy and good vision. Am J Ophthalmol. 2005; 140:8–15.

9. Ferris FL, Podgor MJ, Davis MD. Macular edema in diabetic retinopathy study patients. Diabetic Retinopathy Study Group report number 12. Ophthalmology. 1987; 94:754–60.
10. Nonaka A, Kiryu J, Tsujikawa A, et al. Inflammatory response after scatter laser photocoagulation in nonphotocoagulated retina. Invest Ophthalmol Vis Sci. 2002; 43:1204–9.
11. Funatsu H, Yamashita H, Ikeda T, et al. Relation of diabetic macular edema to cytokines and posterior vitreous detachment. Am J Ophthalmol. 2003; 135:321–7.

12. Brooks HL, Caballero S, Newell CK, et al. Vitreous levels of vascular endothelial growth factor and stromal-derived factor1 in patients with diabetic retinopathy and cystoid macular edema before and after intraocular injection of triamcinolone. Arch Ophthalmol. 2004; 122:1801–7.
13. Jonas JB, Kreissig I, Sofker A, Degenring RF. Intravitreal injection of triamcinolone for diffuse diabetic macular edema. Arch Ophthalmol. 2003; 121:57–61.

14. Bakri SJ, Kaiser PK. Posterior subtenon triamcinolone acetonide for refractory diabetic macular edema. Am J Ophthalmol. 2005; 139:290–4.

15. Massin P, Audren F, Haouchine B, et al. Intravitreal triamcinolone acetonide for diabetic diffuse macular edema. Ophthalmology. 2004; 111:218–24.
16. Martidis A, Duker JS, Greenberg PB, et al. Intravitreal triamcinolone for refractory diabetic macular edema. Ophthalmology. 2002; 109:920–7.

17. Chieh JJ, Roth DB, Liu M, et al. Intravitreal triamcinolone acetonide for diabetic macular edema. Retina. 2005; 25:828–34.

18. Toda J, Fukushima H, Kato S. Injection of triamcinolone acetonide into the posterior subtenon capsule for treatment of diabetic macular edema. Retina. 2007; 27:764–9.

19. Nonaka A, Kiryu J, Tsujikawa A, et al. Inflammatory response after scatter laser photocoagulation in nonphotocoagulated retina. Invest Ophthalmol Vis Sci. 2002; 43:1204–9.
20. Musashi K, Kiryu J, Miyamoto K, et al. Thrombin inhibitor reduces leukocyte-endothelial cell interactions and vascular leakage after scatter laser photocoagulation. Invest Ophthalmol Vis Sci. 2005; 46:2561–6.

21. Er H, Doganay S, Turkoz Y, et al. The levels of cytokine and nitric oxide in rabbit vitreous humor after retinal laser photoco-agulation. Ophthalmic Surg Lasers. 2000; 31:479–83.
22. Brooks HL, Caballero S, Newell CK, et al. Vitreous levels of vascular endothelial growth factor and stromal-derived factor 1 in patients with diabetic retinopathy and cystoid macular edema before and after intraocular injection of triamcinolone. Arch Ophthalmol. 2004; 122:1801–7.
23. Shimura M, Yasuda K, Shiono T. Posterior subtenon's capsule injection of triamcinolone acetonide prevents panretinal photo-coagulation-induced visual dysfunction in patients with severe diabetic retinopathy and good vision. Ophthalmology. 2006; 113:381–7.

24. Kang SB, Park YH. The combined effect of subtenon triamcinolone injection and panretinal photocoagulation on diabetic retinopathy. J Korean Ophthalmol Soc. 2008; 49:71–80.

25. Helm CJ, Holland GN. The effects of posterior subtenon injection of triamcinolone acetonide in patients with intermediate uveitis. Am J Ophthalmol. 1995; 120:55–64.

26. Ozkiris A, Erkilic K. Complications of intravitreal injection of triamcinolone acetonide. Can J Ophthalmol. 2005; 40:63–8.
27. Jonas JB, Degenring RF, Kreissig I, et al. Intraocular pressure elevation after intravitreal triamcinolone acetonide injection. Ophthalmology. 2005; 112:593–8.

28. Mackensen F, Heinz C, Becker MD, Heiligenhaus A. Intravitreal bevacizumab (avastin) as a treatment for refractory macular edema in patients with uveitis: a pilot study. Retina. 2008; 28:41–5.
29. Mason JO 3rd, Albert MA Jr, Vail R. Intravitreal bevacizumab (Avastin) for refractory pseudophakic cystoid macular edema. Retina. 2006; 26:356–7.

30. Barone A, Prascina F, Russo V, et al. Successful treatment of pseudophakic cystoid macular edema with intravitreal bevacizumab. J Cataract Refract Surg. 2008; 34:1210–2.

31. Spitzer MS, Ziemssen F, Yoeruek E, et al. Efficacy of intravitreal bevacizumab in treating postoperative pseudophakic cystoid macular edema. J Cataract Refract Surg. 2008; 34:70–5.

32. Spandau UH, Ihloff AK, Jonas JB. Intravitreal bevacizumab treatment of macular oedema due to central retinal vein occlusion. Acta Ophthalmol Scand. 2006; 84:555–6.

33. Matsumoto Y, Freund KB, Peiretti E, et al. Rebound macular edema following bevacizumab (Avastin) therapy for retinal venous occlusive disease. Retina. 2007; 27:426–31.

34. Costa RA, Jorge R, Calucci D, et al. Intravitreal bevacizumab (avastin) for central and hemicentral retinal vein occlusions: IBeVO study. Retina. 2007; 27:141–9.
35. Haritoglou C, Kook D, Neubauer A, et al. Intravitreal bevacizumab (Avastin) therapy for persistent diffuse diabetic macular edema. Retina. 2006; 26:999–1005.

36. Kook D, Wolf A, Kreutzer T, et al. Long-term effect of intravitreal bevacizumab (avastin) in patients with chronic diffuse diabetic macular edema. Retina. 2008; 28:1053–60.

Figure 1.
Bar graph illustrating percentage of eyes of each group according to best corrected visual acuity (BCVA) 3 months after treatment
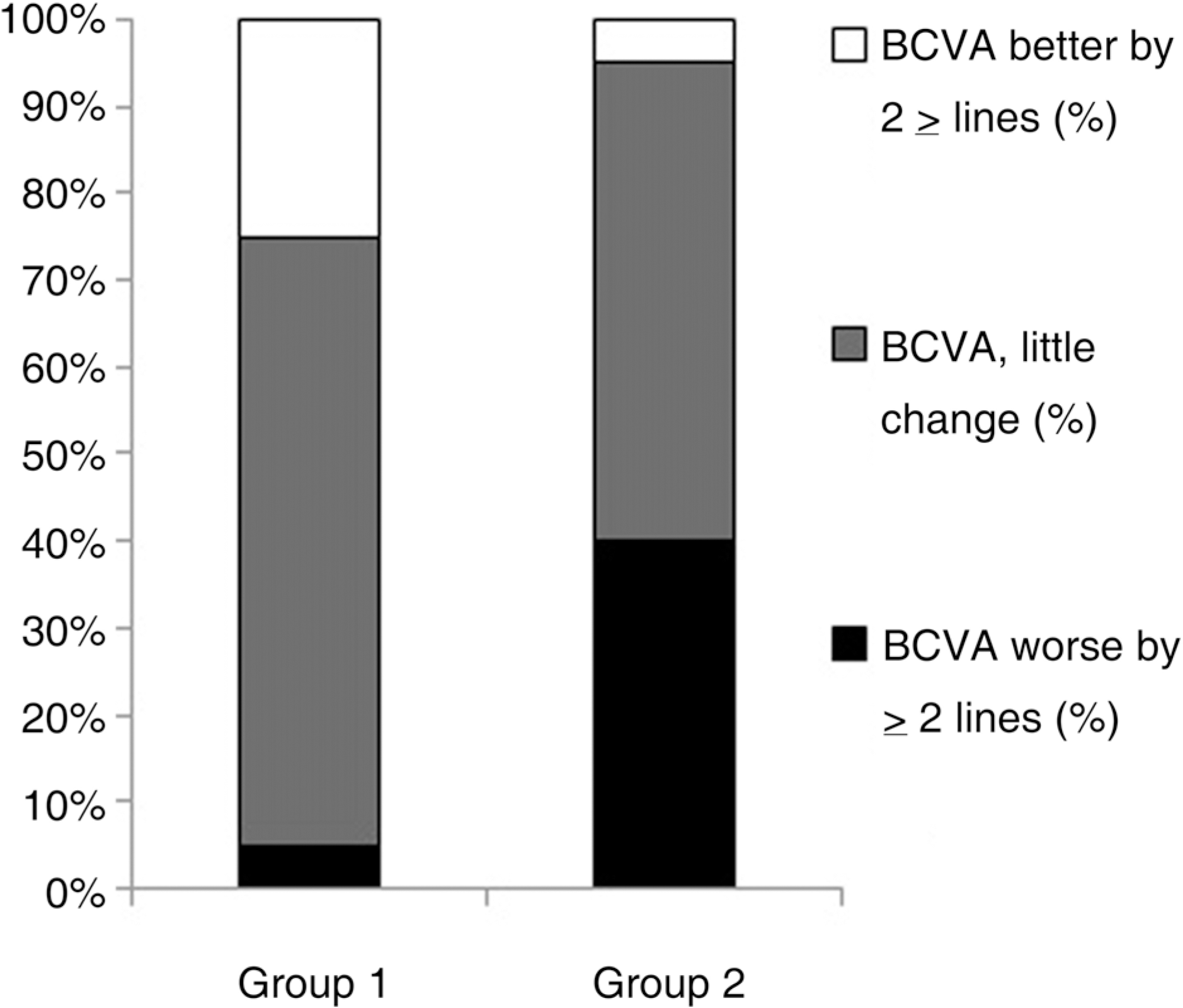
Table 1.
Demographic data of intravitreal bevacizumab injected group (Group 1) and control group (Group 2)
| Variables | Group 1 (n=20) | Group 2 (n=20) | p‡ |
|---|---|---|---|
| Gender | |||
| Male | 12 | 14 | |
| Female | 8 | 6 | |
| Age (years) | 53.5±6.79 | 54.1±6.15 | 0.871 |
| Duration of DM† (years) | 9.15±4.53 | 8.98±5.01 | 0.678 |
| Hemoglobin A1C (%) | 7.16±0.87 | 7.24±0.69 | 0.632 |
| Hypertension | 125.7±12.3 | 128.1±10.3 | 0.545 |
| (systolic/diastolic) | /83.1±14.1 | /87.4±11.7 | /0.501 |
| Lens | |||
| Phakic | 16 | 18 | |
| Pseudophakic | 4 | 2 |
Table 2.
Comparison of BCVA∗, foveal and parafoveal thickness for intravitreal bevacizumab injected group (Group 1) and control group (Group 2)
| Variables | Group 1 (n=20) | Group 2 (n=20) | p‡ |
|---|---|---|---|
| BCVA (logMAR†) | |||
| Baseline | 0.117±0.070 | 0.113±0.055 | 0.844 |
| 1 month | 0.107±0.072 | 0.216±0.144 | 0.005 |
| 3 month | 0.099±0.075 | 0.222±0.171 | 0.007 |
| Foveal thickness (µm) | |||
| Baseline | 240.5±25.9 | 238.4±24.4 | 0.798 |
| 1 month | 227.1±25.4 | 291.2±75.1 | 0.001 |
| 3 month | 222.9±26.2 | 292.8±77.2 | 0.001 |
| Parafoveal thickness(µm) | |||
| Baseline | 302.8±34.7 | 310.3±22.3 | 0.419 |
| 1 month | 300.2±28.6 | 350.5±52.9 | 0.001 |
| 3 month | 295.6±25.3 | 352.9±48.9 | 0.001 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download