Abstract
Purpose
To evaluate corneal endothelial cell changes in Optisol-GSⓇ according to enucleation time at different storage temperatures after death.
Methods
Eight rabbit cadavers (16 eyes) were stored at −3℃ and room temperature, and enucleation was performed 10 and 24 hours postmortem. The samples were divided into four groups (Group 1 was −3℃, 10 hours, Group 2 was room temperature, 10 hours, Group 3 was −3℃, 24 hours, and Group 4 was room temperature, 24 hours). The corneas were stored in Optisol-GSⓇ at 4℃, and we measured corneal endothelial cell density and thickness by specular microscopy on days 1, 3, 5, 7, 10, and 14 of preservation.
Results
The densities and thicknesses of corneal endothelial cells of each of the four groups after enucleation showed no significant difference. Corneal endothelial cell density acceptable for penetrating keratoplasty (CD>2500 cells/mm2) was found in groups 1 and 3 until 14 days, in group 2 until 10 days, and in group 4 until 7 days. In particular, eyes stored at −3℃ had less corneal endothelial cell loss than at room temperature after 7 days and 14 days (P<0.05).
Conclusions
This study demonstrated that when rabbit cadavers were stored at −3℃, corneas could be preserved in Optisol-GSⓇ for 14 days, even if the eyeballs from which they were prepared were extracted within 24 hours postmortem. Within 24 hours postmortem, the storing temperature of the cadavers was found to be more important than the enucleation time for the survival of corneal endothelial cells.
References
1. Dikstein S, Maurice DM. The metabolic basis to the fluid pump in the cornea. J Physiol. 1972; 221:29–41.

2. Filatov VP. Transplantation of cornea from preserved cadaver eyes. Lancet. 1937; 1:1395.
3. Doughman DJ, Harris JE, Schmitt M. Penetrating keratoplasty using 37℃ organ cultured cornea. Trans Sect Am Acad Ophthalmol Otolaryngol. 1976; 81:778–93.
4. Van Horn DL, Schultz RO, De Bruin J. Endothelial survival in corneal tissue stored in M-K medium. Am J Ophthalmol. 1975; 80:642–7.

5. Kaufman HE, Varnell ED, Kaufman S, et al. K-Sol corneal preservation. Am J Ophthalmol. 1985; 100:299–304.

6. Lindstrom RL, Doughman DJ, Skelnik DL, et al. Minnesota system corneal preservation. Br J Ophthalmol. 1986; 70:47–54.

7. Lindstrom RL. Advances in corneal preservation. Trans Am Ophthalmol Soc. 1990; 88:555–648.
8. Taylor MJ. Clinical cryobiology of tissues: preservation of corneas. Cryobiology. 1986; 23:323–53.

9. Lindstrom RL, Kaufman HE, Skelnik DL, et al. Optisol corneal storage medium. Am J Ophthalmol. 1992; 114:345–56.

10. Kim IS, Kim MS. A Comparative Evaluation of 4℃ Corneal Preservation Media, Optisol and Likorol. J Korean Ophthalmol Soc. 1996; 37:776–81.
11. Yang SW, Oh SJ, Kim KS, et al. Morphologic evaluation of cat corneal endothelium preserved in korean corneal storage medium. J Korean Ophthalmol Soc. 2000; 41:2652–62.
12. Baumann G, Fries U, Schnaudigel OE. Viability of corneal endothelium after long-term storage at +4 degrees C in Optisol. Ophthalmology. 1994; 91:624–7.
13. Patel HY, Brookes NH, Moffatt L, et al. The New Zealand National Eye Bank study 1991-2003: a review of the source and management of corneal tissue. Cornea. 2005; 24:576–82.
14. Oh YH, Lee SW. The change of the permeability of the rabbit corneal endothelium in various storage time. J Korean Ophthalmol Soc. 1971; 12:91–5.
15. Hagenah M, Simon B, Bohnke M. Experimental corneal cryopreservation: Impact of postmortem time on corneal endothelial cell survival. Ophthalmic Res. 1993; 25:210–5.

16. Waring Go, Bourne WM, Edelhauser HF, Kenyon KR. The corneal endothelium: Normal and pathologic structure and function. Ophthalmology. 1982; 92:531–90.
17. Green K. Corneal endothelial structure and function under normal and toxic conditions. Cell Biol Rev. 1991; 25:169–207.
Figure 1.
Changes of endothelial cell density according to preservation periods. Endothelial cells over 2500 cells/mm2 is maintained upto 2 weeks in Group 1 and 3, but cells under 2500 cells/mm2 is observed at 2 weeks in Group 2 and after 10 days in Group 4. During the preservation time, the decrease of endothelial cell density of between groups is statistically significant (P<0.05 by repeated measured ANOVA).
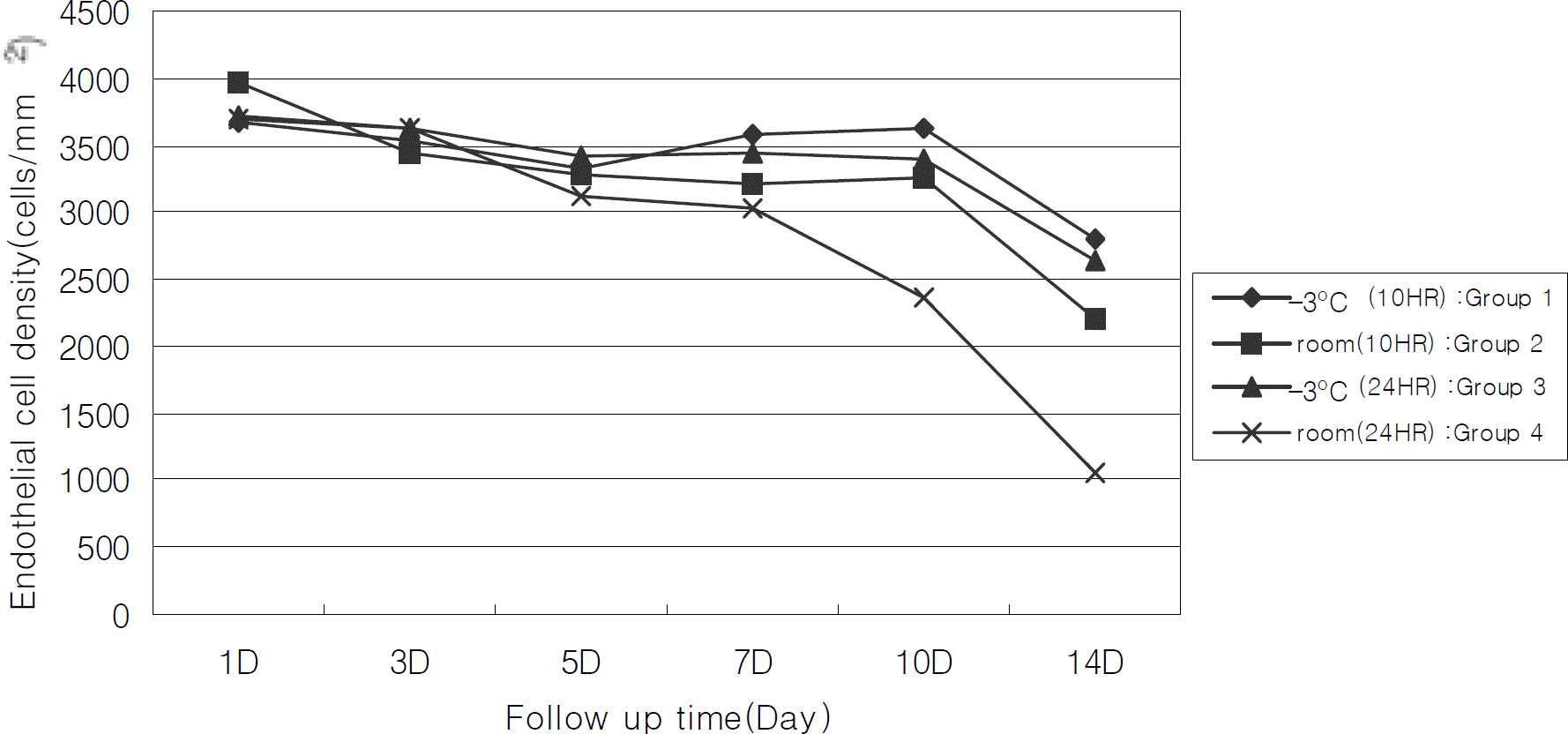
Figure 2.
Endothelial cell loss after 7 and 14 days (A: 1-7 Day, B: 7-14 Day). The decrease of endothelial cell density of −3℃ groups (group 1 and 3) was significantly less than that of room temperature groups (group 2 and 4) (* P<0.05 by two way ANOVA).
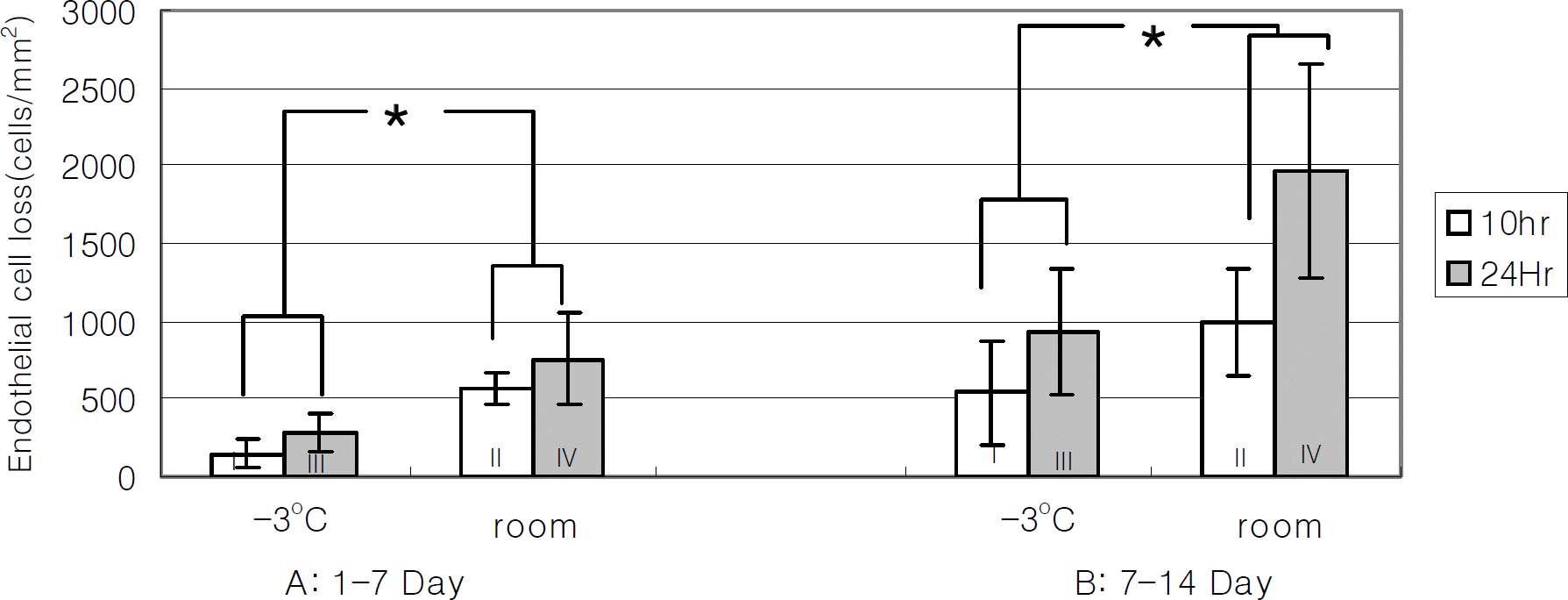
Figure 3.
Changes of corneal thickness according to preservation periods. The corneal thickness of each groups is increased during preservation periods, but no significant change is observed between groups (repeated measured ANOVA).
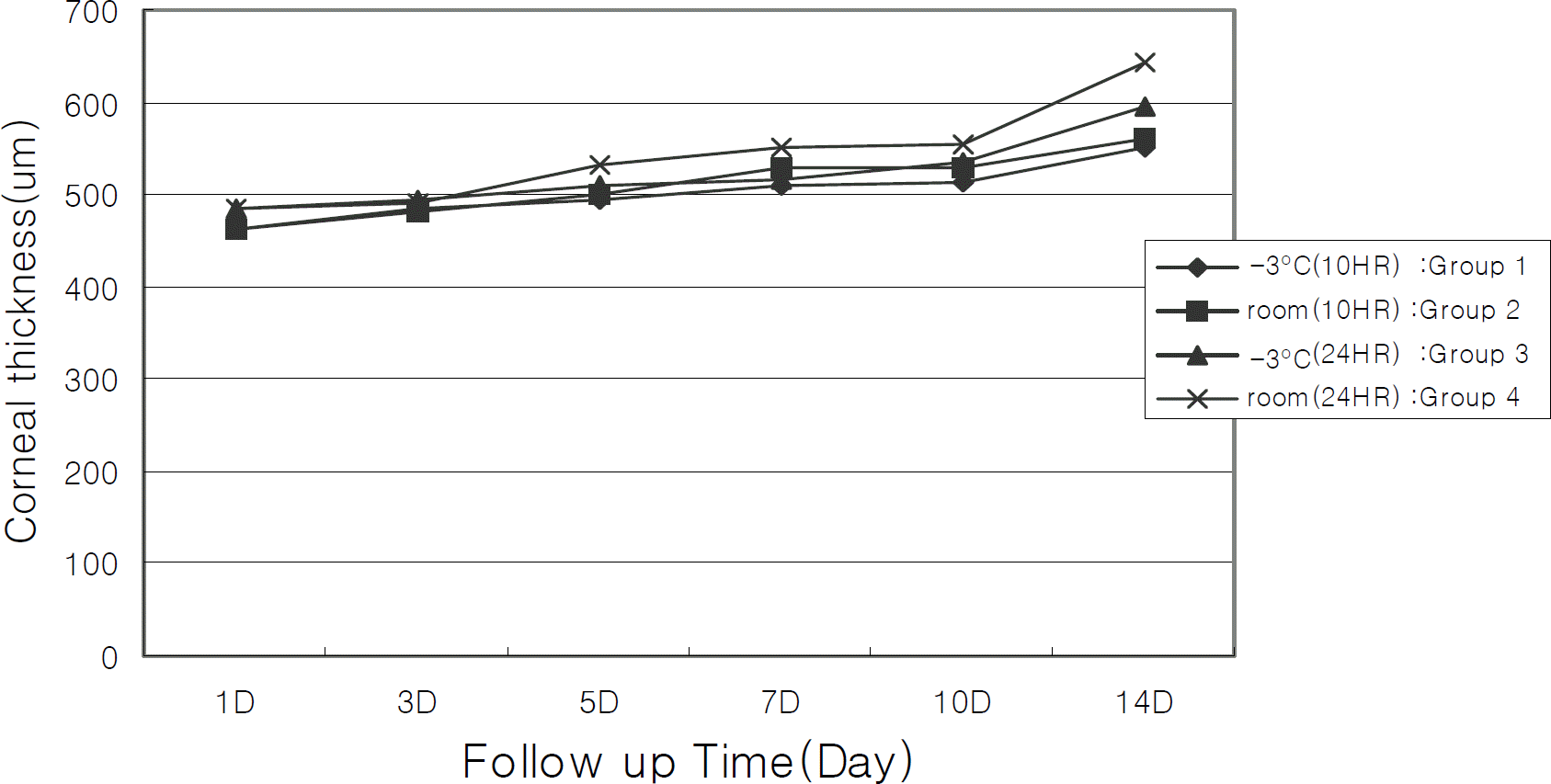
Figure 4.
Changes of corneal endothelial polymegathism (coefficiency of variation) according to preservation periods. The coefficiency of variation of each groups is increased during preservation periods, but no significant change is observed between groups (repeated measured ANOVA).
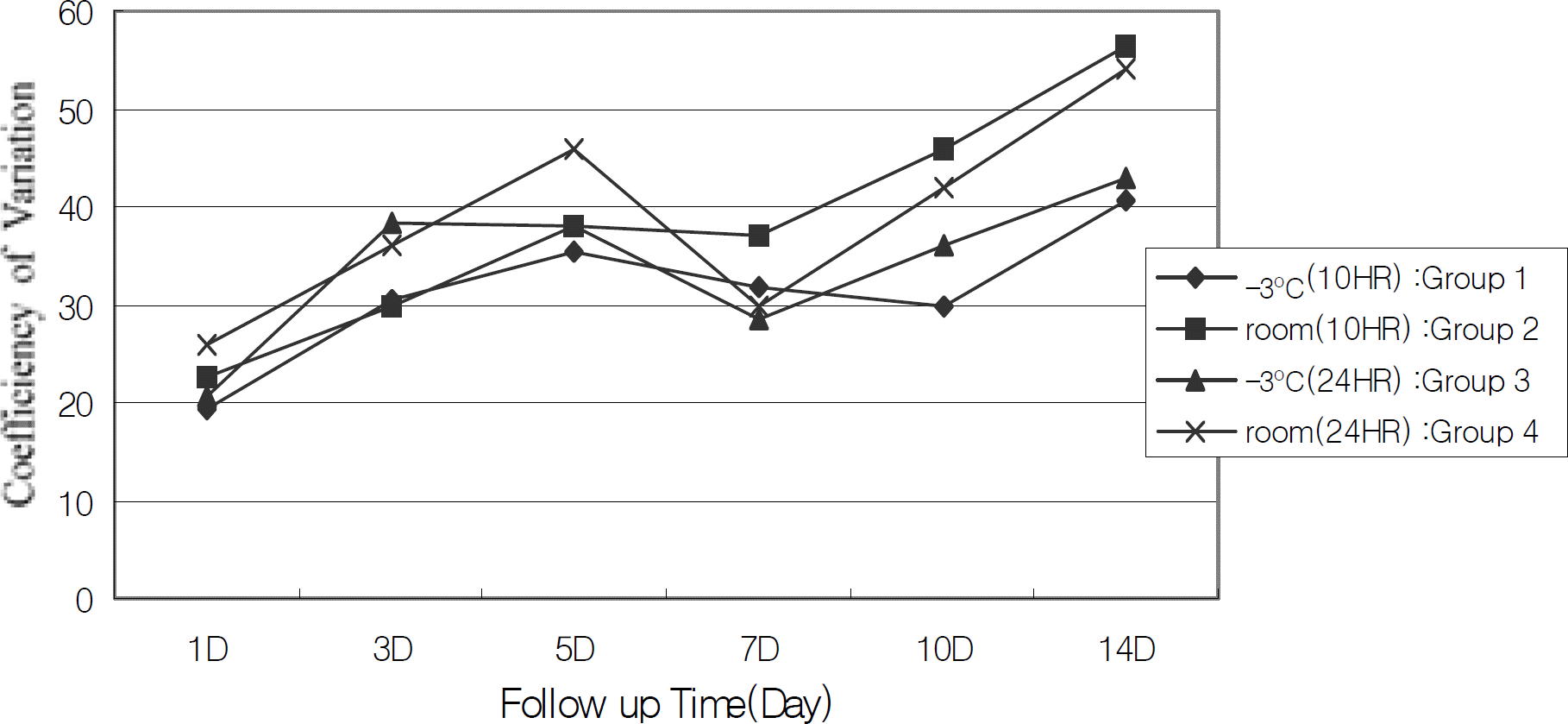
Figure 5.
Changes of corneal endothelial pleomorphism (hexagonality) according to preservation periods. The hexagonality of each groups is decreased during preservation periods, but no significant change is observed between groups (repeated measured ANOVA).
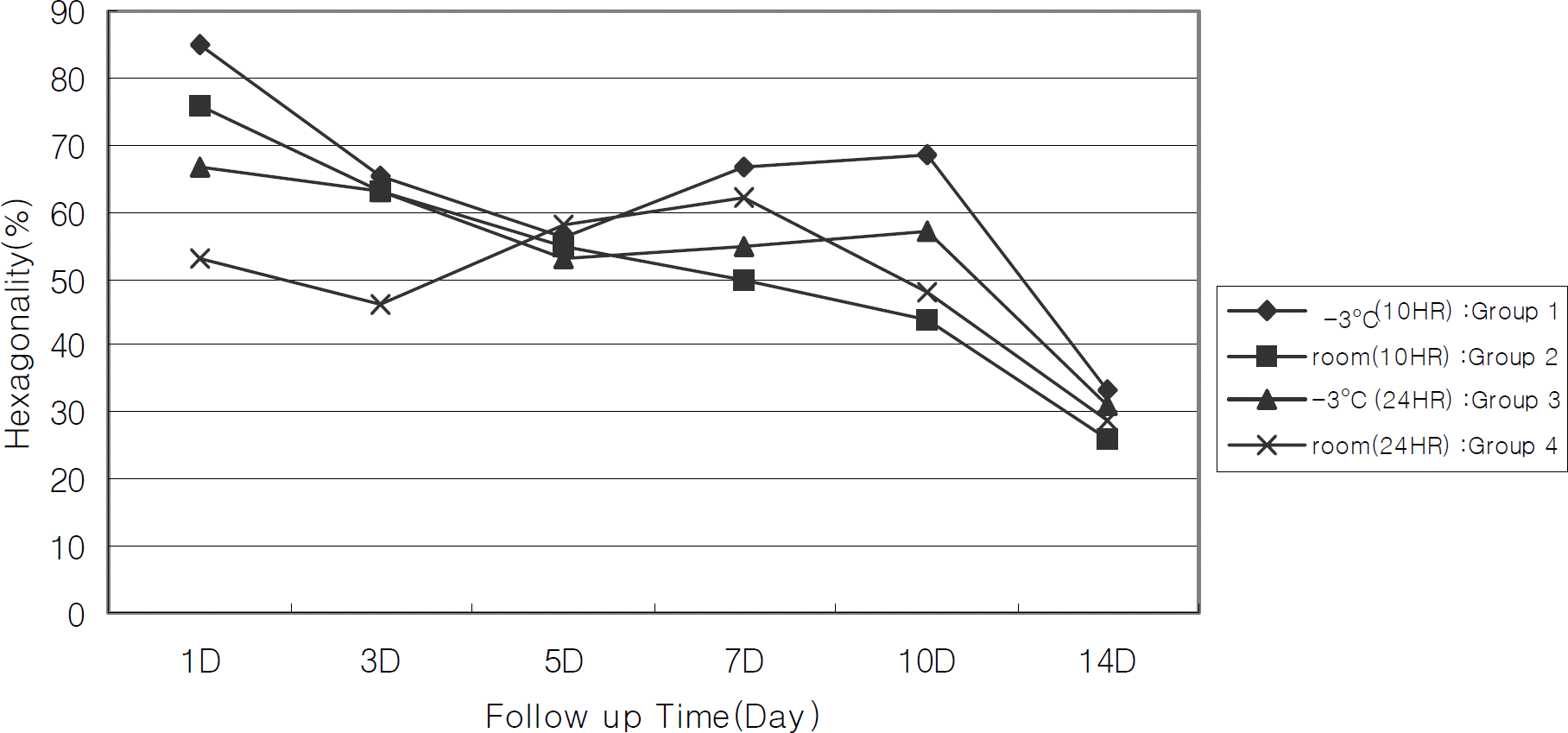
Figure 6.
Corneal specular microscopic findings according to preservation periods. (Group 1: A-D; A-1 Day, B-7 Day, C-10 Day, D-14 Day; Group 2: E-F; E-1 Day, F-7 Day, G-10 Day, H-14 Day)

Figure 7.
Corneal specular microscopic findings according to preservation periods.(Group 3: A-D; A-1 Day, B-7 Day, C-10 Day, D-14 Day; Group 4: E-F; E-1 Day, F-7 Day, G-10 Day, H-14 Day)

Table 1.
Characteristics of rabbit corneas on postoperative 1 day




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download