Abstract
Purpose
To investigate the effects of anterior displacement of four rectus muscles in exposure of porous polyethylene orbital implant (Medpor).
Case summary
This retrospective study reviewed 4 eyes with exposed orbital implant who underwent evisceration with porous polyethylene orbital implant (Medpor®). The technique involves dissection of conjunctiva and tenon's capsule and isolation of four rectus muscles from sclera. Unsure if each muscle was sutured to each other or if a group of muscles were sutured to another group (i.e. superior and inferior to medial and lateral). Posterior tenon's capsule was closed interruptedly and conjunctiva was closed continuously. Orbital implant exposure was occurred at 2∼36 months later after evisceration and implant insertion. One eye was noted wound dehiscence 1 week after operation, then re-suture was done. Re-exposure was notified in 1 eye at postoperative 3 months, but it was small, then we just observed. In other 3 cases, exposure was not identified till last follow up.
References
1. Kaltreider SA, Newman SA. Prevention and management of complications associated with the hydroxyapatite implants. Ophthal Plast Reconstr Surg. 1996; 12:18–31.
2. Nunery WR, Heinz GW, Bonnin JM, et al. Exposure rate of hydroxyapatite spheres in the anophthalmic socket: histopathologic correlation and comparison with silicone sphere implants. Ophthal Plast Reconstr Surg. 1993; 9:96–104.
3. Remulla HD, Rubin PAD, Shore JW, et al. Complications of porous spherical orbital implants. Ophthalmology. 1995; 102:586–93.

4. Buettner H, Bartley GB. Tissue breakdown and exposure associated with orbital hydroxyapatite implants. Am J Ophthalmol. 1992; 113:669–73.

5. Oestreicher JH, Liu E, Berkowitz M. Complications of hydroxyapatite orbital implants: a review of 100 consecutive cases and a comparison of Dexon mesh (polyglycolic acid) with scleral wrapping. Ophthalmology. 1997; 104:324–9.
6. Christmas NJ, Gordon CD, Murray TG, et al. Intraorbital implants after enucleation and their complications: a 10-year review. Arch Ophthalmol. 1998; 116:1199–203.
7. Bosniak SL. Reconstruction of the anophthalmic socket: state of the art. Adv Ophthal Plast Reconstr Surg. 1988; 7:313–48.
8. Beaver HA, Patrinely JR, Holds JB, Soper MP. Periocular autografts in socket reconstruction. Ophthalmology. 1996; 103:1498–502.

9. Wiggs EO, Becker BB. Extrusion of enucleation implants: treatment with secondary implants and autogenous temporalis fascia or fascia lata grafts. Ophthalmic Surg. 1992; 23:472–6.
10. Meltzer MA. Complications of enucleation and evisceration: prevention and treatment. Int Ophthalmol Clin. 1992; 32:213–33.
11. Kim YD, Goldberg RA, Shorr N, Steinsapir KD. Management of exposed orbital implants. Ophthalmology. 1994; 101:1709–15.
12. Goldberg MF. A simplified scleral graft technique for covering an exposed orbital implant. Ophthalmic Surg. 1988; 19:206–11.

13. Goldberg RA, Holds JB, Ebrahimpour J. Exposed hydroxyapatite implants, report of six cases. Ophthalmology. 1992; 99:831–6.
14. Helveston EM. Human bank scleral patch for repair of exposed or extruded orbital implants. Arch Ophthalmol. 1969; 82:83.
15. Massry GG, Holds JB. Frontal periosteum as an exposed orbital implant cover. Ophthal Plast Reconstr Surg. 1999; 15:79–82.

16. Sagoo MS, Olver JM. Autogenous temporalis fascia patch for porous polyethylene (Medpor®) sphere orbital implant exposure. Br J Ophthalmol. 2004; 88:942–6.
17. Lee SY, Lee JH. Clinical experiences of hydroxyapatite implantation. J Korean Ophthalmol Soc. 1993; 34:1275–80.
19. Shields CL, Shields JA, De Potter P. Hydroxyapatite orbital implant after enucleation Experience with initial 100 consecutive cases. Arch Ophthalmol. 1992; 110:333–8.

20. Rubin PA, Popham JK, Bilyk JR, Shore JW. Comparison of fibrovascular ingrowth into hydroxyapatite and porous polyethylene orbital implants. Ophthalmic Plast Reconstr Surg. 1994; 10:96–103.

21. Rosen HM, McFarland MM. The biologic behavior of hydroxyapatite implanted into the maxillofacial skeleton. Plast Reconstr Surg. 1990; 85:718–23.

22. Lee CS, Paik HJ, Song MS. Clinical Evaluation of Medpor® Orbital Implant. J Korean Ophthalmol Soc. 2000; 41:299–306.
23. Karesh JW, Dresner SC. High-density porous polyethylene (Medpor®) as a successful anophthalmic socket implant. Ophthalmology. 1994; 101:1688–96.
24. Baek SH. Clinical effect of porous polyethylene (Medpor®) orbital implants. J Korean Ophthalmol Soc. 2000; 41:1858–63.
25. Karcioglu ZA, Al-mesfer SA, Mullaney PB. Porous polyethylene orbital implants in patients with retinoblastoma. Ophthalmology. 1998; 105:1311–6.
26. Martin P, Ghabrial R. Repair of exposed hydroxyapatite orbital implant by a tarsoconjunctival pedicle flap. Ophthalmology. 1998; 105:1694–7.

27. Fountain JA, Helveston EM. A long-term follow-up study of sclera grafting for exposed or extruded orbital implant. Am J Ophthalmol. 1982; 93:52–6.
28. McCord CD, Tanenbaum M. Oculoplastic Surgery. 3rd ed.New York: Raven Press;1995. p. 609–37.
29. Son MG, Kim YD. Evisceration with hydroxyapatite implants. J Korean Ophthalmol Soc. 2000; 41:598–606.
Figure 1.
Four Rectus muscle anterior displacement. (A & B) After dissection of the conjunctiva and Tenon's capsule with Westcott scissors, we identified 4 rectus muscles with a muscle hook. Then, the rectus muscles were isolated in the insertion site with 6-0 Vicryl. (C & D) After 4 rectus muscles were separated from the sclera, inflammatory and necrotic tissues surrounding exposed orbital implant were removed. (E & F) Among 4 rectus muscles, the medial rectus muscle was sutured with the lateral rectus muscle and the superior rectus muscle was sutured with the inferior rectus muscle interruptedly. Finally, posterior Tenon's capsule was sutured with 5-0 Vicryl, and th conjunctiva was sutured with 6-0 Vicryl.
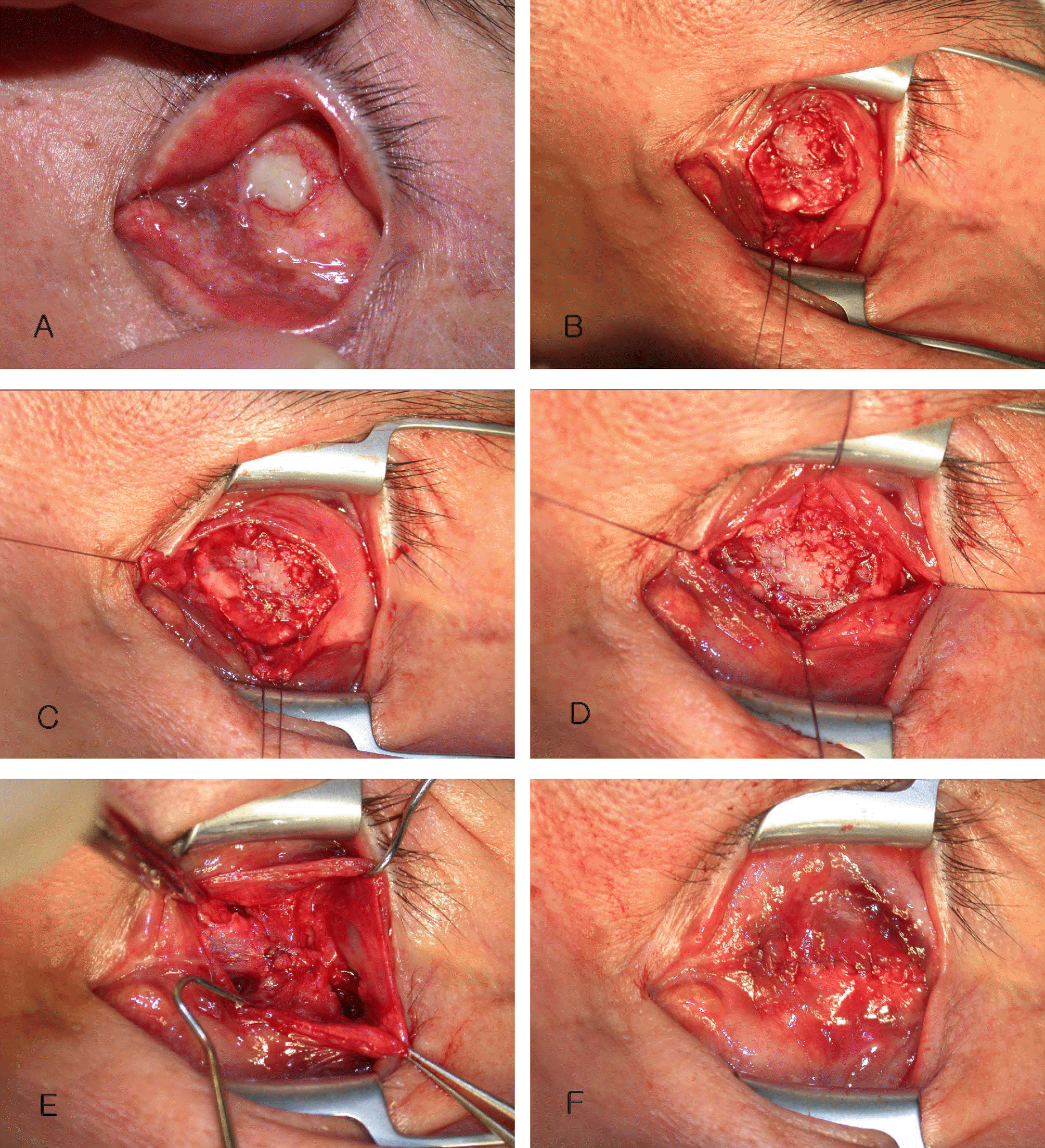
Figure 2.
Case 1 (A) The small exposure site (2 mm×3 mm in size) of orbital implant was noted. (B). At 1 week postoperatively (4 rectus muscle anterior displacement), the conjunctiva was well sutured with 6-0 Vicryl. The conjunctiva was completely healed at the postoperative 3 weeks.
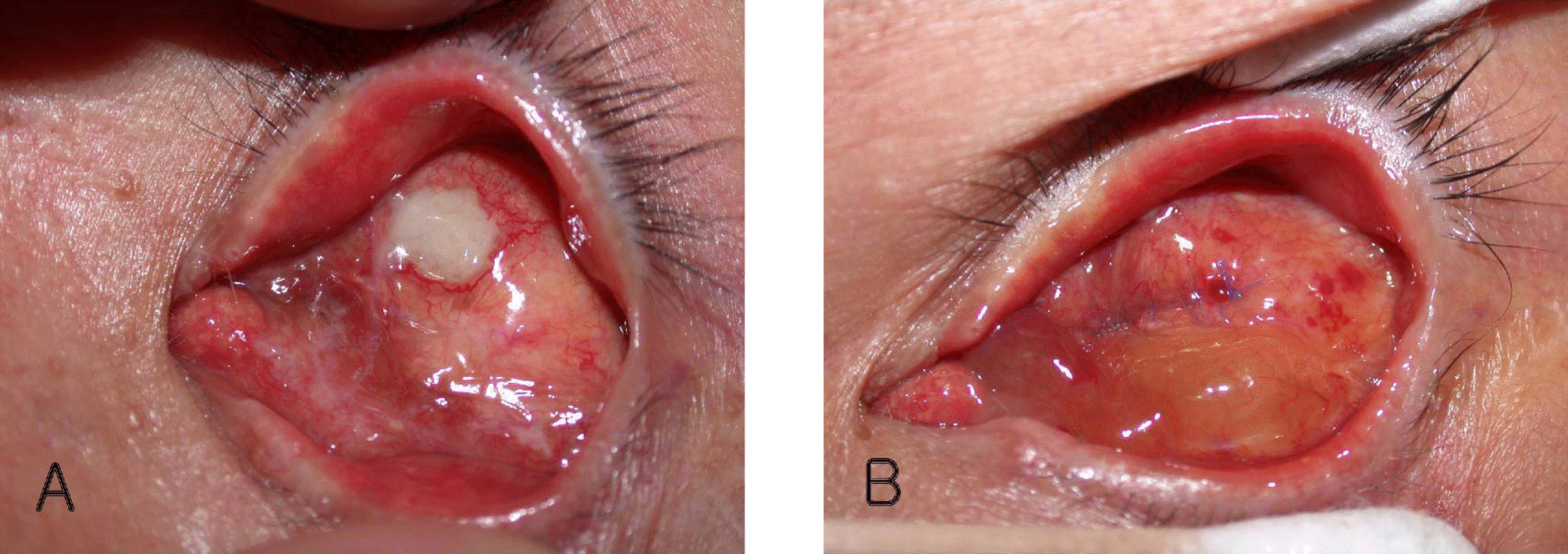
Figure 3.
Case 2 (A) About 6 mm×7 mm-sized conjunctival defect around impacted MCP of an orbital implant was visible at 29 months postoperatively. Antiobiotic and steroid eye solutions were used. Providencia rettgeri was cultured from yellowish discharge. (B) At 4 months postoperatively, the orbital implant was re-exposed measuring about 2 mm×2 mm in size of the exposure. Thereafter the exposure site was not enlarged until the last follow up.
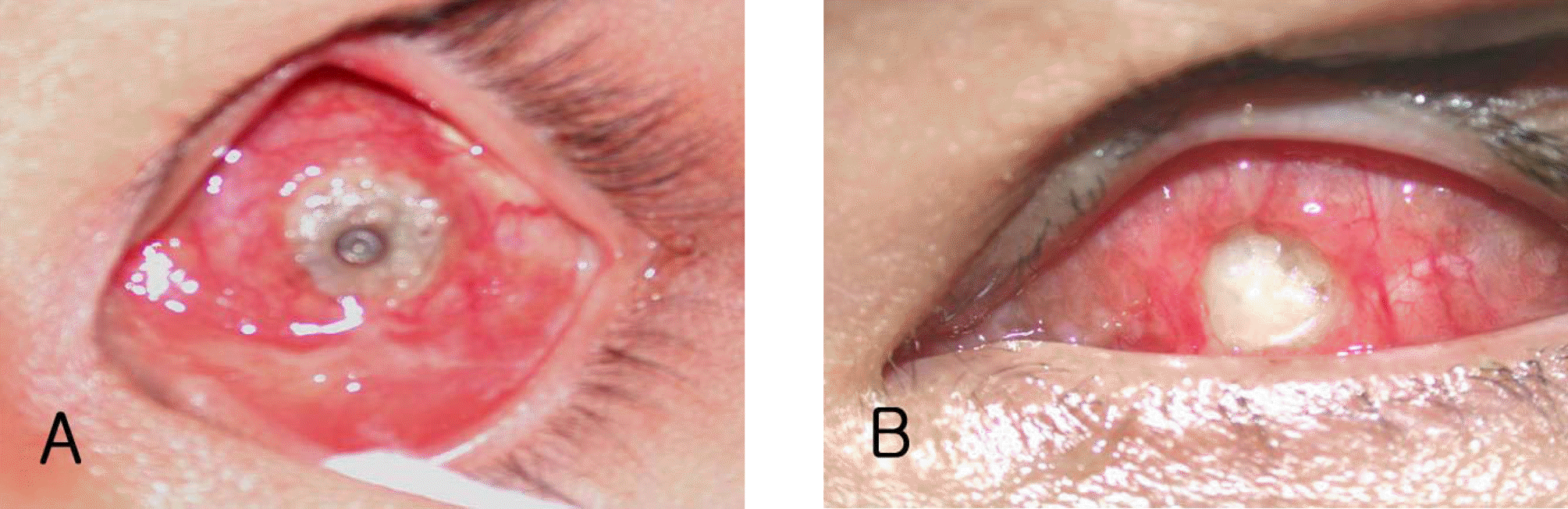
Figure 4.
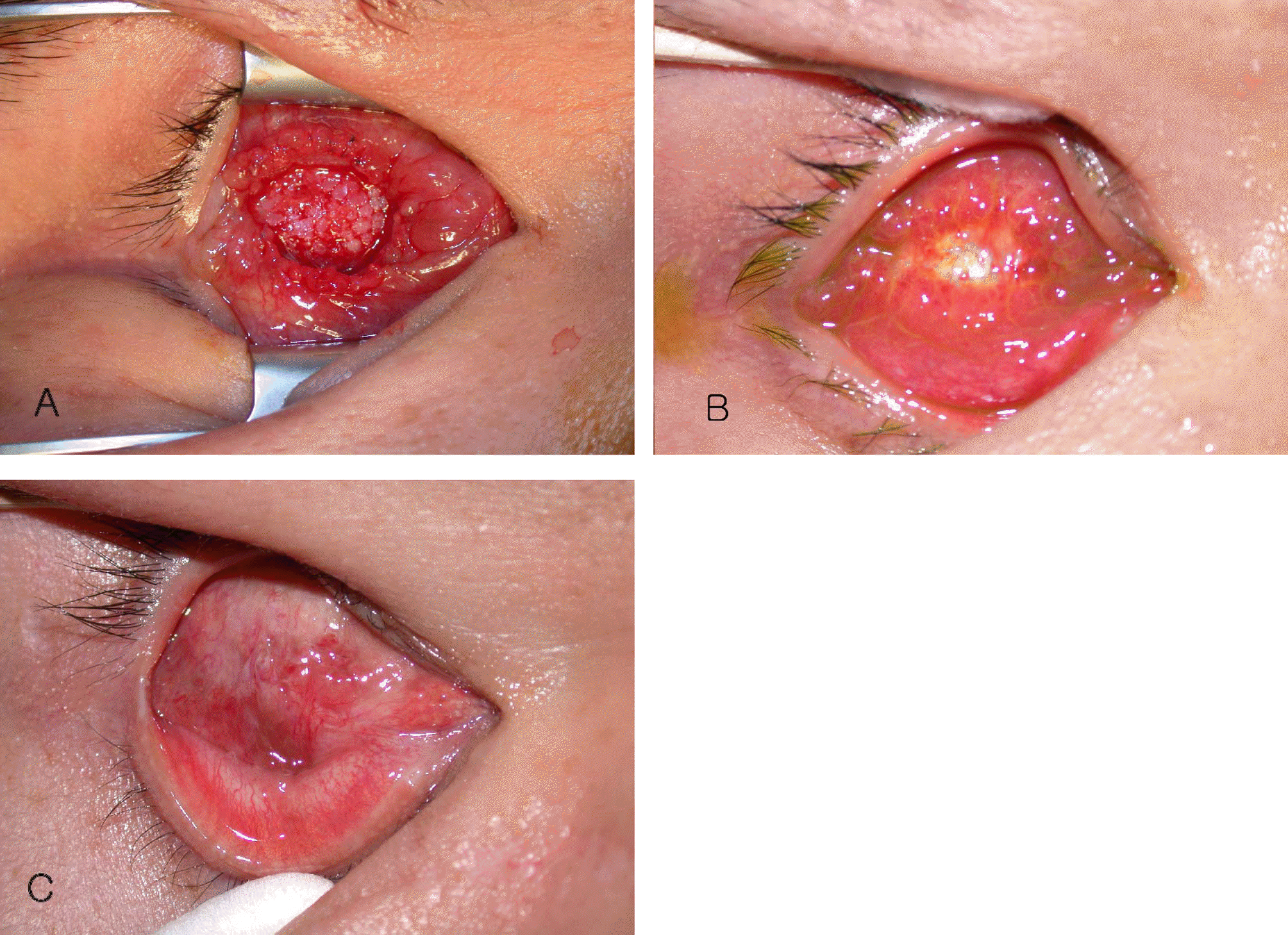
Case 3 About 7 mm×7 mm-sized conjunctival defect around the MCP inserted site of porous polyethylene orbital implant (Medpor®) was visible at postoperative 48 months. And Streptococcus viridans was cultured. After antibiotic therapy, the inflammation was controlled, and 4 rectus muscle anterior displacement was performed. At 6 weeks postoperatively, the conjunctiva was completely healed. (B) At 5 months postoperatively, the granulation tissue was noted, but re-exposure was not detected.
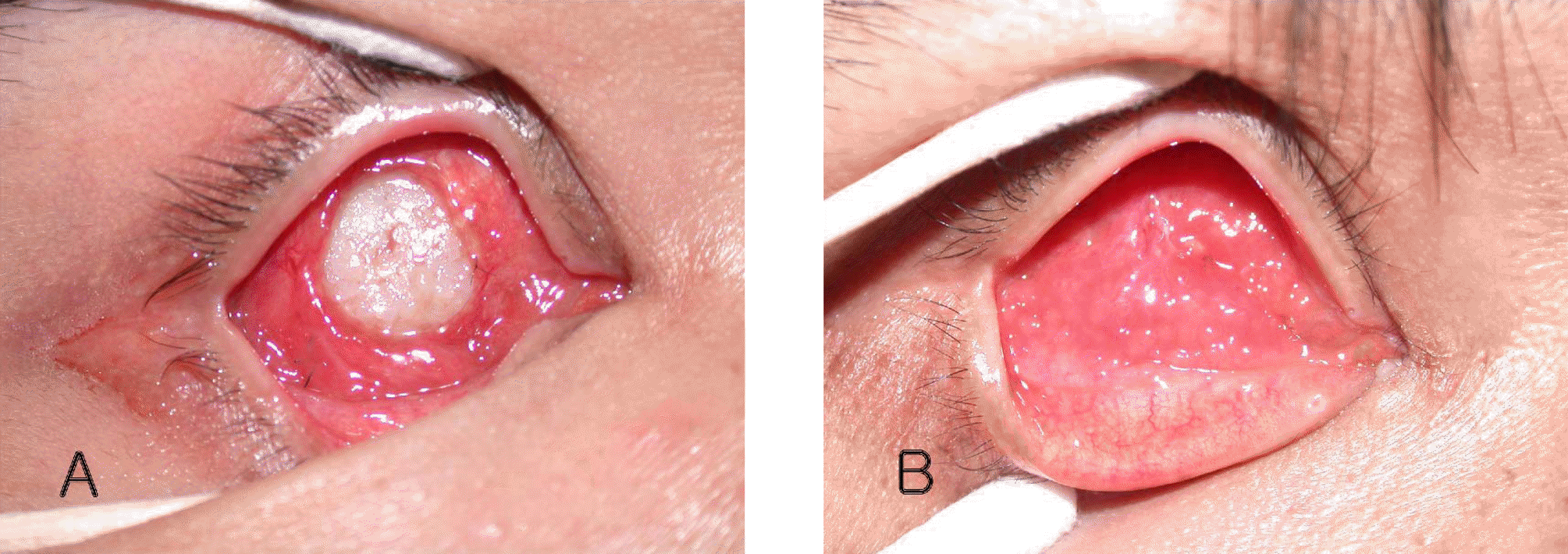
Figure 5.
Case 4 (A) A 6 mm×6 mm-sized conjunctival defect developed at 2 months postoperatively, then the scleral patch graft was performed. Two months later, however, the conjunctival defect reccurred. (B) The dermis fat graft was done, but re-exposure of the orbital implant was noted at 3 months postopteratively. (C) After 4 rectus muscle anterior displacement was performed, the conjunctiva was completely healed at 6 weeks postopetatively.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download