Abstract
Background/Aims
Adequate bowel preparation is an essential factor affecting the visibility of colonic mucosa and safety of related therapeutic interventions. The aim of this study was to assess the efficacy, tolerability, and safety of three bowel preparation agents –2 L polyethylene glycol with ascorbic acid (PEGA), sodium picosulfate magnesium citrate (SPMC), and oral sodium phosphate tablet (NaP)– for morning colonoscopy.
Methods
Here, we analyzed the medical records of patients who had taken bowel preparation agents using the split-dose method and undergone colonoscopy in a single hospital. The efficacy of bowel preparation agents was evaluated using the Ottawa bowel preparation assessment tool. The safety and tolerability of the agents were assessed by measuring the renal function and electrolytes prior to and after the procedure as well as by assessing the self-reported questionnaire.
Results
Of the 365 patients (PEGA:163, SPMC: 93, NaP: 109), 98.6% ingested more than 90% of the agents. NaP showed an inferior cleansing efficacy, and serum phosphate elevation was significantly higher in the NaP group. However, the satisfaction score was lowest in the PEGA group. Age (odds ratio [OR] 0.96, 95% confidence interval [CI] 0.92–0.99, p=0.04) and preparation agents (OR of PEGA versus NaP 5.0, 95% CI 2.28–10.97, p<0.001) (OR of SPMC versus NaP 2.73, 95% CI 1.22–6.08, p=0.01) were independently associated with bowel preparation success.
Conclusions
According to our analysis, NaP showed an inferior cleansing efficacy compared with PEGA and SPMC, which may be attributed to the complex administration method and lower water intake. However, large-volume ingestion remains unsatisfactory for patients. Detailed bowel preparation instructions could enhance bowel cleansing efficacy.
Go to : 
References
1. Harewood GC, Sharma VK, de Garmo P. Impact of colonoscopy preparation quality on detection of suspected colonic neoplasia. Gastrointest Endosc. 2003; 58:76–79.

2. Rex DK, Petrini JL, Baron TH, et al. Quality indicators for colonoscopy. Gastrointest Endosc. 2006; 63(4 Suppl):S16–S28.

3. Hoy SM, Scott LJ, Wagstaff AJ. Sodium picosulfate/magnesium citrate: a review of its use as a colorectal cleanser. Drugs. 2009; 69:123–136.
4. Belsey J, Crosta C, Epstein O, et al. Meta-analysis: the relative abdominal of oral bowel preparations for colonoscopy 1985–2010. Aliment Pharmacol Ther. 2012; 35:222–237.
5. Belsey J, Epstein O, Heresbach D. Systematic review: adverse event reports for oral sodium phosphate and polyethylene glycol. Aliment Pharmacol Ther. 2009; 29:15–28.

6. Choi NK, Lee J, Chang Y, et al. Acute renal failure following oral sodium phosphate bowel preparation: a nationwide case-abdominal study. Endoscopy. 2014; 46:465–470.
7. Rostom A, Jolicoeur E. Validation of a new scale for the abdominal of bowel preparation quality. Gastrointest Endosc. 2004; 59:482–486.
8. Lawrance IC, Willert RP, Murray K. Bowel cleansing for abdominal: prospective randomized assessment of efficacy and of abdominal mucosal abnormality with three preparation agents. Endoscopy. 2011; 43:412–418.
9. Schmidt LM, Williams P, King D, Perera D. Picoprep-3 is a superior colonoscopy preparation to fleet: a randomized, controlled trial comparing the two bowel preparations. Dis Colon Rectum. 2004; 47:238–242.

10. Tjandra JJ, Chan M, Tagkalidis PP. Oral sodium phosphate (fleet) is a superior colonoscopy preparation to picopre (sodium pico-sulfate-based preparation). Dis Colon Rectum. 2006; 49:616–620.
11. Worthington J, Thyssen M, Chapman G, Chapman R, Geraint M. A randomised controlled trial of a new 2 litre polyethylene glycol solution versus sodium picosulphate + magnesium citrate abdominal for bowel cleansing prior to colonoscopy. Curr Med Res Opin. 2008; 24:481–488.
12. Dakkak M, Aziz K, Bennett JR. Short report: comparison of two orally administered bowel preparations for colonoscopy–abdominal glycol and sodium picosulphate. Aliment Pharmacol Ther. 1992; 6:513–519.
13. Saunders BP, Masaki T, Fukumoto M, Halligan S, Williams CB. The quest for a more acceptable bowel preparation: comparison of a polyethylene glycol/electrolyte solution and a mannitol/picolax mixture for colonoscopy. Postgrad Med J. 1995; 71:476–479.

14. Kim HG, Huh KC, Koo HS, et al. Sodium picosulfate with abdominal citrate (SPMC) plus laxative is a good alternative to conventional large volume polyethylene glycol in bowel preparation: a multicenter randomized single-blinded trial. Gut Liver. 2015; 9:494–501.
15. Ness RM, Manam R, Hoen H, Chalasani N. Predictors of abdominal bowel preparation for colonoscopy. Am J Gastroenterol. 2001; 96:1797–1802.
16. Chung YW, Han DS, Park KH, et al. Patient factors predictive of inadequate bowel preparation using polyethylene glycol: a abdominal study in Korea. J Clin Gastroenterol. 2009; 43:448–452.
17. Chan WK, Saravanan A, Manikam J, Goh KL, Mahadeva S. Appointment waiting times and education level influence the quality of bowel preparation in adult patients undergoing colonoscopy. BMC Gastroenterol. 2011; 11:86.

18. Nguyen DL, Wieland M. Risk factors predictive of poor quality preparation during average risk colonoscopy screening: the abdominal of health literacy. J Gastrointestin Liver Dis. 2010; 19:369–372.
Go to : 
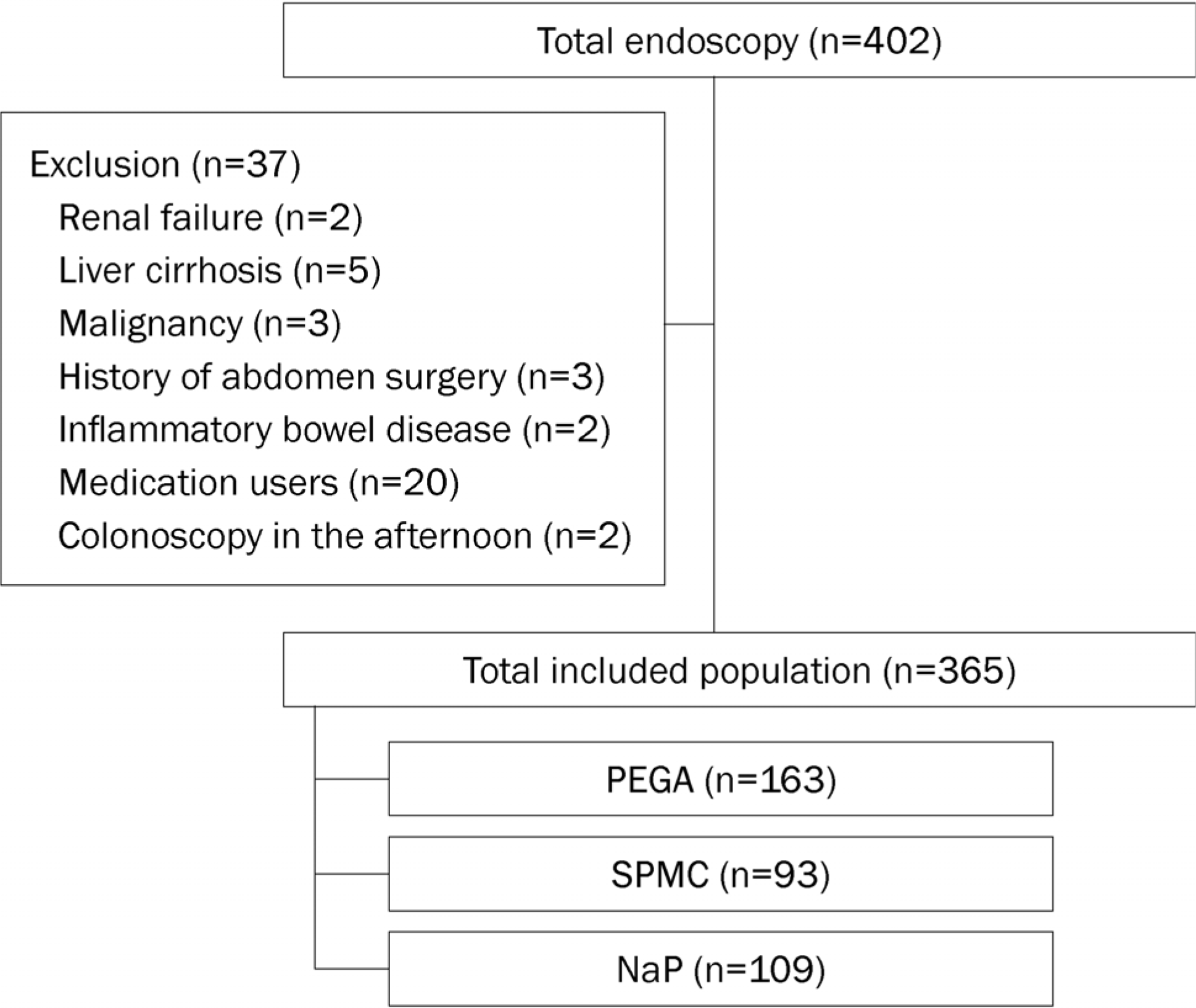 | Fig. 1.Flow diagram of study. PEGA, polyethylene glycol+ascorbic acid; SPMC, sodium picosulfate magnesium citrate; NaP, sodium phosphate. |
Table 1.
Clinical Characteristics of Total Enrolled Population
Table 2.
Cleansing Efficacy of Bowel Preparation Agents
| Component of Ottawa bowel preparation scale | PEGA (n=163) | SPMC (n=93) | NaP (n=109) | p-value |
|---|---|---|---|---|
| Right colon | 0.93±0.69 | 0.97±0.84 | 1.51±1.07 | <0.001 a |
| Mid colon | 1.01±0.56 | 1.05±0.81 | 1.34±0.93 | 0.005 b |
| Rectosigmoid colon | 0.56±0.64 | 0.69±0.88 | 1.04±0.99 | <0.001 c |
| Colonic fluid | 1.06±0.37 | 1.09±0.41 | 1.05±0.48 | 0.77 |
| Total score | 3.57±1.70 | 3.82±2.55 | 4.93±2.96 | <0.001 d |
Table 3.
Safety Profile of Bowel Preparation Agents
| PEGA (n=92) | SPMC (n=72) | NaP (n=83) | p-value | |
|---|---|---|---|---|
| Na (mmol/L) | 0.40 | −0.33 | 0.81 | 0.91 |
| Cl (mmol/L) | 1.29 | −0.80 | −0.11 | 0.03a |
| P (mg/dL) | −0.07 | 0.07 | 3.85 | <0.001b |
| Ca (mg/dL) | 0.07 | 0.14 | −0.35 | <0.001c |
| Cr (mg/dL) | 0.01 | 0.01 | 0.01 | 0.91 |
| eGFR (mL/min) | −1.10 | −0.79 | −0.97 | 0.98 |
Table 4.
Tolerability of Bowel Preparation Agents
| PEGA (n=163) | SPMC (n=93) | NaP (n=109) | p-value | |
|---|---|---|---|---|
| Could not intake all the preparation agents | 3 (1.8) | 1 (1.1) | 1 (0.9) | 0.86 |
| Satisfaction score | 7.29±2.00 | 8.25±1.71 | 8.12±1.60 | <0.001a |
| Refuse to take the same preparation agent in the future | 48 (29.4) | 16 (17.2) | 17 (15.6) | 0.01 |
Table 5.
Multivariable Logistic Regression Analysis Assessing the Associated Factors of Successful Bowel Preparation (Ottawa bowel preparation scale score 7 or less)




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download