Abstract
Background/Aims
In gastric cancer, the rate of recurrence and metastasis following radical resection is high, necessitating improvement in survival and cure rates. Neoadjuvant chemotherapy (NAC) has potential benefits for locally advanced gastric cancer; however, the surgical benefits and effects on survival are unclear. This study evaluates the effectiveness of NAC in locally advanced gastric cancer and compares clinical outcomes of doublet and triplet regimens.
Methods
We reviewed patient medical records of 383 patients who underwent NAC (n=41) or surgery only (n=342) for treatment of locally advanced gastric cancer. The baseline characteristics and clinical outcomes were compared between the groups. Chemotherapy patients were classified according to regimen, doublet (n=28) and triplet (n=13), and NAC-related clinical response, safety, and toxicity were analyzed.
Results
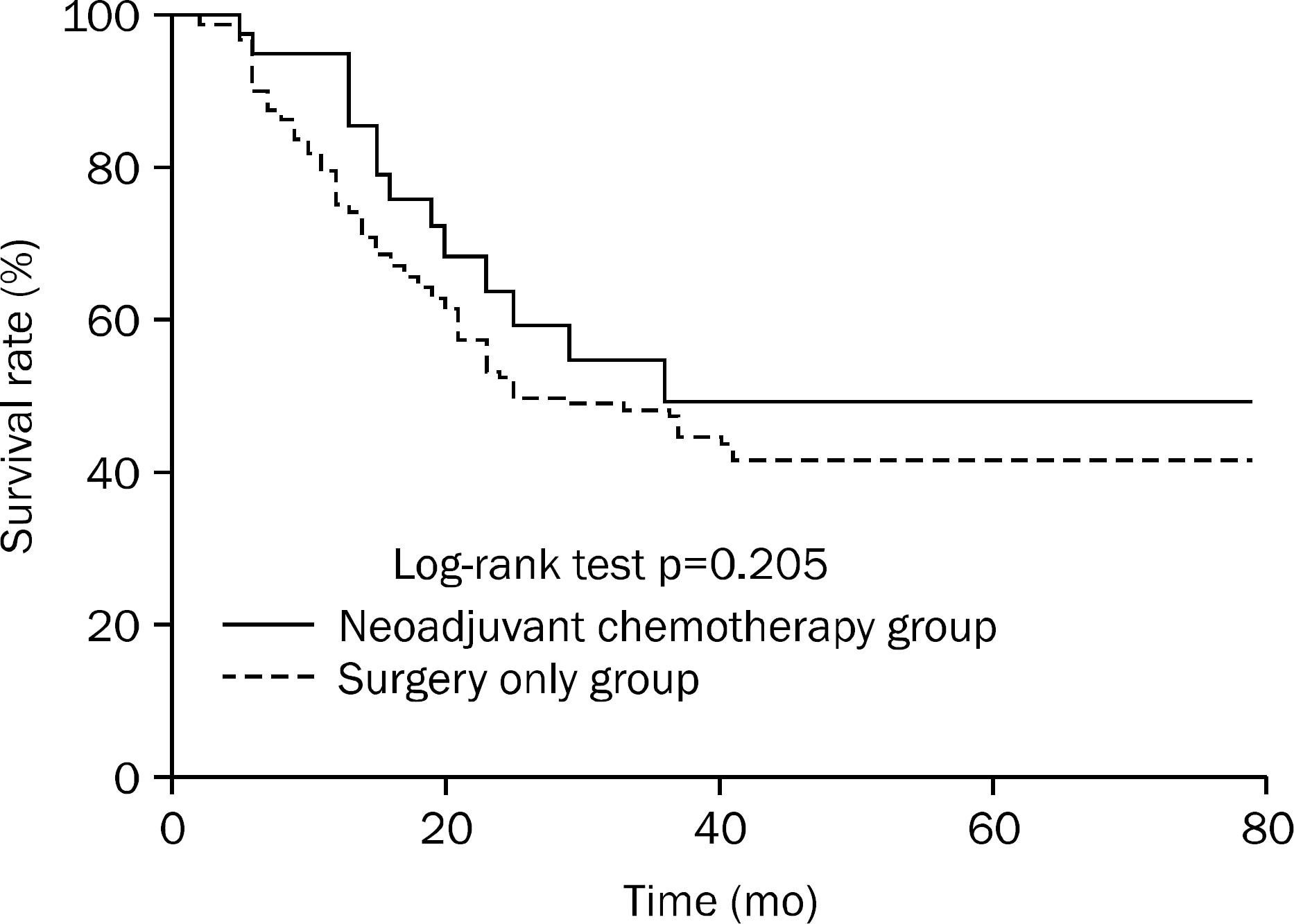
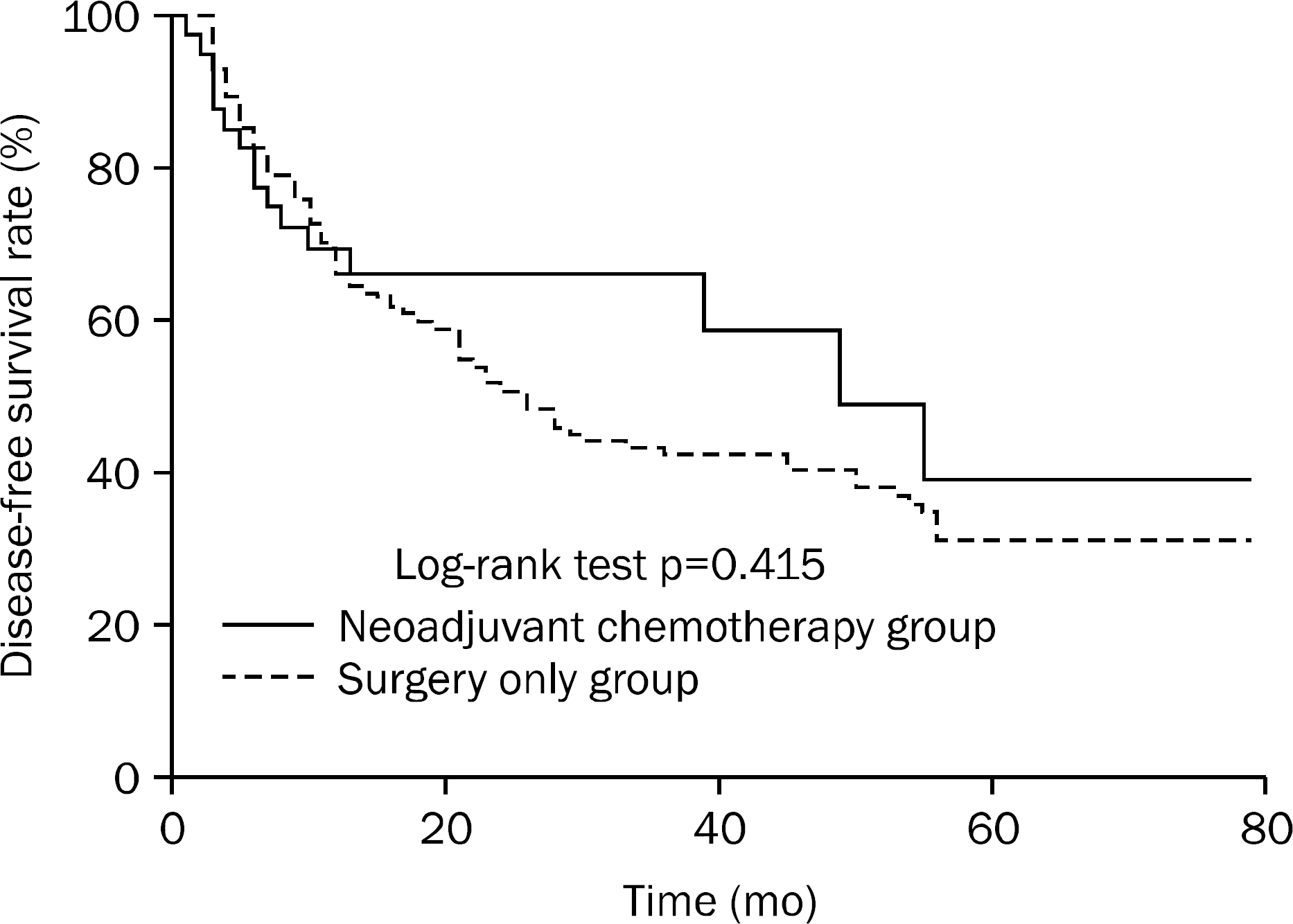
The baseline characteristics did not differ significantly between groups. After NAC, the tumor downstage rate was 51.2% (21/41); however, overall survival (p=0.205) and disease-free survival (p=0.415) were not significantly different between the groups. On subgroup analysis, no significant differences in drug toxicity (p=0.604) or clinical response (p=0.374) were found between outcomes of doublet and triplet chemotherapy regimens.
Conclusions
In patients with locally advanced gastric cancer, NAC showed tolerable drug toxicity and increased tumor downstage, but NAC failed to increase the survival rate, which may be caused by a high D2-lymphadenectomy rate. Therefore, NAC was found to be a therapeutic option for select gastric cancer patients.
Go to : 
References
1. Chung MW, Jeong O, Park YK, et al. Comparison on the long term outcome between endoscopic submucosal dissection and surgical treatment for undifferentiated early gastric cancer. Korean J Gastroenterol. 2014; 63:90–98.

2. Sakuramoto S, Sasako M, Yamaguchi T, et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med. 2007; 357:1810–1820.

3. Kim YH. Chemotherapy for advanced gastric cancer: slow but further progress. Cancer Res Treat. 2005; 37:79–86.

4. Nashimoto A, Nakajima T, Furukawa H, et al. Randomized trial of adjuvant chemotherapy with mitomycin, Fluorouracil, and Cytosine arabinoside followed by oral Fluorouracil in serosa-negative gastric cancer: Japan Clinical Oncology Group 9206–1. J Clin Oncol. 2003; 21:2282–2287.

5. Ronellenfitsch U, Schwarzbach M, Hofheinz R, et al. Preoperative chemo(radio)therapy versus primary surgery for gastroesophageal adenocarcinoma: systematic review with metaanalysis combining individual patient and aggregate data. Eur J Cancer. 2013; 49:3149–3158.

6. Ajani JA, Komaki R, Putnam JB, et al. A three-step strategy of induction chemotherapy then chemoradiation followed by surgery in patients with potentially resectable carcinoma of the esophagus or gastroesophageal junction. Cancer. 2001; 92:279–286.

7. Ross P, Nicolson M, Cunningham D, et al. Prospective randomized trial comparing mitomycin, cisplatin, and protracted venous-infusion fluorouracil (PVI 5-FU) With epirubicin, cisplatin, and PVI 5-FU in advanced esophagogastric cancer. J Clin Oncol. 2002; 20:1996–2004.

8. Li W, Qin J, Sun YH, Liu TS. Neoadjuvant chemotherapy for advanced gastric cancer: a metaanalysis. World J Gastroenterol. 2010; 16:5621–5628.

9. Bang YJ, Kim YW, Yang HK, et al. Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): a phase 3 open-label, randomised controlled trial. Lancet. 2012; 379:315–321.
10. Wang LB, Shen JG, Xu CY, Chen WJ, Song XY, Yuan XM. Neoadjuvant chemotherapy versus surgery alone for locally advanced gastric cancer: a retrospective comparative study. Hepatogastroenterology. 2008; 55:1895–1898.
11. Sumpter K, Harper-Wynne C, Cunningham D, et al. Report of two protocol planned interim analyses in a randomised multicentre phase III study comparing capecitabine with fluorouracil and oxaliplatin with cisplatin in patients with advanced oesophagogastric cancer receiving ECF. Br J Cancer. 2005; 92:1976–1983.

12. Roukos DH, Kappas AM. Perspectives in the treatment of gastric cancer. Nat Clin Pract Oncol. 2005; 2:98–107.

13. Shimizu K, Ito K, Matsunaga N, Shimizu A, Kawakami Y. Diagnosis of gastric cancer with MDCT using the water-filling method and multiplanar reconstruction: CT-histologic correlation. AJR Am J Roentgenol. 2005; 185:1152–1158.

14. Chen CY, Hsu JS, Wu DC, et al. Gastric cancer: preoperative local staging with 3D multidetector row CT–correlation with surgical and histopathologic results. Radiology. 2007; 242:472–482.

15. Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006; 355:11–20.

16. Schuhmacher C, Gretschel S, Lordick F, et al. Neoadjuvant chemotherapy compared with surgery alone for locally advanced cancer of the stomach and cardia: European Organisation for Research and Treatment of Cancer randomized trial 40954. J Clin Oncol. 2010; 28:5210–5218.

17. Ge L, Wang HJ, Yin D, et al. Effectiveness of 5-flurouracil-based neoadjuvant chemotherapy in locally-advanced gastric/gastroesophageal cancer: a metaanalysis. World J Gastroenterol. 2012; 18:7384–7393.

18. Hartgrink HH, van de Velde CJ, Putter H, et al. Neoadjuvant chemotherapy for operable gastric cancer: long term results of the Dutch randomised FAMTX trial. Eur J Surg Oncol. 2004; 30:643–649.

19. Kang YK, Ryu MH, Yoo C, et al. Phase I/II study of a combination of docetaxel, capecitabine, and cisplatin (DXP) as first-line chemotherapy in patients with advanced gastric cancer. Cancer Chemother Pharmacol. 2011; 67:1435–1443.

20. Menges M, Schmidt C, Lindemann W, et al. Low toxic neoadjuvant cisplatin, 5-fluorouracil and folinic acid in locally advanced gastric cancer yields high R-0 resection rate. J Cancer Res Clin Oncol. 2003; 129:423–429.

21. Park I, Ryu MH, Choi YH, et al. A phase II study of neoadjuvant docetaxel, oxaliplatin, and S-1 (DOS) chemotherapy followed by surgery and adjuvant S-1 chemotherapy in potentially resectable gastric or gastroesophageal junction adenocarcinoma. Cancer Chemother Pharmacol. 2013; 72:815–823.

22. Fink U, Schuhmacher C, Stein HJ, et al. Preoperative chemotherapy for stage III-IV gastric carcinoma: feasibility, response and outcome after complete resection. Br J Surg. 1995; 82:1248–1252.

23. Van Cutsem E, Moiseyenko VM, Tjulandin S, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol. 2006; 24:4991–4997.

24. Cunningham D, Starling N, Rao S, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med. 2008; 358:36–46.

25. Valverde CM, Macarulla T, Casado E, Ramos FJ, Martinelli E, Tabernero J. Novel targets in gastric and esophageal cancer. Crit Rev Oncol Hematol. 2006; 59:128–138.

26. Shah MA, Ramanathan RK, Ilson DH, et al. Multicenter phase II study of irinotecan, cisplatin, and bevacizumab in patients with metastatic gastric or gastroesophageal junction adenocarcinoma. J Clin Oncol. 2006; 24:5201–5206.

Go to : 
Table 1.
Patiens Baseline Characteristics
Table 2.
Grade 3 or 4 Toxicity and Clinical Response Assessment during Neoadjuvant Chemotherapy
Table 3.
Surgical and Pathologic Results
Table 4.
Stage Change of the Pretreatment vs. Post Neoadjuvant Chemotherapy
Table 5.
Pattern of Recurrence, Survival Status, and Cause of Death




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download