Abstract
Background/Aims
The prevalence of peptic ulcer disease has not decreased mainly due to an increase in the use of NSAIDs. This study was conducted in order to determine whether a chronic NSAID-induced gastric inflammation model could be established by repeated administration of NSAID.
Methods
Indomethacin (10 mg/kg) was administered once per week for six weeks in 8- and 26-week rats and animals were sacrificed every week after administration. Gross ulcer index, histologic damage index, myeloperoxidase (MPO) activity, and mucus (glucosamine) levels were measured. Small bowel damage was also evaluated.
Results
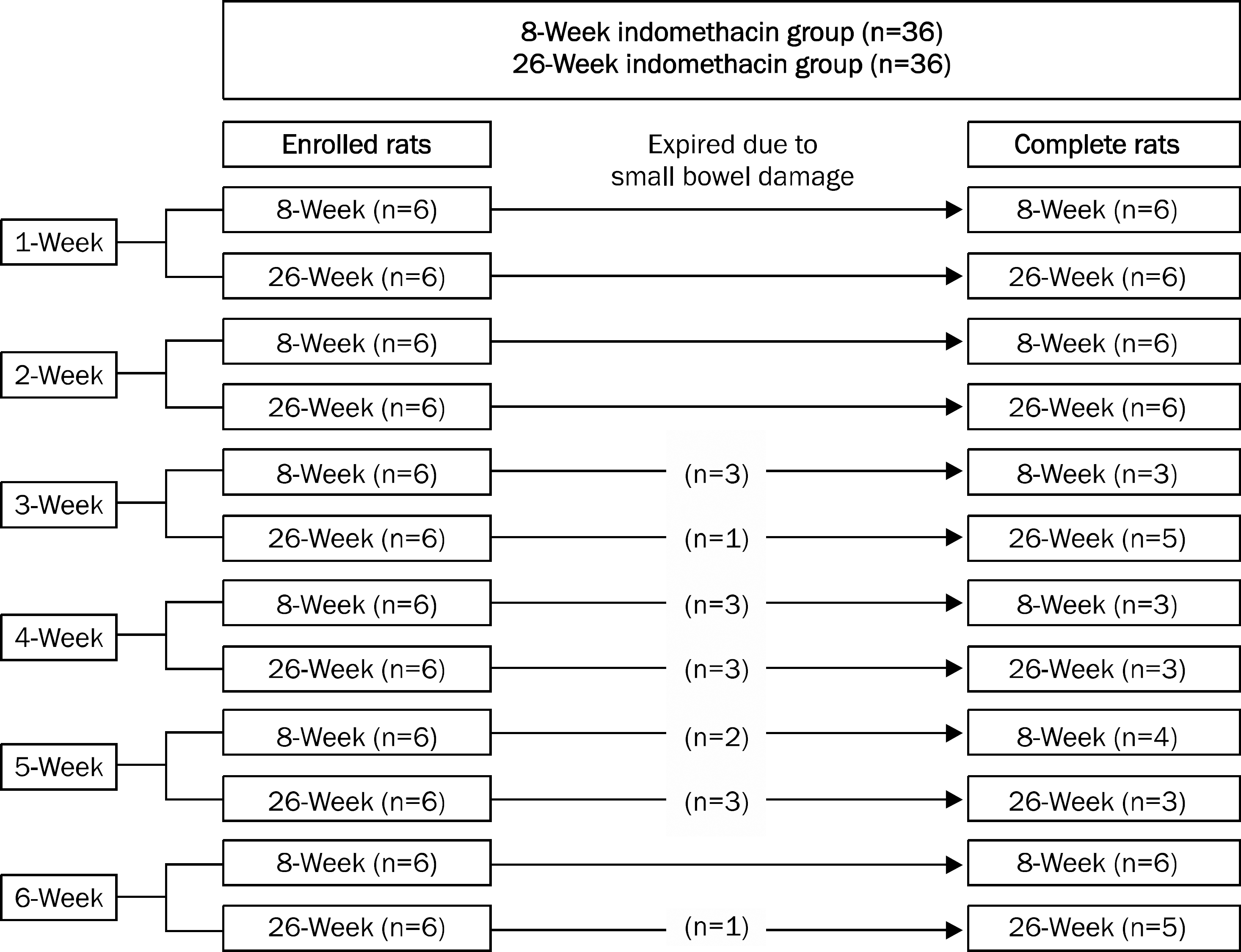
Gross gastric damage index showed a peak level at three weeks and then decreased slowly in the 26-week indomethacin group. Gastric mucosal glucosamine level increased in both the 8-week (p=0.038) and 26-week groups (p=0.007). In addition, gastric mucosal MPO level decreased in the 8-week group (p=0.018) but did not show a decrease in the 26-week group. Small bowel damage began to occur at three weeks during the schedule and eight of 36 rats (22.2%) died due to perforation or peritonitis of the small bowel in the 8- and 26-week indomethacin groups, respectively.
Go to : 
References
1. Wong GL, Wong VW, Chan Y, et al. High incidence of mortality and recurrent bleeding in patients with Helicobacter pylori-negative idiopathic bleeding ulcers. Gastroenterology. 2009; 137:525–531.
2. Gisbert JP, Calvet X. Review article: Helicobacter pylori-negative duodenal ulcer disease. Aliment Pharmacol Ther. 2009; 30:791–815.
3. Sung JJ, Chan FK, Chen M, et al. Asia-Pacific Working Group. Asia-Pacific Working Group consensus on non-variceal upper gastrointestinal bleeding. Gut. 2011; 60:1170–1177.

4. Yang YX, Lewis JD, Epstein S, Metz DC. Longterm proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006; 296:2947–2953.

5. Okabe S, Pfeiffer CJ. Chronicity of acetic acid ulcer in the rat stomach. Am J Dig Dis. 1972; 17:619–629.

6. Kang JM, Kim N, Kim B, et al. Enhancement of gastric ulcer healing and angiogenesis by cochinchina Momordica seed extract in rats. J Korean Med Sci. 2010; 25:875–881.

7. Evangelista S. Role of sensory neurons in restitution and healing of gastric ulcers. Curr Pharm Des. 2006; 12:2977–2984.

8. Fiorucci S, Distrutti E, de Lima OM, et al. Relative contribution of acetylated cyclooxygenase (COX)-2 and 5-lipooxygenase (LOX) in regulating gastric mucosal integrity and adaptation to aspirin. FASEB J. 2003; 17:1171–1173.

9. Polat B, Suleyman H, Alp HH. Adaptation of rat gastric tissue against indomethacin toxicity. Chem Biol Interact. 2010; 186:82–89.

10. Nam SY, Kim N, Lee CS, et al. Gastric mucosal protection via enhancement of MUC5AC and MUC6 by geranylgeranylacetone. Dig Dis Sci. 2005; 50:2110–2120.

11. Lacy ER, Ito S. Microscopic analysis of ethanol damage to rat gastric mucosa after treatment with a prostaglandin. Gastroenterology. 1982; 83:619–625.

12. Bradley PP, Priebat DA, Christensen RD, Rothstein G. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J Invest Dermatol. 1982; 78:206–209.

13. Grisham MB, Benoit JN, Granger DN. Assessment of leukocyte involvement during ischemia and reperfusion of intestine. Methods Enzymol. 1990; 186:729–742.
14. Nishida K, Ohta Y, Ishiguro I. Teprenone, an anti-ulcer agent, increases gastric mucosal mucus level via nitric oxide in rats. Jpn J Pharmacol. 1998; 78:519–522.
15. Alderman BM, Cook GA, Familari M, Yeomans ND, Giraud AS. Resistance to apoptosis is a mechanism of adaptation of rat stomach to aspirin. Am J Physiol Gastrointest Liver Physiol. 2000; 278:G839–G846.

16. Konturek SJ, Brzozowski T, Stachura J, Dembinski A, Majka J. Role of gastric blood flow, neutrophil infiltration, and mucosal cell proliferation in gastric adaptation to aspirin in the rat. Gut. 1994; 35:1189–1196.

17. Konturek PC, Brzozowski T, Pierzchalski P, et al. Activation of genes for spasmolytic peptide, transforming growth factor alpha and for cyclooxygenase (COX)-1 and COX-2 during gastric adaptation to aspirin damage in rats. Aliment Pharmacol Ther. 1998; 12:767–777.

18. Kuroda M, Yoshida N, Ichikawa H, et al. Lansoprazole, a proton pump inhibitor, reduces the severity of indomethacin-induced rat enteritis. Int J Mol Med. 2006; 17:89–93.

19. Saud B, Nandi J, Ong G, Finocchiaro S, Levine RA. Inhibition of TNF-alpha improves indomethacin-induced enteropathy in rats by modulating iNOS expression. Dig Dis Sci. 2005; 50:1677–1683.
20. Stadnyk AW, Dollard C, Issekutz TB, Issekutz AC. Neutrophil migration into indomethacin induced rat small intestinal injury is CD11a/CD18 and CD11b/CD18 co-dependent. Gut. 2002; 50:629–635.

21. Ogihara Y, Okabe S. Mechanism by which indomethacin delays gastric ulcer healing in the rat: inhibited contraction of the ulcer base. Jpn J Pharmacol. 1993; 61:123–131.

22. Penney AG, Malcontenti-Wilson C, O'Brien PE, Andrews FJ. NSAID-induced delay in gastric ulcer healing is not associated with decreased epithelial cell proliferation in rats. Dig Dis Sci. 1995; 40:2684–2693.

23. Wang JY, Yamasaki S, Takeuchi K, Okabe S. Delayed healing of acetic acid-induced gastric ulcers in rats by indomethacin. Gastroenterology. 1989; 96:393–402.

Go to : 
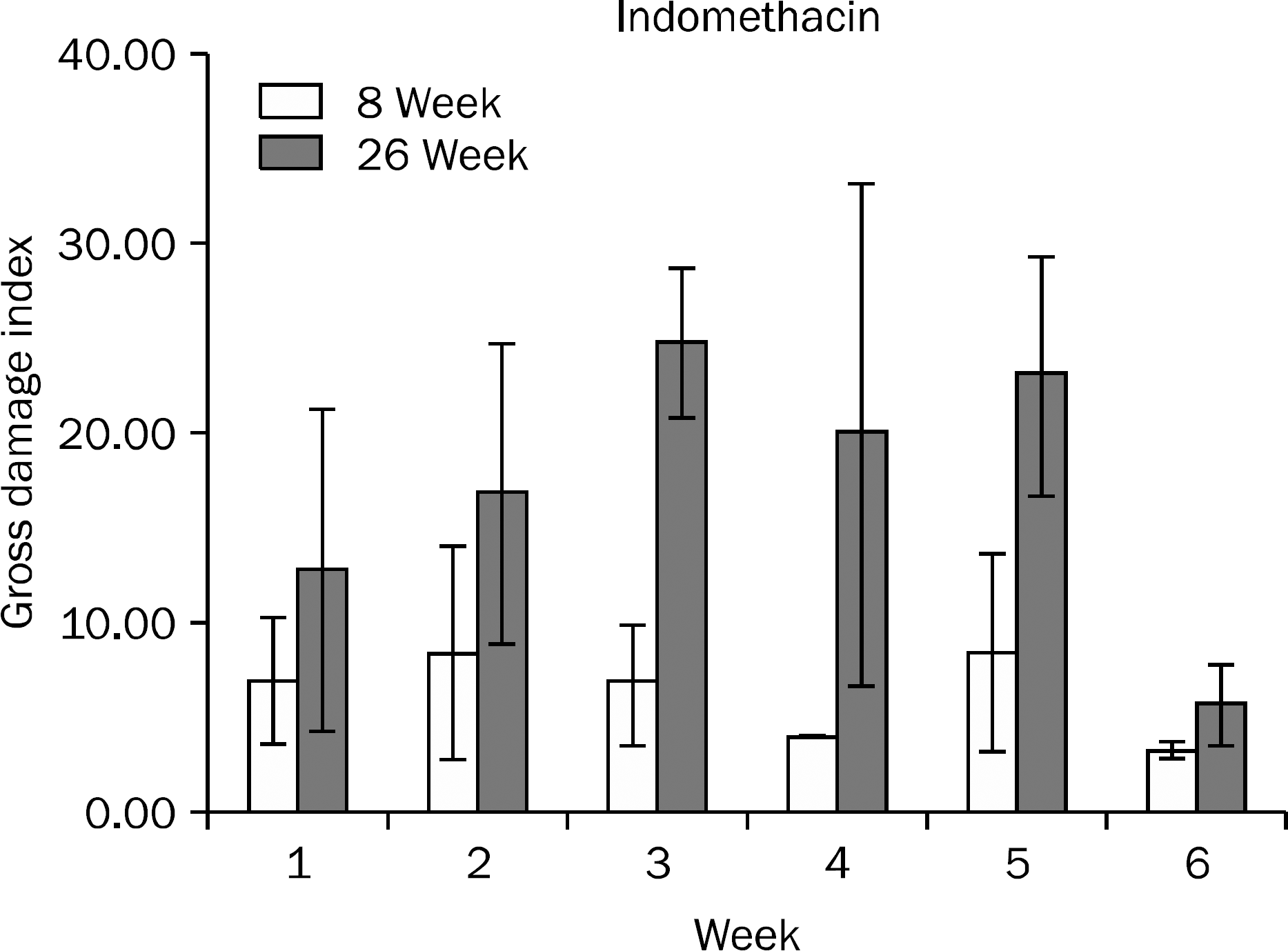 | Fig. 2.Gross gastric damage index during the study period in the 8-week and 26-week indomethacin groups. Data are shown with mean±standard error. |
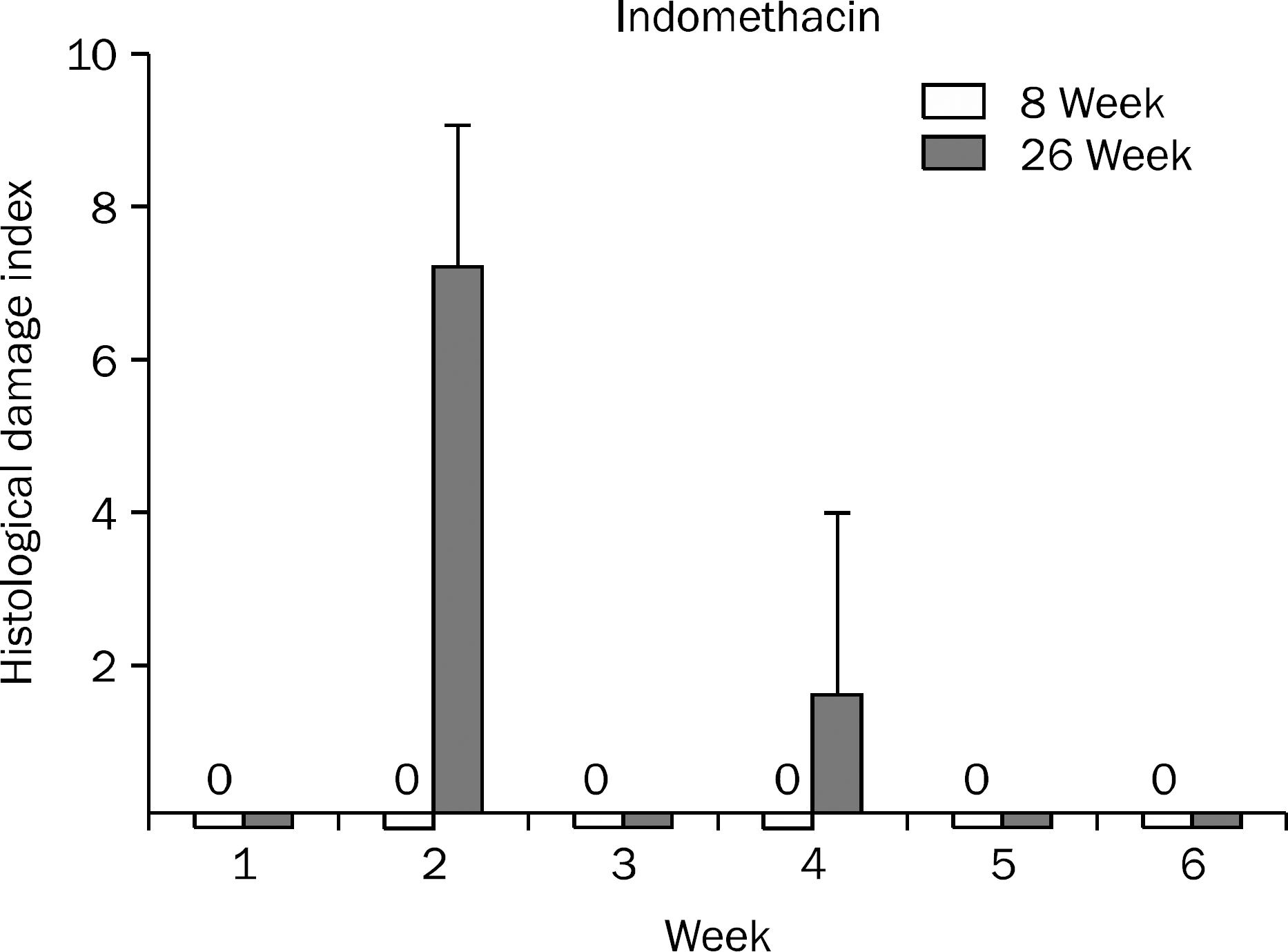 | Fig. 3.Histological index of gastric mucosal damage during the study period in the 8-week and 26-week indomethacin groups. Data are shown with mean±standard error. |
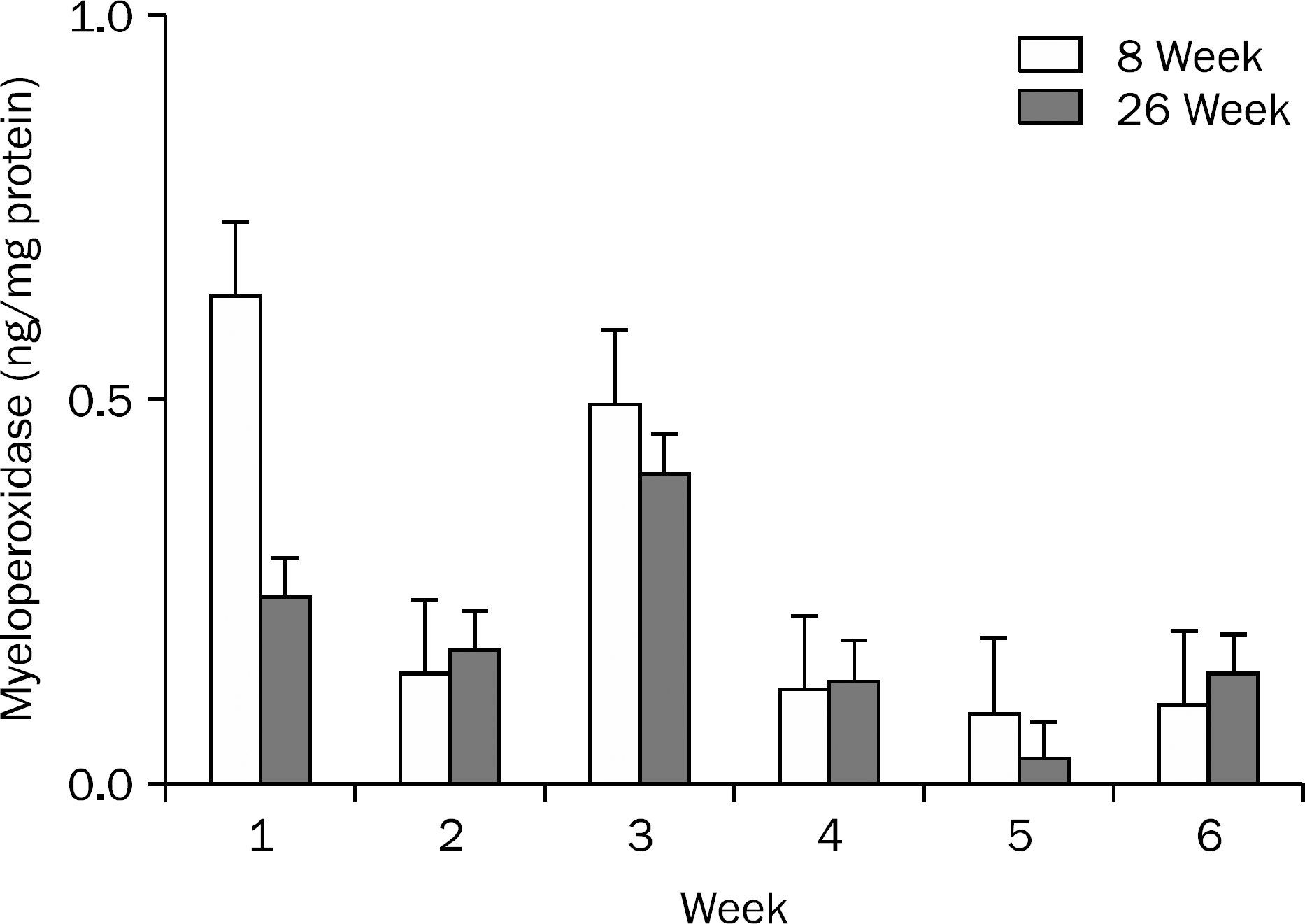 | Fig. 4.Myeloperoxidase levels in the small bowel during the study period in 8- and 26-week groups. No statistically significant trend was observed. Data are shown with mean±standard error. |
Table 1.
Occurrence of Small Bowel Damage in the Indomethacin Group during Six Weeks in 8- or 26-Week Rats
| Rats' age (week) | Perforation or peritonitis a | ||||||
|---|---|---|---|---|---|---|---|
| 1 Week | 2 Week | 3 Week | 4 Week | 5 Week | 6 Week | Total | |
| 8 | 0/6 | 2/6 | 2/3 | 2/3 | 1/4 | 0/6 | 7/28 (25.0%) |
| 16 | 0/6 | 0/6 | 2/5 | 2/3 | 0/3 | 0/5 | 4/28 (14.3%) |
Table 2.
Time Trend of Gastric Mucosal Myeloperoxidase (MPO) and Glucosamine Levels in the Indomethacin Groups
| 1 Week | 2 Week | 3 Week | 4 Week | 5 Week | 6 Week | p-value a | ||
|---|---|---|---|---|---|---|---|---|
| Indomethacin | MPO (ng/mg protein) | 0.68±0.19 | 0.54±0.09 | 0.64±0.12 | 0.26±0.03 | 0.31±0.06 | 0.22±0.05 | 0.018 |
| 8-week-rat group | Glucosamine (mg/g mucosa) | 8.36±1.09 | 8.18±0.59 | 8.01±1.15 | 9.31±1.35 | 9.59±2.10 | 12.24±0.91 | 0.038 |
| Indomethacin | MPO (ng/mg protein) | 0.51±0.07 | 0.72±0.11 | 0.71±0.07 | 0.36±0.06 | 0.31±0.02 | 0.48±0.11 | 0.270 |
| 26-week-rat group | Glucosamine (mg/g mucosa) | 7.74±1.35 | 6.49±1.70 | 10.54±1.31 | 11.19±1.07 | 11.73±2.53 | 14.06±1.95 | 0.007 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download