Abstract
Crohn's disease is characterized by chronic transmural inflammation of the bowel and is associated with serious complications, such as bowel strictures, abscesses, fistula formation, and perforation. As neither medical nor surgical therapy provides a cure for Crohn's disease, the primary goals of therapy are to induce and maintain remission and prevent complications. As a biologic agent, infliximab, a monoclonal antibody to tumor necrosis factor, is indicated for refractory luminal and fistulizing Crohn's disease that does not respond to other medical therapies or surgery. Infliximab has proven to be very effective for inducing and maintaining remission in Crohn's disease; however, infliximab treatment has several potential complications. Here, we report a case of free perforation following a therapeutic response after an initial dose of infliximab for Crohn's disease. This is the first case report describing a free perforation in a Crohn's disease patient after an initial dose of infliximab.
References
1. Hanauer SB, Sandborn W. Practice Parameters Committee of the American College of Gastroenterology. Management of Crohn's disease in adults. Am J Gastroenterol. 2001; 96:635–643.

2. Present DH, Rutgeerts P, Targan S, et al. Infliximab for the treatment of fistulas in patients with Crohn's disease. N Engl J Med. 1999; 340:1398–1405.

3. Rutgeerts P, Van Assche G, Vermeire S. Optimizing anti-TNF treatment in inflammatory bowel disease. Gastroenterology. 2004; 126:1593–1610.

4. de Vries HS, van Oijen MG, de Jong DJ. Serious events with infliximab in patients with inflammatory bowel disease: a 9-year cohort study in the Netherlands. Drug Saf. 2008; 31:1135–1144.
5. Fidder H, Schnitzler F, Ferrante M, et al. Longterm safety of infliximab for the treatment of inflammatory bowel disease: a single-centre cohort study. Gut. 2009; 58:501–508.

6. Eshuis EJ, Griffioen GH, Stokkers PC, Ubbink DT, Bemelman WA. Anti tumour necrosis factor as risk factor for free perforations in Crohn's disease? A casecontrol study. Colorectal Dis. 2012; 14:578–584.

7. Crohn BB. Indications for surgical intervention in regional ileitis. AMA Arch Surg. 1957; 74:305–311.

8. Crohn BB. Acute regional ileitis; clinical aspects and follow-up studies. N Y State J Med. 1965; 65:641–644.
9. Greenstein AJ, Mann D, Sachar DB, Aufses AH Jr. Free perforation in Crohn's disease: I. A survey of 99 cases. Am J Gastroenterol. 1985; 80:682–689.
10. Greenstein AJ, Sachar DB, Mann D, Lachman P, Heimann T, Aufses AH Jr. Spontaneous free perforation and perforated abscess in 30 patients with Crohn's disease. Ann Surg. 1987; 205:72–76.

11. Katz S, Schulman N, Levin L. Free perforation in Crohn's disease: a report of 33 cases and review of literature. Am J Gastroenterol. 1986; 81:38–43.
12. Hanauer SB, Feagan BG, Lichtenstein GR, et al. ACCENT I Study Group. Maintenance infliximab for Crohn's disease: the ACCENT I randomised trial. Lancet. 2002; 359:1541–1549.

13. Sleisenger MH, Feldman M, Friedman LS, Brandt LJ. Sleisenger and Fordtran's gastrointestinal and liver disease. 9th ed.Philadelphia: Saunders;2010. p. 1967–1969.
14. Colombel JF, Loftus EV Jr, Tremaine WJ, et al. The safety profile of infliximab in patients with Crohn's disease: the Mayo clinic experience in 500 patients. Gastroenterology. 2004; 126:19–31.

15. Kronfol Z, Remick DG. Cytokines and the brain: implications for clinical psychiatry. Am J Psychiatry. 2000; 157:683–694.

16. Reichenberg A, Yirmiya R, Schuld A, et al. Cytokine-associated emotional and cognitive disturbances in humans. Arch Gen Psychiatry. 2001; 58:445–452.

17. van Balkom BP, Schoon EJ, Stockbrügger RW, et al. Effects of anti-tumour necrosis factoralpha therapy on the quality of life in Crohn's disease. Aliment Pharmacol Ther. 2002; 16:1101–1107.
Fig. 1.
Colonoscopic findings. (A) Cobblestone appearance in the ascending colon consistent with Crohn's disease. (B) Longitudinal ulcers and luminal narrowing in the terminal ileum consistent with Crohn's disease.
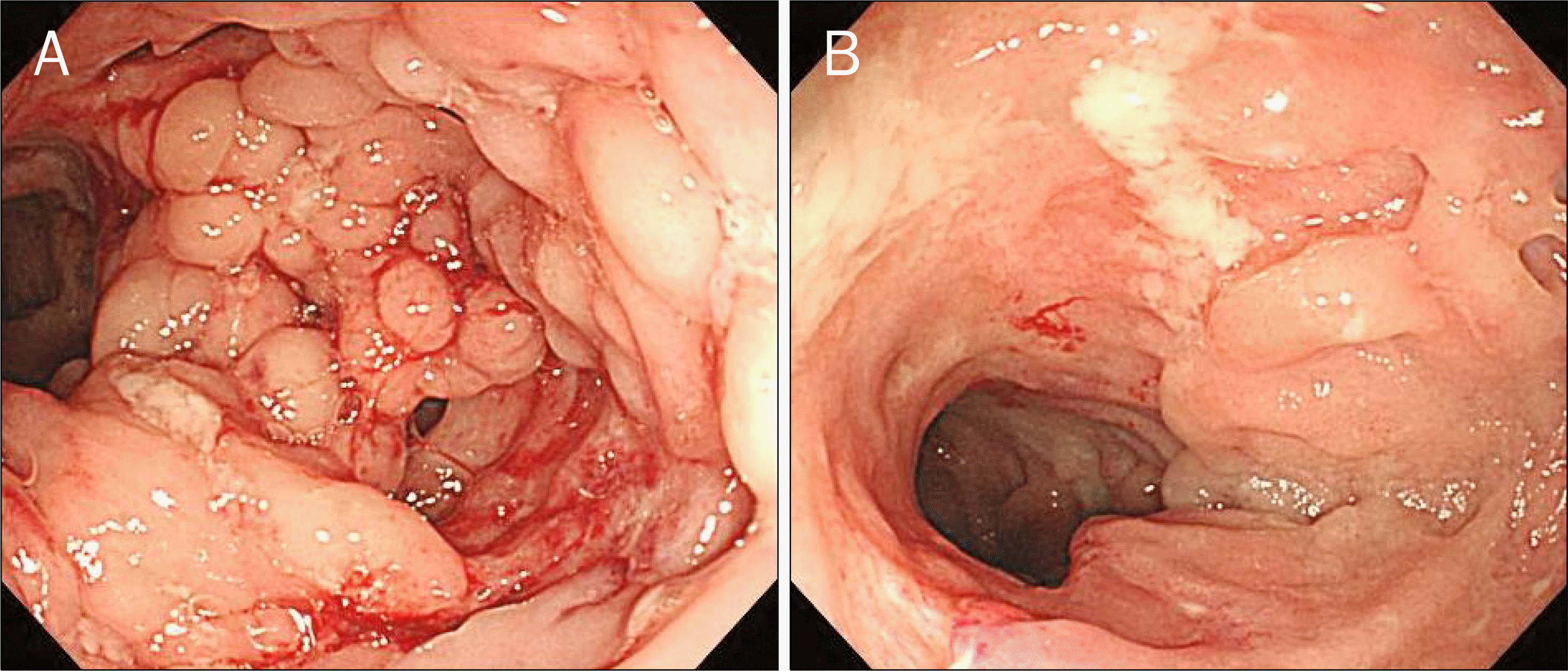
Fig. 2.
A small bowel follow-through at diagnosis demonstrating multiple strictures and pseudosacculations (arrowhead, stricture; arrow, pseudosacculation).
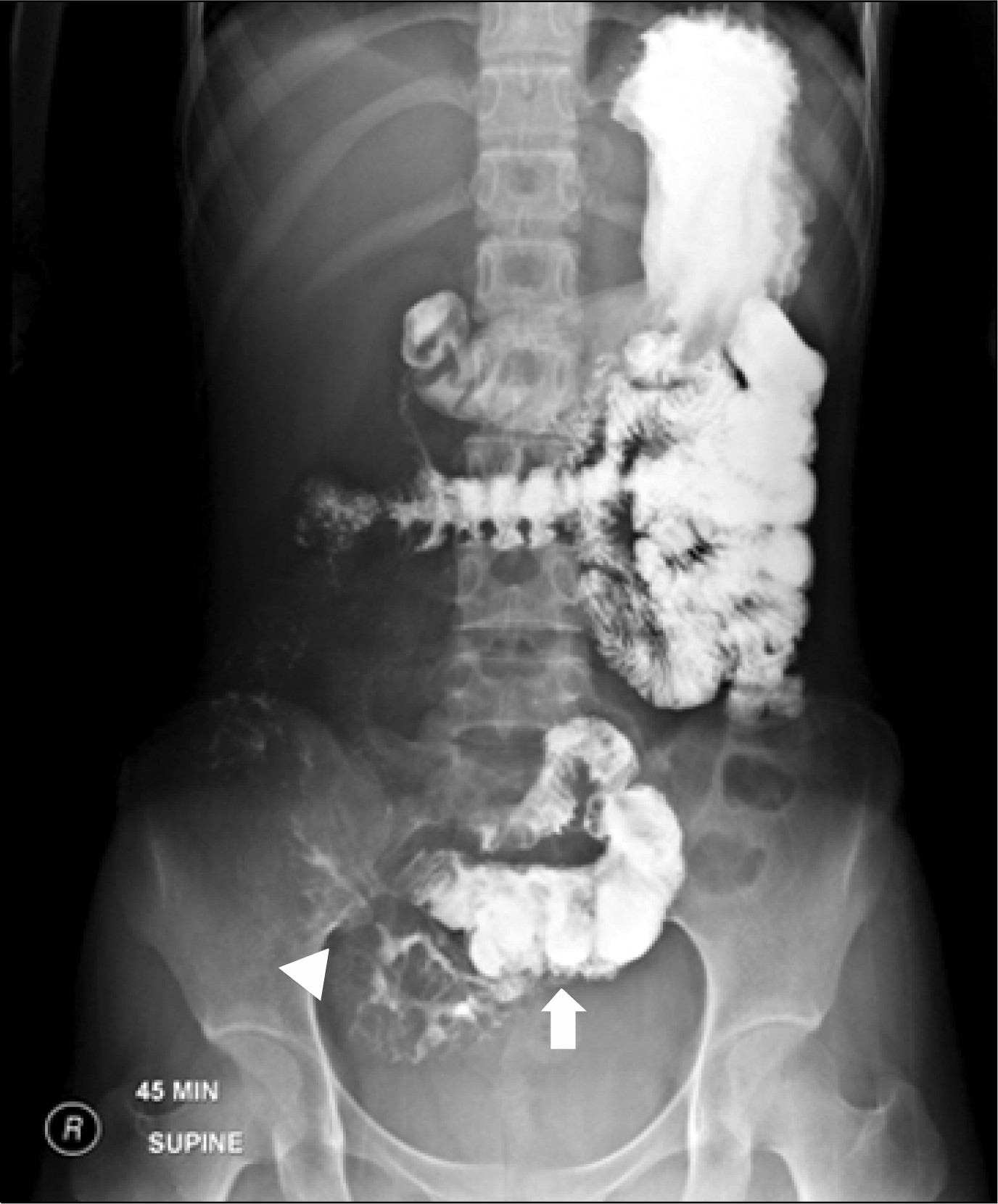
Fig. 3.
Abdominal CT at the time of the emergency room presentation demonstrating a focal, asymmetric wall defect in the distal ileum with extraluminal free fecal density in the peritoneal space. A stricture is noted in the distal part of the perforation lesion. The wall is relatively thin at the level of the distal ileum perforation (arrow, wall defect of the distal ileum; arrowheads, extraluminal free fecal density in the peritoneal space; open arrows, stricture).
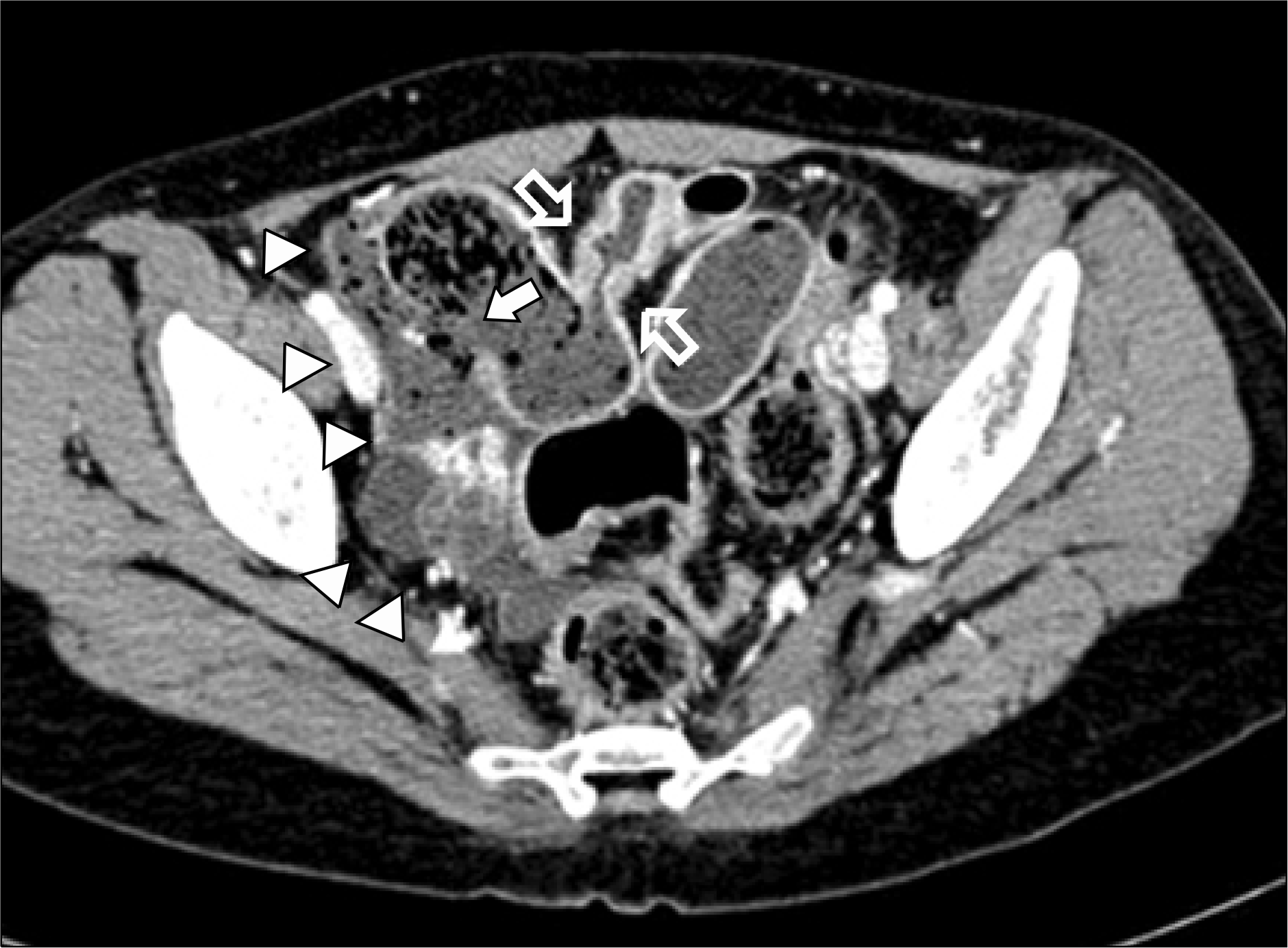
Fig. 4.
Gross findings of the surgical specimen revealing a perforated lesion with a relatively thin ileal wall (arrows).
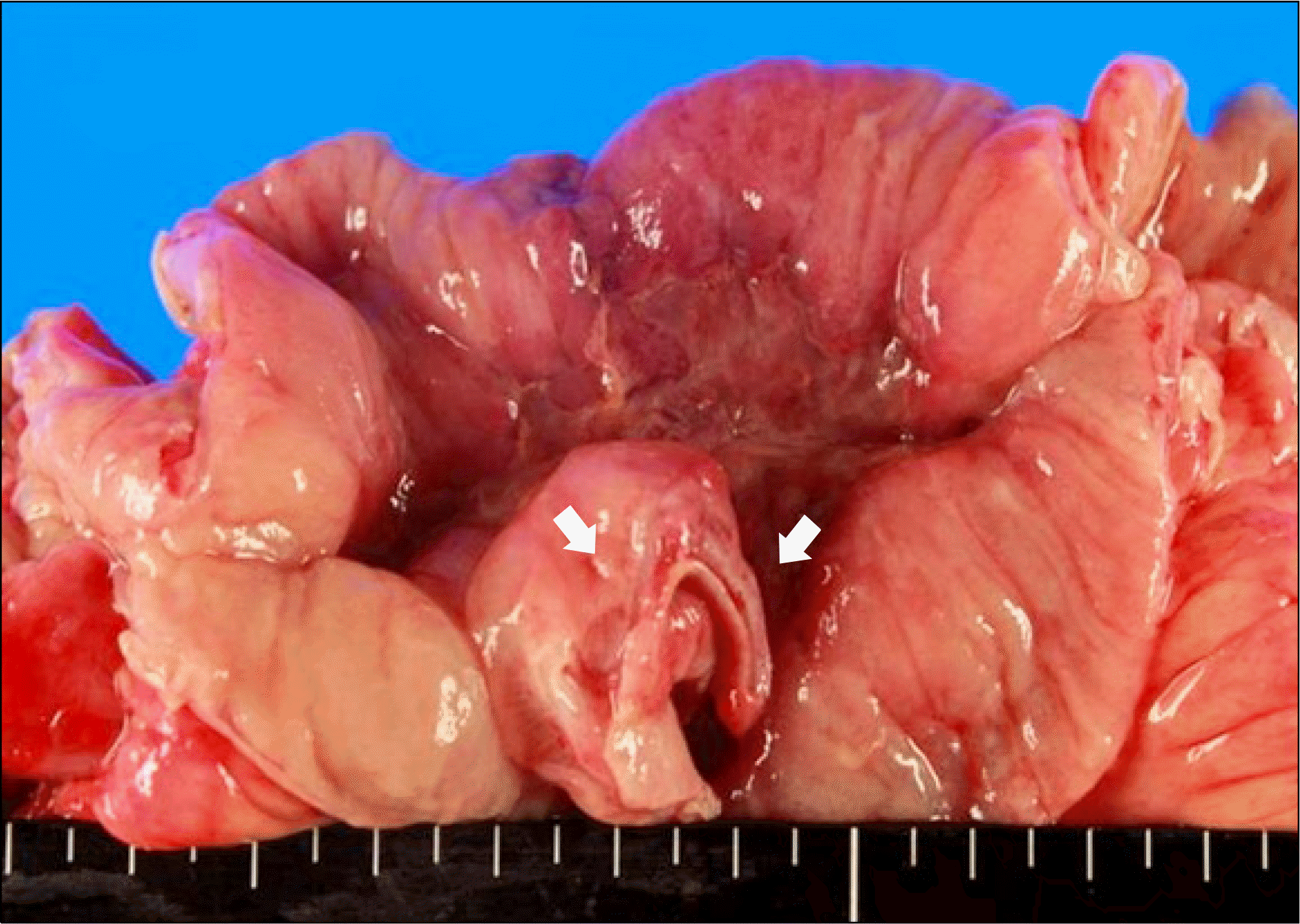
Fig. 5.
Microscopic findings of the surgical specimen (H&E). (A) At the level of the perforation, only mild infiltration of inflammatory cells and fibrosis and a consequently thinned intestinal wall is noted (×40; arrows, perforated margin). (B) Noncaseating epithelioid granulomas are noted (×200; arrows, non-caseating epithelioid granulomas).





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download