Abstract
We report herein a case of intestinal amyloidosis with grave prognosis that caused intractable diarrhea and intestinal pseudo-obstruction, alternately in spite of intensive conservative treatment. A 44-year-old woman was admitted for fever, diarrhea, and crampy abdominal pain which had been continuned during 6 months. Abdomen CT scan showed edematous wall thickening of the small bowel and right colon, and colonoscopic biopsy revealed amyloid deposition in the mucosa. Monoclonal light chains in serum and/or urine were not detected and highly elevated serum amyloid A was shown. In spite of intensive treatment including oral prednisolone and colchicine, diarrhea and intestinal pseudo-obstruction developed alternately, general status rapidly got worsened and died after two months.
Amyloidosis is a rare disease characterized by forming pathological protein deposits (i.e., amyloid) in several different organs and tissues.1 Idiopathic isolated AA amyloidosis affecting the small or large intestine is a very rare condition.2 In this report, we present a case of intestinal amyloidosis with grave prognosis that caused intractable diarrhea and intestinal pseudo-obstruction, alternately in spite of intensive conservative treatment.
A 44-year-old woman was admitted to our hospital with a 6-month history of fever, diarrhea, and crampy abdominal pain. On admission, physical examinations were unremarkable except for decreased skin turgor and a body temperature of 39℃. Her body height and weight were 147 cm and 38 kg.
Laboratory studies showed that hematocrit, 29.8% (normal range, 37-52%); white blood cell count, 3,500/mm3 (normal range, 4,800-10,800/mm3); potassium, 2.6 mEq/L (normal range, 3.5-5 mEq/L); total protein, 5.4 g/dL (normal range, 6.4-8.3 g/dL); albumin, 2.8 g/dL (normal range, 3.5-5.2 g/dL); CRP 9.24 mg/dL (normal range, 0.1-1 mg/dL). Other biochemical data were normal. Stool cultures for Salmonella, Shigella, and Campylobacter species were negative. Toxin A for Clostridium difficile was negative in the stool. Tuberculosis screening tests including tuberculin skin test and interferon gamma release assays were negative. Venereal disease research laboratory test was nonreactive and serology for HIV was negative. No positive results were found for any autoantibodies including rheumatoid factor, anti-nuclear antibody, and anti-neutrophil cytoplasmic antibodies. CH50 activity and concentrations of C3 and C4 were all within normal limits. Urinalysis showed proteinuria with protein excretion of 0.72 g/day. Abdomen CT scan showed edematous wall thickening of the small bowel and right colon (Fig. 1). Colonoscopy showed diffusely distributed petechial mucosal suggillations in the ascending colon and shallow erosions in the terminal ileum (Fig. 2). Histology of the biopsied specimens from the terminal ileum and right colon demonstrated amyloid deposition in the lamina propria of the ileal and colonic mucosa (Fig. 3). Other laboratory data showed negative results in the detection of monoclonal light chains in serum and/or urine. Echocardiography revealed normal cardiac function. Subsequent serum amyloid A (SAA) was 361 µg/mL (normal range 0-8 µg/mL). Initial H2 concentration was over 0.02 mL in the lactulose breath test. Soon after starting antibiotics (ciprofloxacin and metronidazole/rifaximin), the high fever disappeared. Despite treatment with oral prednisolone (60 mg/day) and colchicines (1.2 mg/day), the watery diarrhea and intestinal pseudo-obstruction developed alternately. Her general status rapidly got worsened in parallel with the aggravation of metabolic acidosis and renal dysfunction. She died from intractable diarrhea, intermittent intestinal pseudo-obstruction and renal dysfunction after two months.
Amyloidosis is defined as an extracellular deposit of protein fibrils with a β-sheet fibrillar structure and characteristic properties after staining with Congo red dye.3-5 There are 6 types: primary (AL), secondary (AA), hemodialysis-related, hereditary, senile, and localized.6 AA amyloidosis with acutephase reactant SAA protein (A) is associated with infectious, inflammatory, or, less commonly, neoplastic disorders. This case showed neither physical findings nor laboratory data suggestive of these causative disorders. According to a recent report, no underlying disease was found in nearly 5% of patients with AA amyloidosis.7 In this case, neither chronic inflammatory disease nor malignant tumors were identified despite an intensive systemic evaluation, the AA amyloidosis was regarded as idiopathic.
Gastrointestinal amyloidosis usually shows various clinical manifestations, including mucosal erosions and ulceration, malabsorption, hemorrhages, protein losing enteropathy and intractable diarrhea irrespective of the different precursor proteins of amyloid, and is sometimes the direct cause of death.8
There were 8 cases of gastrointestinal amyloidosis in our hospital for the past 10 years (Table 1). Mean age of these cases was 59 (range 44-74) years and females were dominant (M:F=2:6). The most common site was the rectum. The survival duration of these patients except two alive patients with AA type was 2-27 months. AA amyloidosis with definite cause had a favorable outcome.
Treatment of AA amyloidosis includes control of the primary disease. However, in this case, causeative disorder was not identified despite an intensive systemic survey. To suppress the production of SAA, which was the precursor protein of this disease, and to reduce submucosal edema and inflammation in the gastrointestinal tract, corticosteroid was introduced for treatment of the amyloidosis.9 Also, colchicine that was shown to have promising results in experimental cases of AA amyloidosis was used in the patient.10 But, they were not effective.
Feurle11 reported that decreased gastrointestinal motility in intestinal amyloidosis caused bacterial overgrowth, bile acid deconjugation and consequently, diarrhea, steatorrhea and severe malabsorption in the intestinal amyloidosis. In this case, a lactulose breath test showed overt bacterial overgrowth. However, the patient was not responsive to antibiotic treatment including metronidazole and rifaximin. Pseudo-obstruction due to amyloidosis can involve the small bowel, colon, or both, and carries a particularly grave prognosis.12 It typically presents as a mechanical obstruction with plain films showing a paralytic ileus. In our case, she developed diarrhea and intestinal pseudo-obstruction, alternately. Therefore, we could not use a long-acting somatostatin analogue, octreotide, which has been used in the treatment of severe diarrhea ascribable to amyloidosis. She had a poor prognosis because of intractable diarrhea and intermittent intestinal pseudo-obstruction.
The clinical manifestations of amyloid colonic deposition may mimic other diseases, such as inflammatory bowel disease, malignancy, ischemic colitis and collagenous colitis.13 Also, the diagnosis of amyloidosis must be supported by pathologic finding as it has no pathognomic radiologic or endoscopic findings (Fig. 4).14-19 In reality though, there is a major risk of misunderstanding and diagnostic delay. Therefore, gastrointestinal amyloidosis should be considered among differential diagnoses of chronic diarrhea unresponsive or resistant to conventional treatment.
We have presented herein a case of intestinal amyloidosis without extraintestinal manifestation that caused intractable diarrhea and intestinal pseudo-obstruction, alternately.
Figures and Tables
Fig. 1
Abdominal CT finding. Abdomen CT scan showed edematous wall thickening and mural enhancement of the pelvic small bowel loop and increased vascularity of the corresponding mesentery, mild edematous wall thickening of the colonic loop.
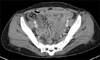
Fig. 2
Colonoscopic findings. Colonoscopy showed diffusely distributed petechial mucosal suggillations in the ascending colon (A) and shallow erosions in the terminal ileum (B).
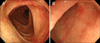
Fig. 3
Microscopic findings. (A) Eosinophilic proteineous material deposition was identified in the lamina propria of the colonic mucosa (H&E, ×100). (B) This material resulted in a positive reaction using the Congo red stain (×200).

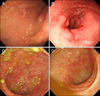
Fig. 4
Endoscopic findings. Endoscopic examinations revealed various findings such as diffuse geographic ulcerations (A), small healing ulcers or erosions (B), yellowish exudate coated hyperemic mucosal lesions (C) and mucosal and submucosal hemorrhagic spots (D).
References
1. Kala Z, Válek V, Kysela P. Amyloidosis of the small intestine. Eur J Radiol. 2007. 63:105–109.
2. Fonnesu C, Giovinale M, Verrecchia E, et al. Gastrointestinal amyloidosis: a case of chronic diarrhoea. Eur Rev Med Pharmacol Sci. 2009. 13:Suppl 1. 45–50.
3. Westermark P. Aspects on human amyloid forms and their fibril polypeptides. FEBS J. 2005. 272:5942–5949.
4. Levine RA. Amyloid disease of the liver. Correlation of clinical, functional and morphologic features in forty-seven patients. Am J Med. 1962. 33:349–357.
5. Naiki H, Nagai Y. Molecular pathogenesis of protein misfolding diseases: pathological molecular environments versus quality control systems against misfolded proteins. J Biochem. 2009. 146:751–756.
6. Ebert EC, Nagar M. Gastrointestinal manifestations of amyloidosis. Am J Gastroenterol. 2008. 103:776–787.
7. Röcken C, Shakespeare A. Pathology, diagnosis and pathogenesis of AA amyloidosis. Virchows Arch. 2002. 440:111–122.
8. Okuda Y, Takasugi K, Oyama T, Oyama H, Nanba S, Miyamoto T. Intractable diarrhoea associated with secondary amyloidosis in rheumatoid arthritis. Ann Rheum Dis. 1997. 56:535–541.
9. Fushimi T, Takahashi Y, Kashima Y, et al. Severe protein losing enteropathy with intractable diarrhea due to systemic AA amyloidosis, successfully treated with corticosteroid and octreotide. Amyloid. 2005. 12:48–53.
10. Cathcart ES. Kelley WN, Harris ED, Ruddy S, Sledge CB, editors. Amyloidosis. Textbook of rheumatology. 1993. Philadelphia: W.B. Saunders Company;1413–1428.
11. Feurle GE. Pathophysiology of diarrhea in patients with familial amyloid neuropathy. Digestion. 1987. 36:13–17.
12. Hiramatsu K, Kaneko S, Shirota Y, et al. Gastrointestinal amyloidosis secondary to hypersensitivity vasculitis presenting with intestinal pseudoobstruction. Dig Dis Sci. 1998. 43:1824–1830.
13. Braunstein JM, Aman A, Warman J. Colonic amyloidosis. Clin Gastroenterol Hepatol. 2007. 5:A30.
14. Yamada M, Hatakeyama S, Tsukagoshi H. Gastrointestinal amyloid deposition in AL (primary or myeloma-associated) and AA (secondary) amyloidosis: diagnostic value of gastric biopsy. Hum Pathol. 1985. 16:1206–1211.
15. Legge DA, Carlson HC, Wollaeger EE. Roentgenologic appearance of systemic amyloidosis involving gastrointestinal tract. Am J Roentgenol Radium Ther Nucl Med. 1970. 110:406–412.
16. Kim SH, Han JK, Lee KH, et al. Abdominal amyloidosis: spectrum of radiological findings. Clin Radiol. 2003. 58:610–620.
17. Schroeder FM, Miller FJ Jr, Nelson JA, Rankin RS. Gastrointestinal angiographic findings in systemic amyloidosis. AJR Am J Roentgenol. 1978. 131:143–146.
18. Tada S, Iida M, Yao T, et al. Endoscopic features in amyloidosis of the small intestine: clinical and morphologic differences between chemical types of amyloid protein. Gastrointest Endosc. 1994. 40:45–50.
19. Michael H, Brandt LJ, Tanaka KE, Berkowitz D, Cardillo M, Weidenheim K. Congo-red negative colonic amyloid with scalloping of the valvulae conniventes. Gastrointest Endosc. 2001. 53:653–655.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download