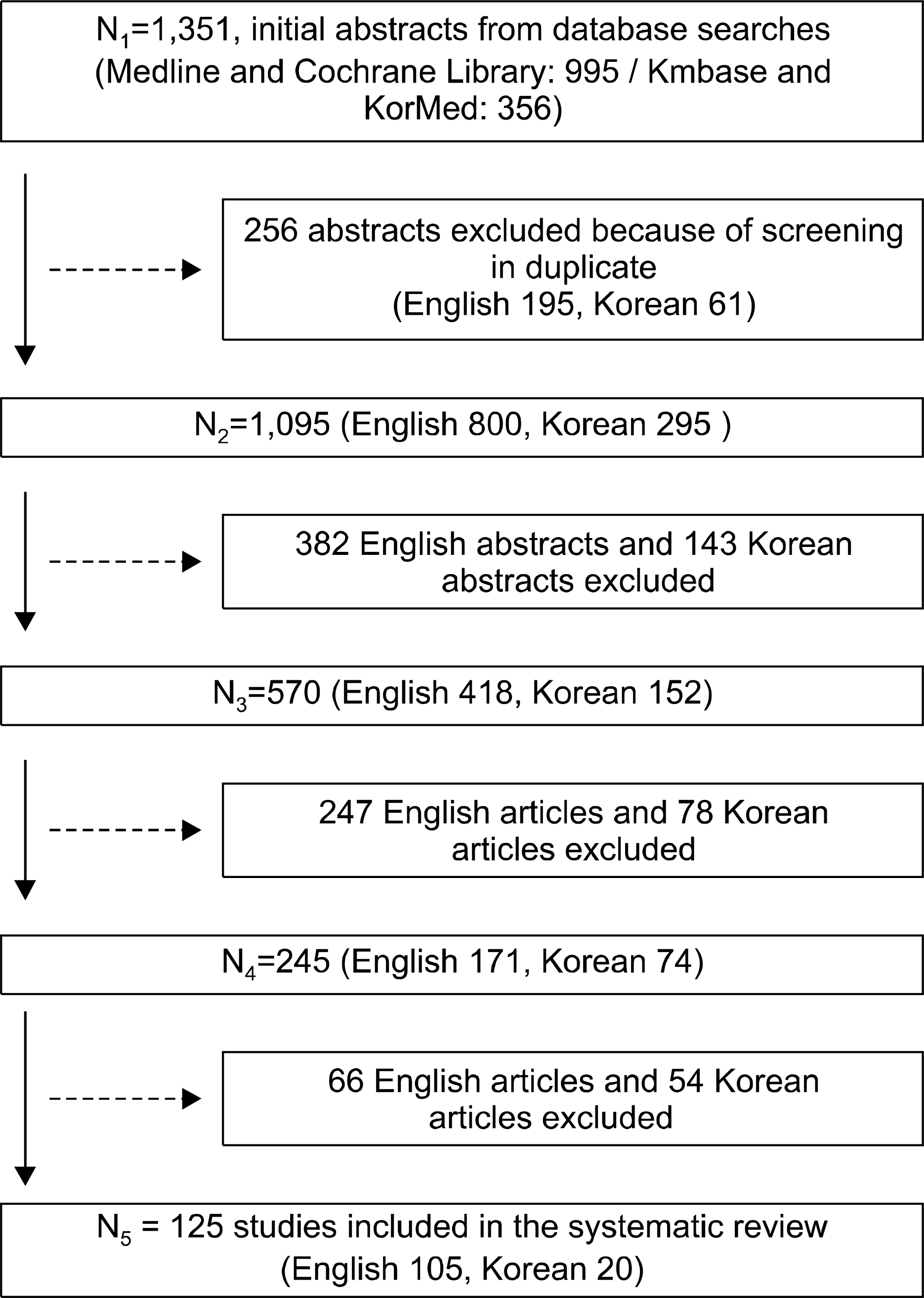
Abstract
While constipation is a common symptom in Korea, there are no existing treatment guidelines. Although constipation may occur as a result of organic cause, there is no obstructive mucosal or structural cause in the vast majority of patients with constipation. The present paper deals with only the management of functional constipation: lifestyle changes; bulking agents and stool softeners; osmotic agents; stimulant laxatives; prokinetics; biofeedback and surgical treatments. Exercise and dietary fiber are helpful in some patients with constipation. Laxatives including bulking agents, stool softeners, osmotic agents, and stimulant laxatives have been found to be more effective than placebo at relieving symptoms of constipation. New enterokinetic agents that affect peristalsis through selective interaction with 5-hydroxytryptamine-4 receptors can be effective in patients with constipation who cannot get adequate relief from current laxatives. Biofeedback can relieve symptoms in selected patients with pelvic floor dyssynergia. Surgical treatments can be helpful in some patients with refractory constipation.
Go to : 
References
1. Talley NJ, Weaver AL, Zinsmeister AR, Melton LJ 3rd. Functional constipation and outlet delay: a population-based study. Gastroenterology. 1993; 105:781–790.

2. Drossman DA, Li Z, Andruzzi E, et al. U.S. householder survey of functional gastrointestinal disorders. Prevalence, socio-demography, and health impact. Dig Dis Sci. 1993; 38:1569–1580.
3. Johanson JF, Sonnenberg A, Koch TR. Clinical epidemiology of chronic constipation. J Clin Gastroenterol. 1989; 11:525–536.

4. Jun DW, Park HY, Lee OY, et al. A population-based study on bowel habits in a Korean community: prevalence of functional constipation and self-reported constipation. Dig Dis Sci. 2006; 51:1471–1477.

5. Park YS, Lee DH. Evidence based guideline for diagnosis and treatment: therapeutic guideline for constipation. Korean J Neurogastroenterol Motil. 2005; 11:51–57.
6. Myung SJ, Lee TH, Huh KC, Choi SC, Sohn CI. Diagnosis of constipation: a systematic review. Korean J Gastroenterol. 2010; 55:316–324.

7. Guyatt GH, Cook DJ, Jaeschke R, Pauker SG, Schünemann HJ. Grades of recommendation for antithrombotic agents: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008; 133(6 Suppl):123S–131S.
8. Hull C, Greco RS, Brooks DL. Alleviation of constipation in the elderly by dietary fiber supplementation. J Am Geriatr Soc. 1980; 28:410–414.

9. Sandman PO, Adolfsson R, Hallmans G, Nygren C, Nystrom L, Winblad B. Treatment of constipation with high-bran bread in long-term care of severely demented elderly patients. J Am Geriatr Soc. 1983; 31:289–293.

10. Brown WJ, Mishra G, Lee C, Bauman A. Leisure time physical activity in Australian women: relationship with well being and symptoms. Res Q Exerc Sport. 2000; 71:206–216.

11. Sullivan SN, Wong C, Heidenheim P. Does running cause gastrointestinal symptoms? A survey of 93 randomly selected run-ners compared with controls. N Z Med J. 1994; 107:328–331.
12. Coenen C, Wegener M, Wedmann B, Schmidt G, Hoffmann S. Does physical exercise influence bowel transit time in healthy young men? Am J Gastroenterol. 1992; 87:292–295.
13. Meshkinpour H, Kemp C, Fairshter R. Effect of aerobic exercise on mouth-to-cecum transit time. Gastroenterology. 1989; 96:938–941.

14. Soffer EE, Summers RW, Gisolfi C. Effect of exercise on intestinal motility and transit in trained athletes. Am J Physiol. 1991; 260:G698–702.

16. Bingham SA, Cummings JH. Effect of exercise and physical fitness on large intestinal function. Gastroenterology. 1989; 97:1389–1399.

17. Keeling WF, Harris A, Martin BJ. Orocecal transit during mild exercise in women. J Appl Physiol. 1990; 68:1350–1353.

18. Keeling WF, Martin BJ. Gastrointestinal transit during mild exercise. J Appl Physiol. 1987; 63:978–981.

19. Rao SS, Beaty J, Chamberlain M, Lambert PG, Gisolfi C. Effects of acute graded exercise on human colonic motility. Am J Physiol. 1999; 276:G1221–1226.

20. Tuteja AK, Talley NJ, Joos SK, Woehl JV, Hickam DH. Is constipation associated with decreased physical activity in normally active subjects? Am J Gastroenterol. 2005; 100:124–129.

21. Kinnunen O. Study of constipation in a geriatric hospital, day hospital, old people's home and at home. Aging (Milano). 1991; 3:161–170.
22. Donald IP, Smith RG, Cruikshank JG, Elton RA, Stoddart ME. A study of constipation in the elderly living at home. Gerontology. 1985; 31:112–118.

23. Meshkinpour H, Selod S, Movahedi H, Nami N, James N, Wilson A. Effects of regular exercise in management of chronic idiopathic constipation. Dig Dis Sci. 1998; 43:2379–2383.
24. Kim I, Cho YN. The effects of abdominal meridian massage on constipation in institutionalized psychiatric patients taking an-tipsychotic Drugs. J Korean Acad Adult Nurs. 2007; 19:809–818.
25. Jeon SY, Jung HM. The effects of abdominal meridian massage on constipation among CVA patients. J Korean Acad Nurs. 2005; 35:135–142.

26. Kim MA, Sakong JK, Kim EJ, Kim EH, Kim EH. Effect of ar-omatherapy massage for the relief of constipation in the elderly. J Korean Acad Nurs. 2005; 35:56–64.

28. de Carvalho EB, Vitolo MR, Gama CM, Lopez FA, Taddei JA, de Morais MB. Fiber intake, constipation, and overweight among adolescents living in Sao Paulo City. Nutrition. 2006; 22:744–749.

29. Preston DM, Lennard-Jones JE. Severe chronic constipation of young women: ‘idiopathic slow transit constipation’. Gut. 1986; 27:41–48.

30. Anderson AS. Dietary factors in the aetiology and treatment of constipation during pregnancy. Br J Obstet Gynaecol. 1986; 93:245–249.

31. Campbell AJ, Busby WJ, Horwath CC. Factors associated with constipation in a community based sample of people aged 70 years and over. J Epidemiol Community Health. 1993; 47:23–26.

32. Towers AL, Burgio KL, Locher JL, Merkel IS, Safaeian M, Wald A. Constipation in the elderly: influence of dietary, psychological, and physiological factors. J Am Geriatr Soc. 1994; 42:701–706.

33. Tramonte SM, Brand MB, Mulrow CD, Amato MG, O'Keefe ME, Ramirez G. The treatment of chronic constipation in adults. A systematic review. J Gen Intern Med. 1997; 12:15–24.

34. Müller-Lissner SA. Effect of wheat bran on weight of stool and gastrointestinal transit time: a meta analysis. Br Med J (Clin Res Ed). 1988; 296:615–617.
35. Müller-Lissner SA, Kamm MA, Scarpignato C, Wald A. Myths and misconceptions about chronic constipation. Am J Gastroenterol. 2005; 100:232–242.
36. Voderholzer WA, Schatke W, Mühldorfer BE, Klauser AG, Birkner B, Müller-Lissner SA. Clinical response to dietary fiber treatment of chronic constipation. Am J Gastroenterol. 1997; 92:95–98.
37. Han YH, Yon MY, Hyun TS. Effect of prune supplementation on dietary fiber intake and constipation relief. Korean J Community Nutr. 2008; 13:426–438.
38. Huh JW, Park YA, Sohn SK, et al. Effect of yogurt enriched wa-ter-soluble fiber on functional constipation. J Korean Soc Coloproctol. 2007; 23:312–320.

39. Kim JY, Kim OY, Yoo HJ, et al. Effects of fiber supplements on functional constipation. Korean J Nutr. 2006; 39:35–43.
40. Kim TI, Park SJ, Choi CH, Lee SK, Kim WH. Effect of ear mush-room (auricularia) on functional constipation. Korean J Gastroenterol. 2004; 44:34–41.
41. Fenn GC, Wilkinson PD, Lee CE, Akbar FA. A general practice study of the efficacy of Regulan in functional constipation. Br J Clin Pract. 1986; 40:192–197.
42. Ashraf W, Park F, Lof J, Quigley EM. Effects of psyllium therapy on stool characteristics, colon transit and anorectal function in chronic idiopathic constipation. Aliment Pharmacol Ther. 1995; 9:639–647.

43. Cheskin LJ, Kamal N, Crowell MD, Schuster MM, Whitehead WE. Mechanisms of constipation in older persons and effects of fiber compared with placebo. J Am Geriatr Soc. 1995; 43:666–669.

44. McRorie JW, Daggy BP, Morel JG, Diersing PS, Miner PB, Robinson M. Psyllium is superior to docusate sodium for treatment of chronic constipation. Aliment Pharmacol Ther. 1998; 12:491–497.

45. Eum JB, Rhee KH, Cho YK, et al. Therapeutic effects of agioⓇ 18 g per day in patients with constipation dominant irritable bowel syndrome and chronic constipation. Korean J Neurogastroenterol Motil. 2008; 14:115–121.
46. Hyland CM, Foran JD. Dioctyl sodium sulphosuccinate as a laxative in the elderly. Practitioner. 1968; 200:698–699.
47. Castle SC, Cantrell M, Israel DS, Samuelson MJ. Constipation prevention: empiric use of stool softeners questioned. Geriatrics. 1991; 46:84–86.
48. Fain AM, Susat R, Herring M, Dorton K. Treatment of constipation in geriatric and chronically ill patients: a comparison. South Med J. 1978; 71:677–680.
49. Chapman RW, Sillery J, Fontana DD, Matthys C, Saunders DR. Effect of oral dioctyl sodium sulfosuccinate on intake-output studies of human small and large intestine. Gastroenterology. 1985; 89:489–493.

50. McRorie JW, Daggy BP, Morel JG, Diersing PS, Miner PB, Robinson M. Psyllium is superior to docusate sodium for treatment of chronic constipation. Aliment Pharmacol Ther. 1998; 12:491–497.

51. Kinnunen O, Salokannel J. Constipation in elderly long-stay patients: its treatment by magnesium hydroxide and bulk-laxative. Ann Clin Res. 1987; 19:321–323.
52. Shin EK, Park SJ, Kim KJ, et al. Effect of combination pretreatment of polyethylene glycol solution and magnesium hydroxide for colonoscopy. Korean J Gastroenterol. 2010; 55:232–236.

53. Andorsky RI, Goldner F. Colonic lavage solution (polyethylene glycol electrolyte lavage solution) as a treatment for chronic constipation: a double-blind, placebo-controlled study. Am J Gastroenterol. 1990; 85:261–265.
54. Klauser AG, Mühldorfer BE, Voderholzer WA, Wenzel G, Müller-Lissner SA. Polyethylene glycol 4000 for slow transit constipation. Z Gastroenterol. 1995; 33:5–8.
55. Corazziari E, Badiali D, Habib FI, et al. Small volume isosmotic polyethylene glycol electrolyte balanced solution (PMF-100) in treatment of chronic nonorganic constipation. Dig Dis Sci. 1996; 41:1636–1642.

56. Freedman MD, Schwartz HJ, Roby R, Fleisher S. Tolerance and efficacy of polyethylene glycol 3350/electrolyte solution versus lactulose in relieving opiate induced constipation: a double-blinded placebo-controlled trial. J Clin Pharmacol. 1997; 37:904–907.

57. Attar A, Lémann M, Ferguson A, et al. Comparison of a low dose polyethylene glycol electrolyte solution with lactulose for treatment of chronic constipation. Gut. 1999; 44:226–230.

58. Bassotti G, Fiorella S, Roselli P, Modesto R. Use of polyethylene glycol solution in slow transit constipation. Ital J Gastroenterol Hepatol. 1999; 31(Suppl 3):S255–S256.
59. Cleveland MV, Flavin DP, Ruben RA, Epstein RM, Clark GE. New polyethylene glycol laxative for treatment of constipation in adults: a randomized, double-blind, placebo-controlled study. South Med J. 2001; 94:478–481.
60. DiPalma JA, DeRidder PH, Orlando RC, Kolts BE, Cleveland MB. A randomized, placebo-controlled, multicenter study of the safety and efficacy of a new polyethylene glycol laxative. Am J Gastroenterol. 2000; 95:446–450.

61. Di Palma JA, Smith JR, Cleveland M. Overnight efficacy of polyethylene glycol laxative. Am J Gastroenterol. 2002; 97:1776–1779.

62. Dipalma JA, Cleveland MV, McGowan J, Herrera JL. A randomized, multicenter, placebo-controlled trial of polyethylene glycol laxative for chronic treatment of chronic constipation. Am J Gastroenterol. 2007; 102:1436–1441.

63. DiPalma JA, Cleveland MB, McGowan J, Herrera JL. A comparison of polyethylene glycol laxative and placebo for relief of constipation from constipating medications. South Med J. 2007; 100:1085–1090.

64. Bae SH. Longterm safety of PEG 4000 in children with chronic functional constipation: A biochemical perspective. Korean J Pediatr. 2010; 53:741–744.

65. Sanders JF. Lactulose syrup assessed in a double-blind study of elderly constipated patients. J Am Geriatr Soc. 1978; 26:236–239.

66. Wesselius-De Casparis A, Braadbaart S, Bergh-Bohlken GE, Mimica M. Treatment of chronic constipation with lactulose syrup: results of a double-blind study. Gut. 1968; 9:84–86.

67. Rouse M, Chapman N, Mahapatra M, Grillage M, Atkinson SN, Prescott P. An open, randomised, parallel group study of lactulose versus ispaghula in the treatment of chronic constipation in adults. Br J Clin Pract. 1991; 45:28–30.
68. Lee JS, Kim YD, Im HH, et al. The effect of lactulose on the colon transit and the various symptoms of constipation in patients with functional constipation. Korean J Neurogastroenterol Motil. 2006; 12:127–135.
69. Stern FH. Constipation–an omnipresent symptom: effect of a preparation containing prune concentrate and cascarin. J Am Geriatr Soc. 1966; 14:1153–1155.

70. Kinnunen O, Winblad I, Koistinen P, Salokannel J. Safety and efficacy of a bulk laxative containing senna versus lactulose in the treatment of chronic constipation in geriatric patients. Pharmacology. 1993; 47(Suppl 1):253–255.

71. Connolly P, Hughes IW, Ryan G. Comparison of "Duphalac" and "irritant" laxatives during and after treatment of chronic constipation: a preliminary study. Curr Med Res Opin. 1974–1975; 2:620–625.
72. Pers M, Pers B. A crossover comparative study with two bulk laxatives. J Int Med Res. 1983; 11:51–53.

73. Williamson J, Coll M, Connolly M. A comparative trial of a new laxative. Nurs Times. 1975; 71:1705–1707.
74. Odes HS, Madar Z. A double-blind trial of a celandin, aloevera and psyllium laxative preparation in adult patients with constipation. Digestion. 1991; 49:65–71.

75. MacLennan WJ, Pooler AFWM. A comparison of sodium picosulphate ("Laxoberal") with standardised senna ("Senokot") in geriatric patients. Curr Med Res Opin. 1974–1975; 2:641–647.

76. Passmore AP, Wilson-Davies K, Stoker C, Scott ME. Chronic constipation in long stay elderly patients: a comparison of lactulose and a senna-fibre combination. BMJ. 1993; 307:769–771.

77. Kasdon SC, Morentin BO. The management of puerperal constipation with a senna preparation. J Int Coll Surg. 1959; 31:455–458.
78. Katz R. Evaluation of standardized senna in the treatment of refractory constipation. Med Press. 1960; 243:335–336.
79. Corman ML. Management of postoperative constipation in anorectal surgery. Dis Colon Rectum. 1979; 22:149–151.

80. DeLuca VA. The management of chronic functional constipation in the severely incapacitated, aged nursing home patient. Med Counterpoint. 1974; 6:25–27.
81. Delegge M, Kaplan R. Efficacy of bowel preparation with the use of a prepackaged, low fibre diet with a low sodium, magnesium citrate cathartic vs. a clear liquid diet with a standard sodium phosphate cathartic. Aliment Pharmacol Ther. 2005; 21:1491–1495.

82. Verghese VJ, Ayub K, Qureshi W, Taupo T, Graham DY. Low-salt bowel cleansing preparation (LoSo Prep) as preparation for colonoscopy: a pilot study. Aliment Pharmacol Ther. 2002; 16:1327–1331.

83. Kienzle-Horn S, Vix JM, Schuijt C, Peil H, Jordan CC, Kamm MA. Efficacy and safety of bisacodyl in the acute treatment of constipation: a double-blind, randomized, placebo-controlled study. Aliment Pharmacol Ther. 2006; 23:1479–1488.

84. Manabe N, Cremonini F, Camilleri M, Sandborn WJ, Burton DD. Effects of bisacodyl on ascending colon emptying and overall colonic transit in healthy volunteers. Aliment Pharmacol Ther. 2009; 30:930–936.

85. Mueller-Lissner S, Kamm MA, Wald A, Hinkel U, Koehler U, Richter EE, Bubeck J. Multicenter, 4-week, double-blind, randomized, placebo-controlled trial of sodium picosulfate in patients with chronic constipation. Am J Gastroenterol. 2010; 105:897–903.

86. Kienzle-Horn S, Vix JM, Schuijt C, Peil H, Jordan CC, Kamm MA. Comparison of bisacodyl and sodium picosulphate in the treatment of chronic constipation. Curr Med Res Opin. 2007; 23:691–699.

87. Kamm MA, Müller-Lissner S, Talley NJ, Tack J, et al. Tegaserod for the treatment of chronic constipation: a randomized, double-blind, placebo-controlled multinational study. Am J Gastroenterol. 2005; 100:362–372.

88. Tack J, Müller-Lissner S, Bytzer P, et al. A randomised controlled trial assessing the efficacy and safety of repeated tegaserod therapy in women with irritable bowel syndrome with constipation. Gut. 2005; 54:1707–1713.

89. Choi H, Yoon SH, Lee WC, et al. Factors affecting the efficacy of tegaserod in patients with chronic constipation. Intest Res. 2007; 5:170–176.
90. Fried M, Johanson JF, Gwee KA, Wagner A, Pecher E, Rueegg P. Efficacy of tegaserod in chronic constipation in men. Am J Gastroenterol. 2007; 102:362–370.

91. Harish K, Hazeena K, Thomas V, Kumar S, Jose T, Narayanan P. Effect of tegaserod on colonic transit time in male patients with constipation-predominant irritable bowel syndrome. J Gastroenterol Hepatol. 2007; 22:1183–1189.

92. Kim YS, Choi SC, Park JM, et al. The effect of tegaserod on symptoms and quality of life in Korean women with irritable bowel syndrome with constipation. J Neurogastroenterol Motil. 2010; 16:61–70.

93. Müller-Lissner S, Kamm MA, Musoglu A, Earnest DL, Dunger-Baldauf C, Shetzline MA. Safety, tolerability, and efficacy of tegaserod over 13 months in patients with chronic constipation. Am J Gastroenterol. 2006; 101:2558–2569.

94. Loughlin J, Quinn S, Rivero E, et al. Tegaserod and the risk of cardiovascular ischemic events: an observational cohort study. J Cardiovasc Pharmacol Ther. 2010; 15:151–157.

95. Camilleri M, Kerstens R, Rykx A, Vandeplassche L. A placebo-controlled trial of prucalopride for severe chronic constipation. N Engl J Med. 2008; 358:2344–2354.

96. Tack J, van Outryve M, Beyens G, Kerstens R, Vandeplassche L. Prucalopride (Resolor) in the treatment of severe chronic constipation in patients dissatisfied with laxatives. Gut. 2009; 58:357–365.

97. Quigley EM, Vandeplassche L, Kerstens R, Ausma J. Clinical trial: the efficacy, impact on quality of life, and safety and tolerability of prucalopride in severe chronic constipation–a 12-week, randomized, double-blind, placebo-controlled study. Aliment Pharmacol Ther. 2009; 29:315–328.
98. Camilleri M, Beyens G, Kerstens R, Robinson P, Vandeplassche L. Safety assessment of prucalopride in elderly patients with constipation: a double-blind, placebo-controlled study. Neurogastroenterol Motil. 2009; 21:1256–e117.

99. Tack J, Middleton SJ, Horne MC, et al. Pilot study of the efficacy of renzapride on gastrointestinal motility and symptoms in patients with constipation-predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2006; 23:1655–1665.

100. George AM, Meyers NL, Hickling RI. Clinical trial: renzapride therapy for constipation-predominant irritable bowel syndrome–multicentre, randomized, placebo-controlled, double-blind study in primary healthcare setting. Aliment Pharmacol Ther. 2008; 27:830–837.
101. Lembo AJ, Cremonini F, Meyers N, Hickling R. Clinical trial: renzapride treatment of women with irritable bowel syndrome and constipation – a double-blind, randomized, placebo-controlled, study. Aliment Pharmacol Ther. 2010; 31:979–990.

102. Camilleri M, Vazquez-Roque MI, Burton D, et al. Pharmacodynamic effects of a novel prokinetic 5-HT receptor agonist, ATI-7505, in humans. Neurogastroenterol Motil. 2007; 19:30–38.
103. Manini ML, Camilleri M, Goldberg M, et al. Effects of Velusetrag (TD-5108) on gastrointestinal transit and bowel function in health and pharmacokinetics in health and constipation. Neurogastroenterol Motil. 2010; 22:42–49.

104. Yang US, Cho JH, Koo JY, et al. The effect of cisapride tartrate in patients with functional dyspepsia and functional constipation. Korean J Gastrointest Motil. 2001; 7:36–46.
105. Emmanuel AV, Kamm MA. Response to a behavioural treatment, biofeedback, in constipated patients is associated with improved gut transit and autonomic innervation. Gut. 2001; 49:214–219.

106. Enck P. Biofeedback training in disordered defecation. A critical review. Dig Dis Sci. 1993; 38:1953–1960.
107. Rao SS, Welcher KD, Pelsang RE. Effects of biofeedback therapy on anorectal function in obstructive defecation. Dig Dis Sci. 1997; 42:2197–2205.
108. Chiarioni G, Salandini L, Whitehead WE. Biofeedback benefits only patients with outlet dysfunction, not patients with isolated slow transit constipation. Gastroenterology. 2005; 129:86–97.

109. Chiotakakou-Faliakou E, Kamm MA, Roy AJ, Storrie JB, Turner IC. Biofeedback provides long-term benefit for patients with intractable, slow and normal transit constipation. Gut. 1998; 42:517–521.
110. Mason HJ, Serrano-Ikkos E, Kamm MA. Psychological state and quality of life in patients having behavioral treatment (biofeedback) for intractable constipation. Am J Gastroenterol. 2002; 97:3154–3159.

111. Rao SS, Valestin J, Brown CK, Zimmerman B, Schulze K. Longterm efficacy of biofeedback therapy for dyssynergic defecation: randomized controlled trial. Am J Gastroenterol. 2010; 105:890–896.

112. Hervé S, Savoye G, Behbahani A, Leroi AM, Denis P, Ducrotté P. Results of 24-h manometric recording of colonic motor activity with endoluminal instillation of bisacodyl in patients with severe chronic slow transit constipation. Neurogastroenterol Motil. 2004; 16:397–402.

113. Jeong JH, Choi JS, Seo YJ, Kim JH. Biofeedback therapy in patients with nonrelaxing puborectalis syndrome: are there differences of therapeutic effect according to methods of diagnosis? J Korean Soc Coloproctol. 2001; 17:26–32.
114. Jung KW, Myung SJ, Byeon JS, et al. Response to biofeedback therapy for patients with rectal hyposensitivity. Intest Res. 2008; 6:56–69.
115. Chang HS, Myung SJ, Yang SK, et al. Electrical stimulation effect in constipated patients with impaired rectal sensation. Korean J Gastrointest Motil. 2002; 8:160–166.
116. Lee BH, Kim N, Kang SB, et al. The long-term clinical efficacy of biofeedback therapy for patients with constipation or fecal incontinence. J Neurogastroenterol Motil. 2010; 16:177–185.

117. Binnie NR, Smith AN, Creasey GH, Edmond P. Constipation associated with chronic spinal cord injury: the effect of pelvic parasympathetic stimulation by the Brindley stimulator. Paraplegia. 1991; 29:463–469.

118. Pescatori M, Meglio M, Cioni B, Colagrande C. Colonic motility in two constipated neurological patients treated by spinal cord stimulation. Wienbeck M, editor. Motility of the digestive tract. New York: Raven;1982. p. 541–546.
119. Ganio E, Masin A, Ratto C, et al. Short-term sacral nerve stimulation for functional anorectal and urinary disturbances: results in 40 patients: evaluation of a new option for anorectal functional disorders. Dis Colon Rectum. 2001; 44:1261–1267.
120. Malouf AJ, Wiesel PH, Nicholls T, Nicholls RJ, Kamm MA. Short-term effects of sacral nerve stimulation for idiopathic slow transit constipation. World J Surg. 2002; 26:166–170.
121. Kenefick NJ, Nicholls RJ, Cohen RG, Kamm MA. Permanent sacral nerve stimulation for treatment of idiopathic constipation. Br J Surg. 2002; 89:882–888.

122. Kenefick NJ, Vaizey CJ, Cohen CR, Nicholls RJ, Kamm MA. Double-blind placebo-controlled crossover study of sacral nerve stimulation for idiopathic constipation. Br J Surg. 2002; 89:1570–1571.

123. Kamm MA, Dudding TC, Melenhorst J, et al. Sacral nerve stimulation for constipation: an international multicentre study. Gastroenterology. 2007; 132(4 Suppl 1):A198.
124. Holzer B, Rosen HR, Novi G, et al. Sacral nerve stimulation in patients with severe constipation. Dis Colon Rectum. 2008; 51:524–529.

125. Vitton V, Roman S, Damon H, Barth X, Mion F. Sacral nerve stimulation and constipation: still a long way to go. Dis Colon Rectum. 2009; 52:752–753.
126. Dinning PG, Fuentealba SE, Kennedy ML, Lubowski DZ, Cook IJ. Sacral nerve stimulation induces pan-colonic propagating pressure waves and increases defecation frequency in patients with slow-transit constipation. Colorectal Dis. 2007; 9:123–132.

127. Song HK, Myung SJ, Yang SK, et al. Noninvasive sacral nerve stimulation for idiopathic slow transit constipation: short term clinical and physiologic effects. Korean J Neurogastroenterol Motil. 2003; 9:134–141.
128. Knowles CH, Scott M, Lunniss PJ. Outcome of colectomy for slow transit constipation. Ann Surg. 1999; 230:627–638.

129. Wexner SD, Daniel N, Jagelman DG. Colectomy for constipation: physiologic investigation is the key to success. Dis Colon Rectum. 1991; 34:851–856.
130. Lee HC, Hong SN, Lee JH, et al. Clinical features and functional outcome of the patients with idiopathic chronic constipation who underwent surgical treatment. Korean J Gastrointest Motil. 2001; 7:204–215.
131. Nam KH, Sohn SK. Clinical results of subtotal colectomy in chronic constipation patients. J Korean Soc Coloproctol. 2000; 16:395–401.
132. Lim SW, Oh SH, Lee WY. Clinical analysis of total colectomy for chronic idiopathic constipation. J Korean Soc Coloproctol. 2000; 16:150–155.
Go to : 
Table 1.
Grading Recommendations7
Table 2.
Summary of Clinical Trials for Prucalopride in Patients with Chronic Constipation




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download