Abstract
Recent advances in chemotherapy lead to improved survival outcomes in patients with colorectal cancer. The 5-fluorouracil (5-FU) is still one of the important chemotherapeutic agents since 1950s, but the introduction of newer cytotoxic agents, irinotecan and oxaliplatin, or targeted agents, bevacizumab and cetuximab, have changed treatment strategies for these patients. A deliberate choice should be made for adjuvant chemotherapy, because it has became complicated more than ever before. Oxaliplatin plus 5-FU seemed to be superior in terms of disease-free and overall survival than 5-FU alone after curative surgery for colon cancers. However not all of these patients seemed to receive benefit from this intensive adjuvant treatment, and some limitations are present according to the postoperative stage, tumor biology and clinical characteristics. For metastatic disease, there is no doubt that more complicated strategies are present because we have more abundant chemotherapeutic agents available for metastatic setting compared to adjuvant setting. Recently, targeted agents, such as bevacizumab or cetuximab, also took an important place in the treatment of metastatic colorectal cancer, and many efforts are also made to find the biomarkers for predicting treatment responses to these targeted agents. In this review, we intended to sort up the standard strategies of chemotherapy for patients with colorectal cancer according to the latest pivotal publications.
REFERENCES
1. O'Connell MJ, Mailliard JA, Kahn MJ, et al. Controlled trial of fluorouracil and low-dose leucovorin given for 6 months as postoperative adjuvant therapy for colon cancer. J Clin Oncol. 1997; 15:246–250.
2. Andre T, Boni C, Navarro M, et al. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol. 2009; 27:3109–3116.
3. Andre T, Boni C, Mounedji-Boudiaf L, et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004; 350:2343–2351.

4. Twelves C, Wong A, Nowacki MP, et al. Capecitabine as adjuvant treatment for stage III colon cancer. N Engl J Med. 2005; 352:2696–2704.

5. Ychou M, Raoul JL, Douillard JY, et al. A phase III randomised trial of LV5FU2+irinotecan versus LV5FU2 alone in adjuvant high-risk colon cancer (FNCLCC Accord02/FFCD-9802). Ann Oncol. 2009; 20:674–680.
6. Saltz LB, Niedzwiecki D, Hollis D, et al. Irinotecan fluorouracil plus leucovorin is not superior to fluorouracil plus leucovorin alone as adjuvant treatment for stage III colon cancer: results of CALGB 89803. J Clin Oncol. 2007; 25:3456–3461.

7. Van Cutsem E, Labianca R, Bodoky G, et al. Randomized phase III trial comparing biweekly infusional fluorouracil/leucovorin alone or with irinotecan in the adjuvant treatment of stage III colon cancer: PETACC-3. J Clin Oncol. 2009; 27:3117–3125.

8. Wolmark N, Yothers G, O'Connell MJ, et al. A phase III trial comparing mFOLFOX6 to mFOLFOX6 plus bevacizumab in stage II or III carcinoma of the colon: results of NSABP Protocol C-08. J Clin Oncol (Meeting Abstracts). 2009; 27.

9. Sauer R, Becker H, Hohenberger W, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004; 351:1731–1740.

10. Gerard JP, Conroy T, Bonnetain F, et al. Preoperative radiotherapy with or without concurrent fluorouracil and leucovorin in T3-4 rectal cancers: results of FFCD 9203. J Clin Oncol. 2006; 24:4620–4625.
11. Bosset JF, Collette L, Calais G, et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med. 2006; 355:1114–1123.

12. Douillard JY, Cunningham D, Roth AD, et al. Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet. 2000; 355:1041–1047.

13. de Gramont A, Figer A, Seymour M, et al. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000; 18:2938–2947.

14. Souglakos J, Androulakis N, Syrigos K, et al. FOLFOXIRI (folinic acid, 5-fluorouracil, oxaliplatin and irinotecan) vs FOLFIRI (folinic acid, 5-fluorouracil and irinotecan) as first-line treatment in metastatic colorectal cancer (MCC): a multicentre randomised phase III trial from the Hellenic Oncology Research Group (HORG). Br J Cancer. 2006; 94:798–805.

15. Falcone A, Ricci S, Brunetti I, et al. Phase III trial of infusional fluorouracil, leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) compared with infusional fluorouracil, leucovorin, and irinotecan (FOLFIRI) as first-line treatment for metastatic colorectal cancer: the Gruppo Oncologico Nord Ovest. J Clin Oncol. 2007; 25:1670–1676.

16. Tournigand C, Andre T, Achille E, et al. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004; 22:229–237.

17. Hochster HS, Hart LL, Ramanathan RK, et al. Safety and efficacy of oxaliplatin and fluoropyrimidine regimens with or without bevacizumab as first-line treatment of metastatic colorectal cancer: results of the TREE study. J Clin Oncol. 2008; 26:3523–3529.

18. Rothenberg ML, Cox JV, Butts C, et al. Capecitabine plus oxaliplatin (XELOX) versus 5-fluorouracil/folinic acid plus oxaliplatin (FOLFOX-4) as second-line therapy in metastatic colorectal cancer: a randomized phase III noninferiority study. Ann Oncol. 2008; 19:1720–1726.

19. Fuchs CS, Marshall J, Mitchell E, et al. Randomized, controlled trial of irinotecan plus infusional, bolus, or oral fluoropyrimidines in first-line treatment of metastatic colorectal cancer: results from the BICC-C study. J Clin Oncol. 2007; 25:4779–4786.

20. Fuchs CS, Marshall J, Barrueco J. Randomized, controlled trial of irinotecan plus infusional, bolus, or oral fluoropyrimidines in first-line treatment of metastatic colorectal cancer: updated results from the BICC-C study. J Clin Oncol. 2008; 26:689–690.

21. Hurwitz H, Fehrenbacher L, Novotny W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004; 350:2335–2342.

22. Saltz LB, Clarke S, Diaz-Rubio E, et al. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol. 2008; 26:2013–2019.

23. Giantonio BJ, Catalano PJ, Meropol NJ, et al. Bevacizumab in combination with oxaliplatin, fluorouracil, and leucovorin (FOLFOX4) for previously treated metastatic colorectal cancer: results from the Eastern Cooperative Oncology Group Study E3200. J Clin Oncol. 2007; 25:1539–1544.

24. Van Cutsem E, Rivera F, Berry S, et al. Safety and efficacy of first-line bevacizumab with FOLFOX, XELOX, FOLFIRI and fluoropyrimidines in metastatic colorectal cancer: the BEAT study. Ann Oncol. 2009; 20:1842–1847.

25. Cunningham D, Humblet Y, Siena S, et al. Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med. 2004; 351:337–345.

26. Sobrero AF, Maurel J, Fehrenbacher L, et al. EPIC: phase III trial of cetuximab plus irinotecan after fluoropyrimidine and oxaliplatin failure in patients with metastatic colorectal cancer. J Clin Oncol. 2008; 26:2311–2319.

27. Van Cutsem E, Kohne CH, Hitre E, et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N Engl J Med. 2009; 360:1408–1417.

28. Bokemeyer C, Bondarenko I, Makhson A, et al. Fluorouracil, leucovorin, and oxaliplatin with and without cetuximab in the first-line treatment of metastatic colorectal cancer. J Clin Oncol. 2009; 27:663–671.

29. Han HS, Chang HJ, Hong YS, Kim SY, Lee KS, Jung KH. Epidermal growth factor receptor expression discrepancies in metastatic colorectal cancer patients treated with cetuximab plus irinotecan-based chemotherapy refractory to irinotecan and oxaliplatin. Dis Colon Rectum. 2009; 52:1144–1151.

30. Chung KY, Shia J, Kemeny NE, et al. Cetuximab shows activity in colorectal cancer patients with tumors that do not express the epidermal growth factor receptor by immunohistochemistry. J Clin Oncol. 2005; 23:1803–1810.

31. De Roock W, Piessevaux H, De Schutter J, et al. KRAS wild-type state predicts survival and is associated to early radiological response in metastatic colorectal cancer treated with cetuximab. Ann Oncol. 2008; 19:508–515.

32. Lievre A, Bachet JB, Le Corre D, et al. KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res. 2006; 66:3992–3995.

33. Dieterle CP, Conzelmann M, Linnemann U, Berger MR. Detection of isolated tumor cells by polymerase chain reaction-restriction fragment length polymorphism for Kras mutations in tissue samples of 199 colorectal cancer patients. Clin Cancer Res. 2004; 10:641–650.

34. Khambata-Ford S, Garrett CR, Meropol NJ, et al. Expression of epiregulin and amphiregulin and Kras mutation status predict disease control in metastatic colorectal cancer patients treated with cetuximab. J Clin Oncol. 2007; 25:3230–3237.

35. Sohn BS, Kim TW, Lee JL, et al. The role of KRAS mutations in predicting the efficacy of cetuximab-plus-irinotecan therapy in irinotecan-refractory korean metastatic colorectal cancer patients. Oncology. 2009; 77:224–230.
36. Di Nicolantonio F, Martini M, Molinari F, et al. Wild-type BRAF is required for response to panitumumab or cetuximab in metastatic colorectal cancer. J Clin Oncol. 2008; 26:57055712.

37. Loupakis F, Pollina L, Stasi I, et al. PTEN expression and KRAS mutations on primary tumors and metastases in the prediction of benefit from cetuximab plus irinotecan for patients with metastatic colorectal cancer. J Clin Oncol. 2009; 27:2622–2629.

Fig. 1.
Improvement of overall survival in patients with metastatic colorectal cancer according to the recent advances in chemotherapy.
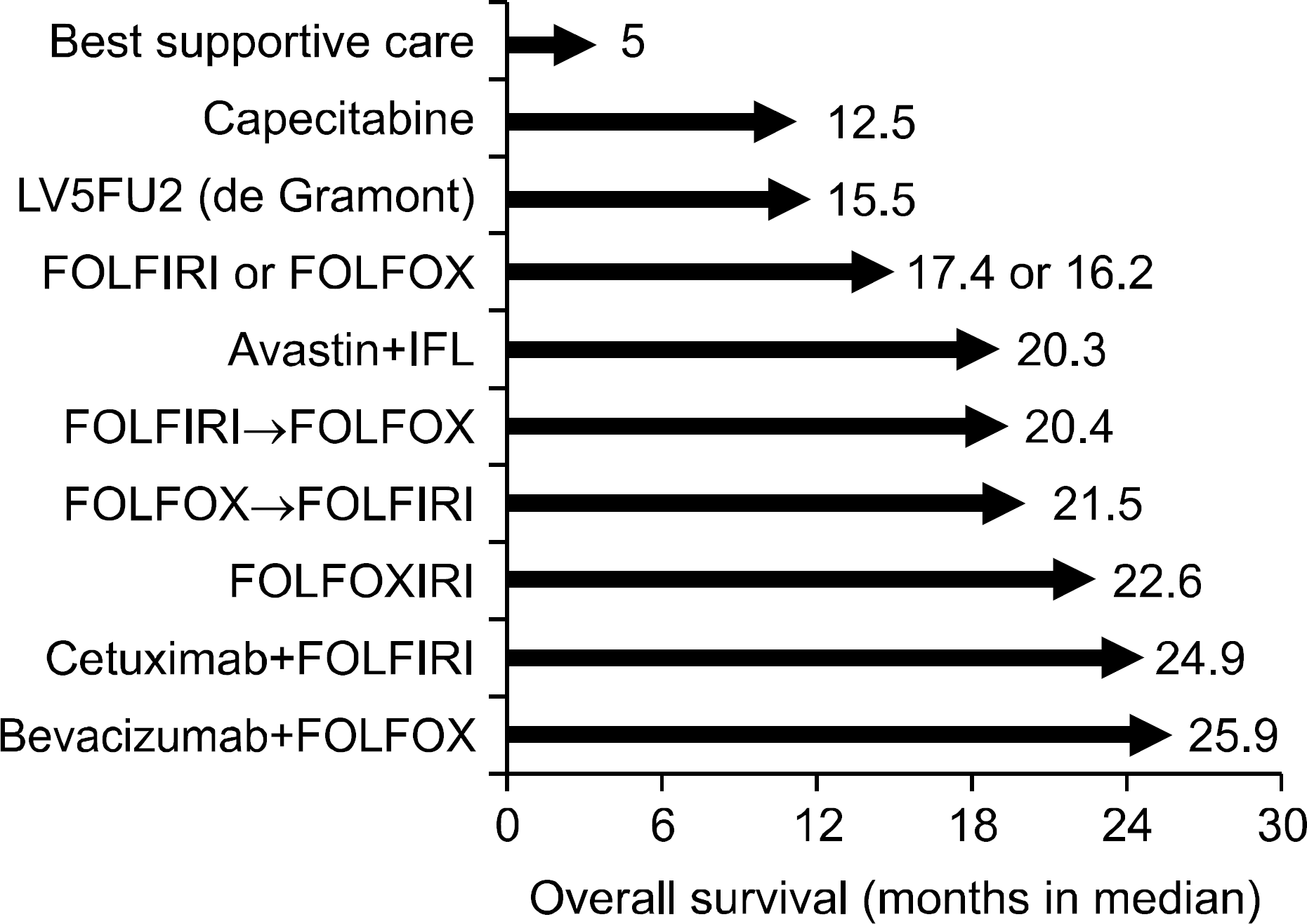
Fig. 2.
Cetuximab and Kras modulate signaling through the epidermal growth factor receptor (EGFR) pathway. (A) Binding of ligand to EGFR triggers signaling through ras/MAPK pathway to multiple targets that may regulate ligand levels. (B) In the presence of cetuximab, ligand binding is prevented, and there is deactivation of EGFR signaling in cells dependent on this pathway. (C) Kras mutations can lead to dysregulation of MAPK pathway and downstream signaling in the absence of ligand-dependent receptor activation.
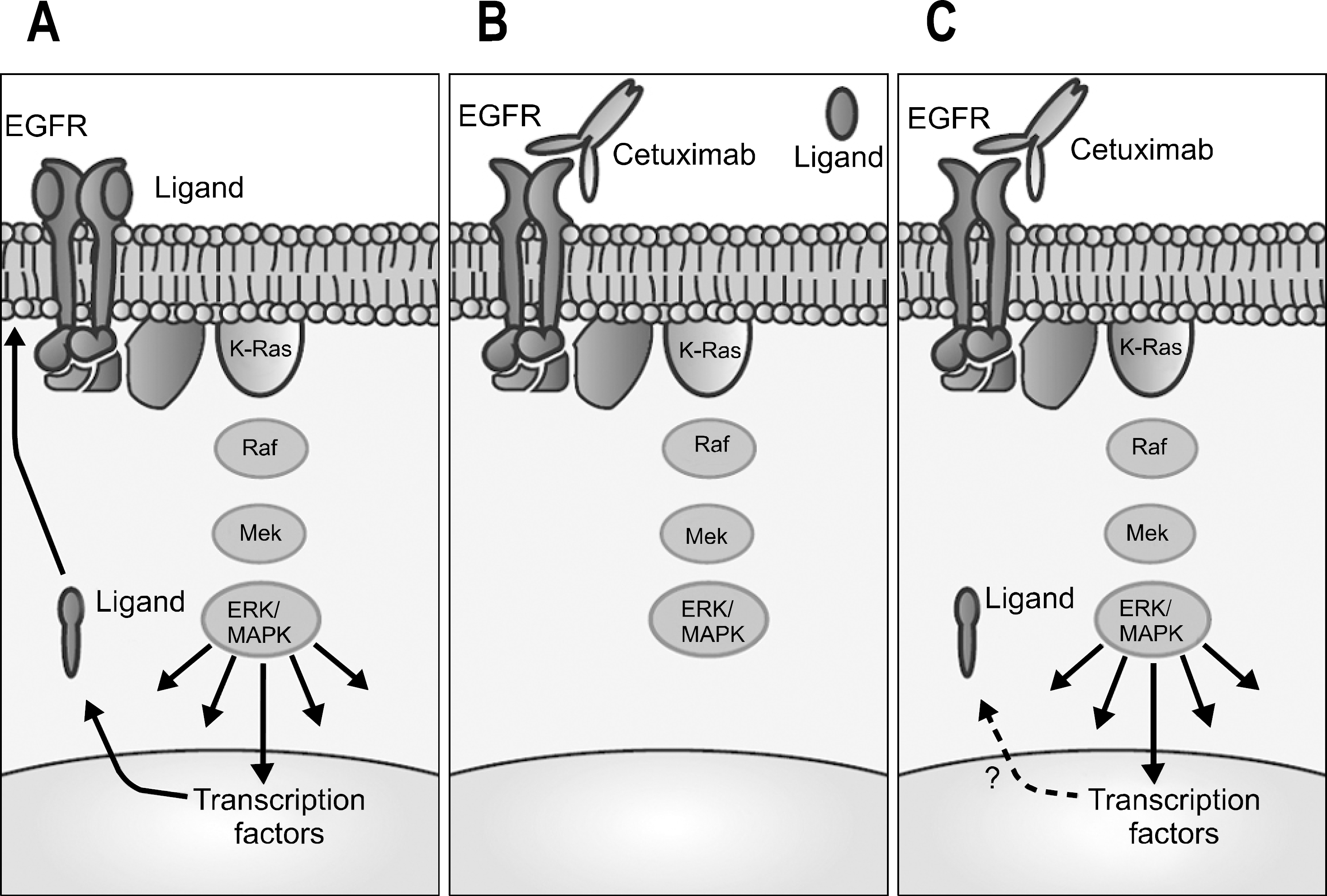
Table 1.
Survival Outcomes in Patients with Stage II and III Colon Cancer (MOSAIC Trial)
DFS, disease-free survival; OS, overall survival; HR, hazard ratio. ∗ FOLFOX did not result in survival benefit in patients with high-risk stage II colon cancers (T4 lesion, perforated, obstructive, poorly differentiated grade of tumor, positive venous invasion, harvested less than 10 lymph nodes during surgery).
Table 2.
FOLFIRI Followed by FOLFOX or the Reverse Sequence in Metastatic Colorectal Cancer, GERCOR Study
Table 3.
Pivotal Randomized Phase III Trials of Bevacizumab in Patients with Metastatic Colorectal Cancers
| Hurwitz et al.21 | Saltz et al.22 | |||||
|---|---|---|---|---|---|---|
| IFL | IFL+Bev | p-value | OxF | OxF+Bev | p-value | |
| RR | 34.8% | 44.8% | 0.004 | 49% | 47% | 0.31 |
| PFS (mo) | 6.2 | 10.6 | <0.001 | 8.0 | 9.4 | 0.0023 |
| OS (mo) | 15.6 | 20.3 | <0.001 | 19.9 | 21.3 | 0.0769 |
Table 4.
Bevacizumab-associated Adverse Events (from the BEAT Study)
| Any grade | Grade 3 or 4 | |
|---|---|---|
| Hypertension | 30% | 5% |
| Proteinuria | 10% | 1% |
| Bleeding | 31% | 3% |
| Wound healing complication | 4% | 1% |
| Arterial thromboembolism | 2% | 1% |
| GI perforation | 2% | 2% |
Table 5.
Different Efficacy Outcomes according to the Kras Mutation Status (CRYSTAL and OPUS Trial)




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download