Abstract
Objective
Alendronate, a widely used bisphosphonates, acts to inhibit bone resorption by interfering with the activity of osteoclasts. Recently, it has been reported that alendronate also may increase bone proliferation and osteoblastic differentiation. However, little is known about mechanism of the action of alendronate on osteoblast differentiation, especially in transcription level. Inhibitors of DNA binding/differentiation (Ids) are helix-loop-helix (HLH) transcription factors and play an important role in BMP-induced osteoblast lineage-specific differentiation. Therefore, this study was aimed to investigate the effect of alendronate on osteoblast differentiation and expression of Id-1 and Id-2.
Methods
MC3T3-E1, pre-osteoblast cell line, were treated with alendronate of various concentrations (10-9 M-10-4 M) and time periods (24, 48 and 72 hours). And then, the effect of alendronate on osteoblast differentiation was examined by alkaline phosphatase (ALP) activity and RT-PCR for osteoblast differentiation markers such as ALP, type 1 collagen (Col 1), and osteocalcin (OCN). The expressions of Id-1 and Id-2 were measured by RT-PCR.
Figures and Tables
Figure 1
Effects of alendronate (Aln) on osteoblastic cell viability and differentiation. Alendronate were treated with various concentrations (10-9 M-10-4 M) for various duration (24, 48, and 72 hours) in MC3T3-E1 cells. (A) Cell viability was determined by MTT assay. (B) For evaluation of osteoblast differentiation, ALP activity was measured at 405 nm. The experiment was repeated three times and each performed with triple samples. *P <0.05, †P <0.01 (vs control).

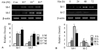
Figure 2
Effect of alendronate (Aln) of different concentrations and times on osteogenic gene marker expression. (A) MC3T3-E1 cells were treated with increasing concentrations (10-6, 10-7 and 10-8 M) for 48 hours. (B) MC3T3-E1 cells were treated with different time periods (24, 48, and 72 hours) at 10-8 M. ALP, Col 1, OCN, and CTSK mRNA levels were detected by RT-PCR with β-actin as loading control. Data shown are representative of three independent experiments. The relative density of each gene was quantitated and statistically analyzed by ImageJ version 1.35d (NIH Image). *P <0.01, †P <0.005 (vs control).
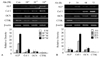
Figure 3
Expression of Id-1 and Id-2 genes during alendronate (Aln)-induced osteoblast differentiation. MC3T3-E1 cells were treated with increasing concentrations (10-6, 10-7 and 10-8 M) for 48 hours (A) and different time periods (24, 48, and 72 hours) at 10-8 M (B). Total RNA was prepared and analyzed for the expression of the Id-1 and Id-2 mRNA by RT-PCR as described. Data shown are representative of four independent experiments. The relative density of each gene was quantitated and statistically analyzed by ImageJ version 1.35d (NIH Image). *P <0.05, †P <0.01, ‡P <0.005 (vs control).
References
1. Katagiri T, Takahashi N. Regulatory mechanisms of osteoblast and osteoclast differentiation. Oral Dis. 2002. 8:147–159.

2. Harada S, Rodan GA. Control of osteoblast function and regulation of bone mass. Nature. 2003. 423:349–355.

3. Suda T, Takahashi N, Martin TJ. Modulation of osteoclast differentiation. Endocr Rev. 1992. 13:66–80.

5. Lane JM, Khan SN, O'Connor WJ, Nydick M, Hommen JP, Schneider R, et al. Bisphosphonate therapy in fibrous dysplasia. Clin Orthop. 2001. 382:6–12.

6. Devogelaer JP. New uses of bisphosphonates: osteogenesis imperfecta. Curr Opin Pharmacol. 2002. 2:748–753.

7. Lehmann HJ, Mouritzen U, Christgau S, Cloos PA, Christiansen C. Effect of bisphosphonates on cartilage turnover assessed with a newly developed assay for collagen type II degradation products. Ann Rheum Dis. 2002. 61:530–533.

8. Maksymowych WP. Bisphosphonates for arthritis - a confusing rationale. J Rheumatol. 2003. 30:430–434.
9. Fromigué O, Body JJ. Bisphosphonates influence the proliferation and the maturation of normal human osteoblasts. J Endocrinol Invest. 2002. 25:539–546.

10. Im GI, Qureshi SA, Kenney J, Rubash HE, Shanbhag AS. Osteoblast proliferation and maturation by bisphosphonates. Biomaterials. 2004. 25:4105–4115.

11. Marie PJ. Transcription factors controlling osteoblastogenesis. Arch Biochem Biophys. 2008. 473:98–105.

12. Malaval L, Liu F, Roche P, Aubin JE. Kinetics of osteoprogenitor proliferation and osteoblast differentiation in vitro. J Cell Biochem. 1999. 74:616–627.

13. Aubin JE, Liu F, Malaval L, Gupta AK. Osteoblast and chondroblast differentiation. Bone. 1995. 17:2 suppl. 77S–83S.

14. Yamaguchi A, Komori T, Suda T. Regulation of osteoblast differentiation mediated by bone morphogenetic proteins, hedgehogs, and Cbfa1. Endocr Rev. 2000. 21:393–411.

15. Peng Y, Kang Q, Luo Q, Jiang W, Si W, Liu BA, et al. Inhibitor of DNA binding/differentiation helix-loop-helix proteins mediate bone morphogenetic protein-induced osteoblast differentiation of mesenchymal stem cells. J Biol Chem. 2004. 279:32941–32949.

16. Giuliani N, Girasole G, Pedrazzoni M, Passeri G, Gatti C, Passeri M. Alendronate stimulates b-FGF productin and mineralized nodule formation in hman osteoblastic cells and osteoblastogenesis in human bone marrow cultures. J Bone Miner Res. 1995. 10:Suppl. S171.
17. Kang Y, Chen CR, Massagué J. A self-enabling TGFbeta response coupled to stress signaling: Smad engages stress response factor ATF3 for Id1 repression in epithelial cells. Mol Cell. 2003. 11:915–926.
18. Kreider BL, Benezra R, Rovera G, Kadesch T. Inhibition of myeloid differentiation by the helix-loop-helix protein Id. Science. 1992. 255:1700–1702.

19. Ruzinova MB, Benezra R. Id proteins in development, cell cycle and cancer. Trends Cell Biol. 2003. 13:410–418.

21. García-Moreno C, Serrano S, Nacher M, Farré M, Díez A, Mariñoso ML, et al. Effect of alendronate on cultured normal human osteoblasts. Bone. 1998. 22:233–239.

22. Sama AA, Khan SN, Myers ER, Huang RC, Cammisa FP, Sandhu HS, et al. High-dose alendronate uncouples osteoclast and osteoblast function: a study in a rat spine pseudarthrosis model. Clin Orthop Relat Res. 2004. 425:135–142.
23. Klein BY, Ben-Bassat H, Breuer E, Solomon V, Golomb G. Structurally different bisphosphonates exert opposing effects on alkaline phosphatase and mineralization in marrow osteoprogenitors. J Cell Biochem. 1998. 68:186–194.

24. Reinholz GG, Getz B, Pederson L, Sanders ES, Subramaniam M, Ingle JN, et al. Bisphosphonates directly regulate cell proliferation, differentiation, and gene expression in human osteoblasts. Cancer Res. 2000. 60:6001–6007.
26. Einhorn TA. The cell and fracture healing. Clin Orthop Ralat Res. 1988. 355 suppl. S7–S21.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download